10.1039/c6ra05385h
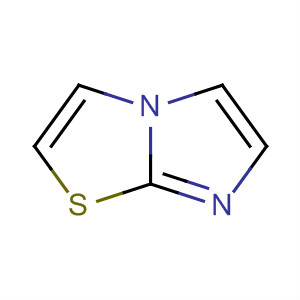
The study presents a visible light promoted, catalyst-free synthesis of thiazoles and imidazo[2,1-b]thiazoles in an EtOH:H2O green medium. The key chemicals involved are phenacyl bromide, N-phenylthiourea, and 2-aminothiazole. Phenacyl bromide serves as a reactant that undergoes homolytic fission of its C-Br bond under visible light, generating a free radical. N-phenylthiourea also forms a free radical through the homolytic fission of its S-H bond. These radicals combine to form intermediate compounds, which further react to produce the desired thiazoles and imidazo[2,1-b]thiazoles via cyclization and removal of a water molecule. The use of visible light provides the activation energy needed for the reaction, eliminating the need for catalysts or photosensitizers. The EtOH:H2O solvent system enhances the solubility of the reactants and stabilizes the transition states, contributing to the eco-efficiency and high yield of the products. This method is notable for its cost-effectiveness, short reaction time, and alignment with green chemistry principles.