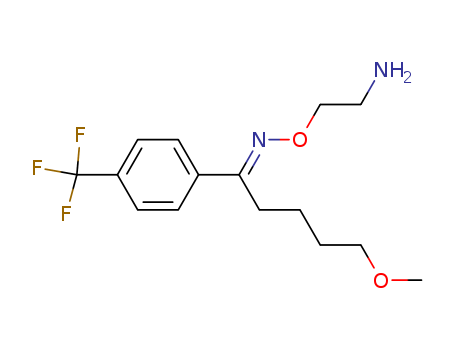
- Chemical Name:Fluvoxamine
- CAS No.:54739-18-3
- Molecular Formula:C15H21F3N2O2
- Molecular Weight:318.339
- Hs Code.:2928000090
- European Community (EC) Number:611-193-1
- UNII:O4L1XPO44W
- DSSTox Substance ID:DTXSID2044002
- Nikkaji Number:J1.245.131H,J453.182E,J11.936I
- Wikipedia:Fluvoxamine
- Wikidata:Q27259474
- NCI Thesaurus Code:C61769
- RXCUI:42355
- Pharos Ligand ID:CH1FF2M6Z9W4
- Metabolomics Workbench ID:42584
- ChEMBL ID:CHEMBL814
- Mol file:54739-18-3.mol
Synonyms:Desiflu;DU 23000;DU-23000;DU23000;Dumirox;Faverin;Fevarin;Floxyfral;Fluvoxadura;Fluvoxamin AL;Fluvoxamin beta;Fluvoxamin neuraxpharm;Fluvoxamin ratiopharm;Fluvoxamin Stada;Fluvoxamin-neuraxpharm;Fluvoxamin-ratiopharm;Fluvoxamina Geminis;Fluvoxamine;Fluvoxamine Maleate;Fluvoxamine Maleate, (E)-Isomer;Fluvoxamine, (Z)-Isomer;Geminis, Fluvoxamina;Luvox;Novo Fluvoxamine;Novo-Fluvoxamine;Nu Fluvoxamine;Nu-Fluvoxamine;PMS Fluvoxamine;PMS-Fluvoxamine;ratio Fluvoxamine;ratio-Fluvoxamine