Products Categories
Basic Information
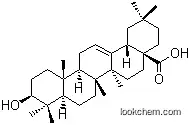
| CAS No.: | 218600-53-4 |
|---|---|
| Name: | Bardoxolone methyl |
| Article Data: | 11 |
| Molecular Structure: | |
|
|
|
| Formula: | C32H43 N O4 |
| Molecular Weight: | 505.698 |
| Synonyms: | 2-Cyano-3,12-dioxooleana-1,9(11)-dien-28-oicacid methyl ester; Bardoxolone methyl; CDDO Methylester; CDDO-Me; Methyl 2-cyano-3,12-dioxoleana-1,9(11)-dien-28-oate; NSC713200; RTA 402 |
| Density: | 1.15g/cm3 |
| Melting Point: | 215-223°C |
| Boiling Point: | 600.8°Cat760mmHg |
| Flash Point: | 256.5°C |
| PSA: | 84.23000 |
| LogP: | 6.37898 |
Related products
- 483366-12-7(2S,4R)-1-Boc-2-cyano-4-hydroxypyrrolidine
- 361456-36-2METHYL (R)-(+)-ISOCYANATO-3-PHENYLPROPI&
- 5156-58-1N-(1-Benzyl-4-pipperidinyl)-N-phenylpropanamide HCl
- 81281-59-67-Benzylideneaminotheophylline
- 50288-62-5threo-Phenyl-2-piperidyl acetamide
- 82993-81-5D-threo-Ritalinic acid hydrochloride
- 47087-37-6Z-D-Glu-OMe
- 73441-42-6METHYL-5-CHLORO-2,2-DIMETHYLVALERATE
- 240444-70-62,3,5,6-Tetrafluor-4-(methoxymethyl)benzyl-2,2-dimethyl-3-(prop-1-en-1-yl)cyclopropancarboxylat
- 68439-39-4Poly(oxy-1,2-ethanediyl), alpha-(2-ethylhexyl)-omega-hydroxy-,
Synthetic route
- 1428550-98-4
2-bromo-3,12-dioxo-oleanane-1,9 (11)-diene-28-carboxylic acid methyl ester

- 218600-53-4
bardoxolone methyl
Conditions

| Conditions | Yield |
|---|---|
| With potassium iodide In N,N-dimethyl-formamide at 120℃; for 30h; | 83.6% |
| With potassium iodide In N,N-dimethyl-formamide at 120℃; | 73% |
| With potassium iodide In N,N-dimethyl-formamide at 120℃; | 73% |
| With potassium iodide In N,N-dimethyl-formamide at 120℃; for 24h; Temperature; Time; Reagent/catalyst; Inert atmosphere; | 66% |
Conditions

| Conditions | Yield |
|---|---|
| Stage #1: bardoxolone With oxalyl dichloride; N,N-dimethyl-formamide In dichloromethane at 20℃; for 1.16667h; Cooling with ice; Stage #2: methanol In dichloromethane for 1h; | 96% |

- 218600-53-4
bardoxolone methyl
Conditions

| Conditions | Yield |
|---|---|
| With 2,3-dicyano-5,6-dichloro-p-benzoquinone In benzene for 0.5h; Reflux; Inert atmosphere; | 87% |
| Stage #1: methyl 2-cyano-3,12-dioxoolean-9(11)-en-28-oate With Phenylselenyl chloride In ethyl acetate Stage #2: With dihydrogen peroxide In tetrahydrofuran; water | 40% |
| With Phenylselenyl chloride; dihydrogen peroxide 1.) ethyl acetate, 2.) THF; Yield given; Multistep reaction; |
Conditions

| Conditions | Yield |
|---|---|
| With potassium iodide In N,N-dimethyl-formamide at 120℃; for 16h; Inert atmosphere; | 89% |
- 305818-40-0
1,2-dihydro-2-cyano-3,12-dioxooleana-1,9(11)-dien-28-oic acid methyl ester

- 218600-53-4
bardoxolone methyl
Conditions

| Conditions | Yield |
|---|---|
| With 2,3-dicyano-5,6-dichloro-p-benzoquinone In benzene for 0.5h; Heating; | 92% |

- 218600-53-4
bardoxolone methyl
Conditions

| Conditions | Yield |
|---|---|
| With porcine liver esterase; γ-glutamyl transpeptidase In aq. buffer at 37℃; for 8h; pH=7.4; Enzymatic reaction; | 88% |
- 508-02-1
Oleanolic acid

- 218600-53-4
bardoxolone methyl
Conditions

| Conditions | Yield |
|---|---|
| Multi-step reaction with 4 steps 1.1: potassium carbonate / N,N-dimethyl-formamide / 24 h / 0 - 20 °C / Inert atmosphere 2.1: 1-hydroxy-3H-benz[d][1,2]iodoxole-1,3-dione; fluorobenzene / dimethyl sulfoxide / 24 h / 85 °C / Inert atmosphere 3.1: 3-chloro-benzenecarboperoxoic acid / dichloromethane / 24 h / 0 - 20 °C / Inert atmosphere 3.2: 24 h / 35 °C / Inert atmosphere 4.1: potassium iodide / N,N-dimethyl-formamide / 24 h / 120 °C / Inert atmosphere View Scheme | |
| Multi-step reaction with 4 steps 1.1: potassium carbonate / N,N-dimethyl-formamide / 24 h / 0 - 20 °C / Inert atmosphere 2.1: 1-hydroxy-3H-benz[d][1,2]iodoxole-1,3-dione; fluorobenzene / dimethyl sulfoxide / 24 h / 85 °C / Inert atmosphere 3.1: 3-chloro-benzenecarboperoxoic acid / dichloromethane / 24 h / 0 - 20 °C / Inert atmosphere 3.2: 90 °C / Inert atmosphere; Darkness 3.3: 18 h / 37 °C / Inert atmosphere 4.1: potassium iodide / N,N-dimethyl-formamide / 16 h / 120 °C / Inert atmosphere View Scheme | |
| Multi-step reaction with 4 steps 1.1: potassium carbonate / N,N-dimethyl-formamide / 24 h / 0 - 20 °C / Inert atmosphere 2.1: 1-hydroxy-3H-benz[d][1,2]iodoxole-1,3-dione; fluorobenzene / dimethyl sulfoxide / 24 h / 85 °C / Inert atmosphere 3.1: 3-chloro-benzenecarboperoxoic acid / dichloromethane / 24 h / 0 - 20 °C / Inert atmosphere 3.2: 90 °C / Inert atmosphere; Darkness 4.1: potassium iodide / N,N-dimethyl-formamide / 16 h / 120 °C / Inert atmosphere View Scheme |
- 69660-90-8
(4aS,6aS,6bR,8aR,12aR,12bR,14bS)-methyl2,2,6a,6b,9,9,12a-heptamethyl-10-oxo-1,2,3,4,4a,5,6,6a,6b,7,8,8a,9,10,12a,12b,13,14b-octadecahydropicene-4a-carboxylate

- 218600-53-4
bardoxolone methyl
Conditions

| Conditions | Yield |
|---|---|
| Multi-step reaction with 2 steps 1.1: 3-chloro-benzenecarboperoxoic acid / dichloromethane / 24 h / 0 - 20 °C / Inert atmosphere 1.2: 24 h / 35 °C / Inert atmosphere 2.1: potassium iodide / N,N-dimethyl-formamide / 24 h / 120 °C / Inert atmosphere View Scheme | |
| Multi-step reaction with 2 steps 1.1: 3-chloro-benzenecarboperoxoic acid / dichloromethane / 24 h / 0 - 20 °C / Inert atmosphere 1.2: 90 °C / Inert atmosphere; Darkness 1.3: 18 h / 37 °C / Inert atmosphere 2.1: potassium iodide / N,N-dimethyl-formamide / 16 h / 120 °C / Inert atmosphere View Scheme | |
| Multi-step reaction with 3 steps 1: 3-chloro-benzenecarboperoxoic acid / dichloromethane 2: hydrogen bromide; bromine; acetic acid 3: potassium iodide / N,N-dimethyl-formamide / 120 °C View Scheme | |
| Multi-step reaction with 3 steps 1: 3-chloro-benzenecarboperoxoic acid / dichloromethane 2: hydrogen bromide; bromine; acetic acid 3: potassium iodide / N,N-dimethyl-formamide / 120 °C View Scheme | |
| Multi-step reaction with 2 steps 1.1: 3-chloro-benzenecarboperoxoic acid; hydrogen bromide / dichloromethane / 26 h / 20 °C 1.2: 24 h / 35 °C 2.1: potassium iodide / N,N-dimethyl-formamide / 30 h / 120 °C View Scheme |
- 1724-17-0, 73584-64-2
oleanolic acid methyl ester

- 218600-53-4
bardoxolone methyl
Conditions

| Conditions | Yield |
|---|---|
| Multi-step reaction with 3 steps 1.1: 1-hydroxy-3H-benz[d][1,2]iodoxole-1,3-dione; fluorobenzene / dimethyl sulfoxide / 24 h / 85 °C / Inert atmosphere 2.1: 3-chloro-benzenecarboperoxoic acid / dichloromethane / 24 h / 0 - 20 °C / Inert atmosphere 2.2: 24 h / 35 °C / Inert atmosphere 3.1: potassium iodide / N,N-dimethyl-formamide / 24 h / 120 °C / Inert atmosphere View Scheme | |
| Multi-step reaction with 3 steps 1.1: 1-hydroxy-3H-benz[d][1,2]iodoxole-1,3-dione; fluorobenzene / dimethyl sulfoxide / 24 h / 85 °C / Inert atmosphere 2.1: 3-chloro-benzenecarboperoxoic acid / dichloromethane / 24 h / 0 - 20 °C / Inert atmosphere 2.2: 90 °C / Inert atmosphere; Darkness 2.3: 18 h / 37 °C / Inert atmosphere 3.1: potassium iodide / N,N-dimethyl-formamide / 16 h / 120 °C / Inert atmosphere View Scheme | |
| Multi-step reaction with 3 steps 1.1: 1-hydroxy-3H-benz[d][1,2]iodoxole-1,3-dione; fluorobenzene / dimethyl sulfoxide / 24 h / 85 °C / Inert atmosphere 2.1: 3-chloro-benzenecarboperoxoic acid / dichloromethane / 24 h / 0 - 20 °C / Inert atmosphere 2.2: 90 °C / Inert atmosphere; Darkness 3.1: potassium iodide / N,N-dimethyl-formamide / 16 h / 120 °C / Inert atmosphere View Scheme |
- 65023-20-3
methyl 3β-acetoxy-12-oxoolean-9(11)-en-28-oate

- 218600-53-4
bardoxolone methyl
Conditions

| Conditions | Yield |
|---|---|
| Multi-step reaction with 6 steps 1: 97 percent / methanolic KOH / 0.5 h / Heating 2: 92 percent / Jones reagent / acetone / 0.17 h / 20 °C 3: 99 percent / NaOMe / benzene / 2 h / 20 °C 4: 66 percent / NH2OH*HCl / ethanol; H2O / 1 h / Heating 5: 100 percent / NaOMe / methanol; diethyl ether / 0.75 h / 20 °C 6: 92 percent / DDQ / benzene / 0.5 h / Heating View Scheme | |
| Multi-step reaction with 6 steps 1: aq. KOH / methanol 2: CrO3, H2SO4 3: 100 percent / sodium methoxide / benzene 4: 61 percent / NH2OH*HCl / aq. ethanol 5: 100 percent / sodium methoxide / methanol; diethyl ether 6: 1.) phenylselenenyl chloride, 2.) 30 percent aq. H2O2 / 1.) ethyl acetate, 2.) THF View Scheme | |
| Multi-step reaction with 5 steps 1: potassium hydroxide; water / methanol 2: sodium methylate / benzene 3: hydroxylamine hydrochloride / water; ethanol 4: sodium methylate / methanol; diethyl ether 5: Phenylselenyl chloride / ethyl acetate View Scheme | |
| Multi-step reaction with 4 steps 1.1: potassium hydroxide / methanol / 1 h / Reflux 2.1: chromium(VI) oxide; sulfuric acid / water; acetone / 0 - 20 °C 3.1: lithium diisopropyl amide / tetrahydrofuran; n-heptane; ethylbenzene / 0.33 h / -78 - 20 °C / Inert atmosphere 3.2: 0.08 h / -78 - 20 °C 4.1: 2,3-dicyano-5,6-dichloro-p-benzoquinone / benzene / 0.5 h / Reflux; Inert atmosphere View Scheme |
Raw Materials
Downstream Products
Hot Products
- 37296-80-3Colestipol, hydrochloride
- 116-02-93,3,5-Trimethylcyclohexanol
- 149845-06-7Saquinavir mesylate
- 7721-01-9Tantalum chloride(TaCl5)
- 583-91-52-Hydroxy-4-(methylthio)butanoic acid
- 128517-07-7Cyclo[(2Z)-2-amino-2-butenoyl-L-valyl-(3S,4E)-3-hydroxy-7-mercapto-4-heptenoyl-D-valyl-D-cysteinyl],cyclic (3®5)-disulfide
- 136-53-8Hexanoic acid,2-ethyl-, zinc salt (2:1)
- 23504-03-21,3-Benzenediol,4-(1-methylethyl)-
- 104987-11-3Tacrolimus
- 141-53-7Sodium formate
- 8001-54-5Quaternary ammonium compounds, alkylbenzyldimethyl, chlorides
- 9003-39-8Povidone
- 10161-34-9Trenbolone acetate
- 402957-28-2Telaprevir
- 68-19-9Cyanocobalamin