10.1016/S0040-4039(01)85858-5
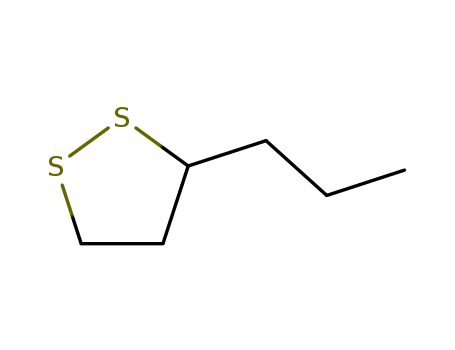
The study investigates the major malodorous component from the anal gland secretion of the male stoat (Mustela erminea). Through gas chromatographic analysis of a diethyl ether extract of the viscous yellow fluid from the anal glands, four volatile components were identified, with 2-propylthietane being the major and most volatile one (50%). The chemical structure of 2-propylthietane was determined based on its mass spectral data (m/e 116,101,87,82,74,73,69,67) and proton magnetic resonance (p.m.r.) spectrum, which showed a substituted thietane with a tertiary hydrogen adjacent to the sulphur atom and four other hydrogens on the ring. The compound was synthesized by desulphurisation of 3-propyl-1,2-dithiolane with tris(diethylamino) phosphine, and the mass spectrum, retention times, and p.m.r. spectra of the synthetic compound matched those of the natural substance. The study also mentions that the sulphur-containing constituents of the anal gland secretion from the mink are likely derived from an isoprenoid biosynthesis pathway, but 2-propylthietane from the stoat does not appear to be so derived. The nature of the remaining components and those from the female stoat are still under investigation.