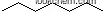Products Categories
| CAS No.: | 149-57-5 |
|---|---|
| Name: | 2-Ethylhexanoic acid |
| Article Data: | 123 |
| Molecular Structure: | |
|
|
|
| Formula: | C8H16O2 |
| Molecular Weight: | 144.214 |
| Synonyms: | Caproicacid, a-ethyl- (4CI);2-Butylbutanoic acid;2-Ethyl-1-hexanoic acid;3-Heptanecarboxylic acid;NSC8881;iso-Caprylic acid; |
| EINECS: | 205-743-6 |
| Density: | 0.926 g/cm3 |
| Melting Point: | -59 °C |
| Boiling Point: | 228 °C at 760 mmHg |
| Flash Point: | 116.6 °C |
| Solubility: | water: 2 g/L (20 °C ) |
| Appearance: | colourless liquid |
| Hazard Symbols: |
 Xn Xn
|
| Risk Codes: | 63 |
| Safety: | 36/37 |
| Transport Information: | UN 3265 8/PG 2 |
| PSA: | 37.30000 |
| LogP: | 2.28740 |
- 483366-12-7(2S,4R)-1-Boc-2-cyano-4-hydroxypyrrolidine
- 173606-50-3BOC-10-AMINODECANOIC ACID
- 361456-36-2METHYL (R)-(+)-ISOCYANATO-3-PHENYLPROPI&
- 5156-58-1N-(1-Benzyl-4-pipperidinyl)-N-phenylpropanamide HCl
- 81281-59-67-Benzylideneaminotheophylline
- 50288-62-5threo-Phenyl-2-piperidyl acetamide
- 82993-81-5D-threo-Ritalinic acid hydrochloride
- 47087-37-6Z-D-Glu-OMe
- 73441-42-6METHYL-5-CHLORO-2,2-DIMETHYLVALERATE
- 211914-50-0N-[[2-[[[4-(Aminoiminomethyl)phenyl]amino]methyl]-1-methyl-1H-benzimidazol-5-yl]carbonyl]-N-(2-pyridinyl)-beta-alanine ethyl ester hydrochloride

| Conditions | Yield |
|---|---|
| With copper acetylacetonate; oxygen; sodium hydroxide; 1,3-bis(2,4,6-trimethylphenyl)-2-imidazolidinylidene In water at 50℃; under 760.051 Torr; for 12h; Sealed tube; | 99% |
| With C43H56IO4P; potassium acetate; caesium carbonate at 35℃; for 6h; Reagent/catalyst; Cooling; | 99.1% |
| With oxygen; copper(II) acetate monohydrate; cobalt(II) diacetate tetrahydrate In water at 40℃; under 760.051 Torr; for 3h; | 94% |

| Conditions | Yield |
|---|---|
| With toluene-4-sulfonic acid In water; acetone at 100℃; under 11103.3 Torr; for 4h; | 98% |

- 149-57-5
2-Ethylhexanoic acid

| Conditions | Yield |
|---|---|
| With iron(III) chloride In dichloromethane at 20℃; for 0.0833333h; Inert atmosphere; Green chemistry; | 96% |

- 149-57-5
2-Ethylhexanoic acid

| Conditions | Yield |
|---|---|
| With dimethylfumarate at 48℃; for 3h; Temperature; | 96% |
- 62251-85-8
4-Ethyl-3-oxo-2-(triphenyl-λ5-phosphanylidene)-octanoic acid ethyl ester

A
- 149-57-5
2-Ethylhexanoic acid

B
- 791-28-6
Triphenylphosphine oxide

| Conditions | Yield |
|---|---|
| With sodium hydroxide; sodium hypochlorite In tetrahydrofuran at 25℃; for 19h; | A 93% B n/a |
- 1426824-91-0
2-methoxybenzyl 2-ethylhexanoate

- 149-57-5
2-Ethylhexanoic acid

| Conditions | Yield |
|---|---|
| With N1,N1,N12,N12-tetramethyl-7,8-dihydro-6H-dipyrido[1,2-a:2,1'-c][1,4]diazepine-2,12-diamine In N,N-dimethyl-formamide for 24h; Inert atmosphere; Glovebox; UV-irradiation; | 91% |

| Conditions | Yield |
|---|---|
| With sulfuric acid; water; oxygen In water at 60℃; under 2280.15 Torr; for 16h; pH=1; | A 36% B 90% |

- 1426824-92-1
2-ethoxybenzyl 2-ethylhexanoate

- 149-57-5
2-Ethylhexanoic acid

| Conditions | Yield |
|---|---|
| With N1,N1,N12,N12-tetramethyl-7,8-dihydro-6H-dipyrido[1,2-a:2,1'-c][1,4]diazepine-2,12-diamine In N,N-dimethyl-formamide for 24h; Inert atmosphere; Glovebox; UV-irradiation; | 89% |

| Conditions | Yield |
|---|---|
| at 70℃; for 7h; Product distribution; electrolysis: nickel net anode, cylindrical stainless steel cathode; electrolyte: 1M NaOH/water; variation of temperature and reaction time; | 76% |
| at 70℃; for 7h; electrolysis: nickel net anode, cylindrical stainless steel cathode; electrolyte: 1M NaOH/H2O; | 76% |
| With sodium tungstate; dihydrogen peroxide In water at 90℃; for 4h; | 68% |
- 68439-49-6Alcohols C16-18 ethoxylated
- 1191-15-7Aluminum,hydrobis(2-methylpropyl)-
- 841205-47-8Propanamide, 3-(4-cyanophenoxy)-N-[4-cyano-3-(trifluoromethyl)phenyl]-2-hydroxy-2-methyl-, (2S)-
- 598-62-9Manganese carbonate
- 79-04-9Acetyl chloride,2-chloro-
- 1314-80-3Phosphorus pentasulfide
- 182349-12-8Rupatadine fumarate
- 90-72-2Tris(dimethylaminomethyl)phenol
- 104987-11-3Tacrolimus
- 141-53-7Sodium formate
- 8001-54-5Quaternary ammonium compounds, alkylbenzyldimethyl, chlorides
- 9003-39-8Povidone
- 10161-34-9Trenbolone acetate
- 402957-28-2Telaprevir
- 68-19-9Cyanocobalamin
Consensus Reports
Standards and Recommendations
Specification
The Butyl ethyl acetic acid is an organic compound with the formula C8H16O2. The IUPAC name of this chemical is 2-ethylhexanoic acid. With the CAS registry number 149-57-5, it is also named as hexanoic acid, 2-ethyl-. The product's category is Industrial/Fine Chemicals. Besides, it is colourless liquid, which should be stored in a closed cool and dry palce.
The main use of Butyl ethyl acetic acid is to prepare a dryer for coatings and paints. It is also used as a heat stabilizer of polyvinyl chloride plastics. It is still an important medical material of carbenicillin. Besides, it is used in synthesis of dyes and fragrance.
Physical properties about Butyl ethyl acetic acid are: (1)ACD/LogP: 2.72; (2)ACD/LogD (pH 5.5): 1.96; (3)ACD/LogD (pH 7.4): 0.17; (4)ACD/BCF (pH 5.5): 11.85; (5)ACD/BCF (pH 7.4): 1; (6)ACD/KOC (pH 5.5): 123.98; (7)ACD/KOC (pH 7.4): 2.01; (8)#H bond acceptors: 2; (9)#H bond donors: 1; (10)#Freely Rotating Bonds: 5; (11)Polar Surface Area: 26.3 Å2; (12)Index of Refraction: 1.435; (13)Molar Refractivity: 40.63 cm3; (14)Molar Volume: 155.5 cm3; (15)Polarizability: 16.1×10-24cm3; (16)Surface Tension: 31.8 dyne/cm; (17)Density: 0.926 g/cm3; (18)Flash Point: 116.6 °C; (19)Enthalpy of Vaporization: 51.18 kJ/mol; (20)Boiling Point: 228 °C at 760 mmHg; (21)Vapour Pressure: 0.027 mmHg at 25°C.
Preparation: this chemical can be prepared by 2-ethyl-hexan-1-ol. This reaction will need reagent KMnO4 and alkali.

Uses of Butyl ethyl acetic acid: it can be used to produce 2-ethyl-hexanoic acid-anhydride. It will need reagent acetic acid anhydride and sulfuric acid.

When you are using this chemical, please be cautious about it as the following:
This chemical is possible risk of harm to the unborn child. When you are using it, wear suitable protective clothing and gloves.
You can still convert the following datas into molecular structure:
(1)SMILES: O=C(O)C(CC)CCCC
(2)InChI: InChI=1/C8H16O2/c1-3-5-6-7(4-2)8(9)10/h7H,3-6H2,1-2H3,(H,9,10)
(3)InChIKey: OBETXYAYXDNJHR-UHFFFAOYAO
(4)Std. InChI: InChI=1S/C8H16O2/c1-3-5-6-7(4-2)8(9)10/h7H,3-6H2,1-2H3,(H,9,10)
(5)Std. InChIKey: OBETXYAYXDNJHR-UHFFFAOYSA-N
The toxicity data is as follows:
| Organism | Test Type | Route | Reported Dose (Normalized Dose) | Effect | Source |
|---|---|---|---|---|---|
| guinea pig | LD50 | skin | 6300uL/kg (6.3mL/kg) | Journal of Industrial Hygiene and Toxicology. Vol. 26, Pg. 269, 1944. | |
| rabbit | LD50 | skin | 1260uL/kg (1.26mL/kg) | Union Carbide Data Sheet. Vol. 11/4/1971, | |
| rat | LC | inhalation | > 400ppm/6H (400ppm) | Kodak Company Reports. Vol. 21MAY1971, | |
| rat | LD50 | oral | 3gm/kg (3000mg/kg) | Journal of Industrial Hygiene and Toxicology. Vol. 26, Pg. 269, 1944. |