Products Categories
| CAS No.: | 123846-66-2 |
|---|---|
| Name: | 5-Nitro-2-pyridineacetonitrile |
| Article Data: | 7 |
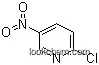
| Molecular Structure: | |
|
|
|
| Formula: | C7H5N3O2 |
| Molecular Weight: | 163.136 |
| Synonyms: | 2-(5-Nitropyridin-2-yl)acetonitrile;2-(5-nitropyridin-2-yl)acetonitrile;2-(5-Nitro-2-pyridinyl)acetonitrile; |
| Density: | 1.352 g/cm3 |
| Melting Point: | 66.0-66.5 °C |
| Boiling Point: | 342.237 °C at 760 mmHg |
| Flash Point: | 160.78 °C |
| PSA: | 82.50000 |
| LogP: | 1.57908 |
- 144851-82-1METHYL2-AMINO-3-FLUOROBENZOATE
- 483366-12-7(2S,4R)-1-Boc-2-cyano-4-hydroxypyrrolidine
- 173606-50-3BOC-10-AMINODECANOIC ACID
- 361456-36-2METHYL (R)-(+)-ISOCYANATO-3-PHENYLPROPI&
- 5156-58-1N-(1-Benzyl-4-pipperidinyl)-N-phenylpropanamide HCl
- 81281-59-67-Benzylideneaminotheophylline
- 50288-62-5threo-Phenyl-2-piperidyl acetamide
- 82993-81-5D-threo-Ritalinic acid hydrochloride
- 47087-37-6Z-D-Glu-OMe
- 73441-42-6METHYL-5-CHLORO-2,2-DIMETHYLVALERATE
- 914223-27-1
tert-butyl 2-cyano-2-(5-nitropyridin-2-yl)acetate

- 123846-66-2
2-(5-nitropyridin-2-yl)acetonitrile

| Conditions | Yield |
|---|---|
| With toluene-4-sulfonic acid In toluene for 2h; Reflux; | 76.3% |
| With hydrogenchloride In ethanol at 80℃; for 4h; | 42% |
| With toluene-4-sulfonic acid In toluene for 2h; Heating; Yield given; | |
| With toluene-4-sulfonic acid In toluene for 2h; Heating; | 4.95 g |
| With hydrogenchloride In ethanol at 80℃; for 4h; |
- 4548-45-2
2-chloro-5-nitropyridine

- 1116-98-9
cyanoacetic acid tert-butyl ester

- 123846-66-2
2-(5-nitropyridin-2-yl)acetonitrile

| Conditions | Yield |
|---|---|
| Stage #1: 2-chloro-5-nitropyridine; cyanoacetic acid tert-butyl ester With potassium carbonate In tetrahydrofuran at 20℃; for 20h; Molecular sieve; Heating / reflux; Stage #2: With toluene-4-sulfonic acid In toluene at 20℃; for 18h; Heating / reflux; | 66% |
- 5418-51-9
5-nitro-2-hydroxypyridine

- 123846-66-2
2-(5-nitropyridin-2-yl)acetonitrile

| Conditions | Yield |
|---|---|
| Multi-step reaction with 3 steps 1: 66 percent / POCl3; PCl5 / 3 h / 110 °C 2: K2CO3 / tetrahydrofuran / 24 h / Heating 3: 4.95 g / p-TsOH / toluene / 2 h / Heating View Scheme |
- 4214-76-0
5-nitro-pyridin-2-ylamine

- 123846-66-2
2-(5-nitropyridin-2-yl)acetonitrile

| Conditions | Yield |
|---|---|
| Multi-step reaction with 4 steps 1: 77 percent / aq. H2SO4; NaNO2 / 0 - 20 °C 2: 66 percent / POCl3; PCl5 / 3 h / 110 °C 3: K2CO3 / tetrahydrofuran / 24 h / Heating 4: 4.95 g / p-TsOH / toluene / 2 h / Heating View Scheme |
- 4548-45-2
2-chloro-5-nitropyridine

- 123846-66-2
2-(5-nitropyridin-2-yl)acetonitrile

| Conditions | Yield |
|---|---|
| Multi-step reaction with 2 steps 1: K2CO3 / tetrahydrofuran / 24 h / Heating 2: 4.95 g / p-TsOH / toluene / 2 h / Heating View Scheme | |
| Multi-step reaction with 2 steps 1: potassium carbonate / tetrahydrofuran / 20 °C / Reflux 2: hydrogenchloride / ethanol / 4 h / 80 °C View Scheme | |
| Multi-step reaction with 2 steps 1: potassium carbonate / tetrahydrofuran / 70 °C 2: toluene-4-sulfonic acid / toluene / 2 h / Reflux View Scheme | |
| Multi-step reaction with 2 steps 1: potassium carbonate / tetrahydrofuran / 20 °C / Reflux 2: hydrogenchloride / ethanol / 4 h / 80 °C View Scheme |

| Conditions | Yield |
|---|---|
| Multi-step reaction with 2 steps 1: K2CO3 / tetrahydrofuran / 24 h / Heating 2: TsOH / toluene / 2 h / Heating View Scheme |
- 123846-66-2
2-(5-nitropyridin-2-yl)acetonitrile

- 100-10-7
4-dimethylamino-benzaldehyde

| Conditions | Yield |
|---|---|
| With piperidine; acetic acid In toluene Heating; | 90% |
- 123846-66-2
2-(5-nitropyridin-2-yl)acetonitrile

- 74-88-4
methyl iodide

- 1256633-31-4
2-methyl-2-(5-nitropyridin-2-yl)-propanenitrile

| Conditions | Yield |
|---|---|
| Stage #1: 2-(5-nitropyridin-2-yl)acetonitrile With sodium hydride In N,N-dimethyl-formamide at -15 - -10℃; for 1h; Stage #2: methyl iodide In N,N-dimethyl-formamide at -15 - 25℃; | 83% |
| With potassium carbonate In acetonitrile at 20 - 40℃; | 48% |
| Stage #1: 2-(5-nitropyridin-2-yl)acetonitrile With sodium hydride In tetrahydrofuran at 0℃; for 0.5h; Stage #2: methyl iodide In tetrahydrofuran at 20℃; for 24h; | |
| Stage #1: 2-(5-nitropyridin-2-yl)acetonitrile With sodium hydride In tetrahydrofuran at 0℃; for 0.5h; Stage #2: methyl iodide In tetrahydrofuran at 0 - 20℃; for 24h; |
- 123846-66-2
2-(5-nitropyridin-2-yl)acetonitrile

- 883993-15-5
2-(5-amino-2-pyridyl)acetonitrile

| Conditions | Yield |
|---|---|
| With palladium 10% on activated carbon; hydrogen In tetrahydrofuran at 20℃; under 7500.75 Torr; for 2h; | 82.8% |
| With hydrogenchloride; tin(ll) chloride In ethanol; water at 0 - 20℃; for 2h; | 74% |
- 123846-66-2
2-(5-nitropyridin-2-yl)acetonitrile

- 105-07-7
4-cyanobenzaldehyde

| Conditions | Yield |
|---|---|
| With piperidine; acetic acid In toluene Heating; | 71% |

What can I do for you?
Get Best Price
Specification
The 5-Nitro-2-pyridineacetonitrile with cas registry number of 123846-66-2, has the systematic name of (5-nitropyridin-2-yl)acetonitrile. And it is also named 2-pyridineacetonitrile, 5-nitro-.
Physical properties about this chemical are: (1)ACD/LogP: -0.11; (2)# of Rule of 5 Violations: 0; (3)ACD/LogD (pH 5.5): 0; (4)ACD/LogD (pH 7.4): 0; (5)ACD/BCF (pH 5.5): 1; (6)ACD/BCF (pH 7.4): 1; (7)ACD/KOC (pH 5.5): 18; (8)ACD/KOC (pH 7.4): 18; (9)#H bond acceptors: 5; (10)#H bond donors: 0; (11)#Freely Rotating Bonds: 2; (12)Polar Surface Area: 82.5 Å2; (13)Index of Refraction: 1.583; (14)Molar Refractivity: 40.352 cm3; (15)Molar Volume: 120.699 cm3; (16)Polarizability: 15.997×10-24cm3; (17)Surface Tension: 64.306 dyne/cm; (18)Enthalpy of Vaporization: 58.597 kJ/mol; (19)Vapour Pressure: 0 mmHg at 25°C.
You can still convert the following datas into molecular structure:
(1)SMILES: N#CCc1ccc(cn1)[N+]([O-])=O;
(2)InChI: InChI=1/C7H5N3O2/c8-4-3-6-1-2-7(5-9-6)10(11)12/h1-2,5H,3H2;
(3)InChIKey: PHCKNSBYAFTJKZ-UHFFFAOYAF;
(4)Std. InChI: InChI=1S/C7H5N3O2/c8-4-3-6-1-2-7(5-9-6)10(11)12/h1-2,5H,3H2;
(5)Std. InChIKey: PHCKNSBYAFTJKZ-UHFFFAOYSA-N