Products Categories
| CAS No.: | 58-63-9 |
|---|---|
| Name: | Inosine |
| Article Data: | 92 |
| Molecular Structure: | |
|
|
|
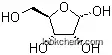
| Formula: | C10H12N4O5 |
| Molecular Weight: | 268.229 |
| Synonyms: | 9-[(2R,3R,4S,5R)-3,4-dihydroxy-5-(hydroxymethyl)oxolan-2-yl]-3H-purin-6-one;4-acetamidobenzoic acid; 1-[(2R,4S,5S)-4-azido-5-(hydroxymethyl)oxolan-2-yl]-5-methyl-pyrimidine-2,4-dione; 9-[(2R,3R,4R,5R)-3,4-dihydroxy-5-(hydroxymethyl)oxolan-2-yl]-3H-purin-6-one; 1-dimethylaminopropan-2-ol;6-Hydroxy-9-(β-D-ribofuranosyl)-9H-purine;2',3',5'–Triacetylinosine;iso-prinosine;Inosine (8CI,9CI);hypoxanthine-ribose;.beta.-Inosine;Inosina [INN-Spanish];9-.beta.-D-Ribofuranosylhypoxanthine;Hypoxanthine 9-beta-D-ribofuranoside;Oxiamin;556-08-1;Hypoxanthine, 9-beta-D-ribofuranosyl-;Inosine-Pranobex;Ribonosine;(-)-Inosine;Inotin (TN);Hypoxanthine ribonucleoside;9H-purin-6-ol, 9-pentofuranosyl-;9-[(2S,3R,4S,5R)-3,4-dihydroxy-5-(hydroxymethyl)oxolan-2-yl]-3H-purin-6-one;Inosine [INN:JAN];Atorel;9-[(2R,3R,4R,5R)-3,4-dihydroxy-5-(hydroxymethyl)oxolan-2-yl]-3H-purin-6-one;Inosine (JAN);1,9-Dihydro-9-beta-D-ribofuranosyl-6H-purin-6-one;beta-D-Ribofuranoside, hypoxanthine-9; |
| EINECS: | 200-390-4 |
| Density: | 2.085 g/cm3 |
| Melting Point: | 222-226 °C (dec.)(lit.) |
| Boiling Point: | 732.815 °C at 760 mmHg |
| Flash Point: | 396.993 °C |
| Solubility: | 2.1 g/100 mL (20 °C) in water |
| Appearance: | White crystalline powder |
| Hazard Symbols: |
 Xi Xi
|
| Risk Codes: | 36/37/38 |
| Safety: | 24/25-36-26 |
| PSA: | 133.49000 |
| LogP: | -2.26890 |
- 81281-59-67-Benzylideneaminotheophylline
- 82993-81-5D-threo-Ritalinic acid hydrochloride
- 73441-42-6METHYL-5-CHLORO-2,2-DIMETHYLVALERATE
- 68439-39-4Poly(oxy-1,2-ethanediyl), alpha-(2-ethylhexyl)-omega-hydroxy-,
- 852475-26-4MC1568
- 958254-66-51H-Imidazo[4,5-b]pyridine-2-carboxaldehyde, 1-methyl-, hydrochloride
- 99170-93-1N-Methyl-2-oxazolamine
- 717878-06-31-(4-fluorophenyl)-4-nitro-1H-imidazole
- 914458-26-7[5-(2-fluorophenyl)-1-pentyl-1H-pyrrol-3-yl]-1-naphthalenyl-Methanone
- 894852-01-87-BROMO-2,2-DIMETHYL-2H-PYRIDO[3,2-B][1,4]OXAZIN-3(4H)-ONE

| Conditions | Yield |
|---|---|
| Multistep reaction; |

| Conditions | Yield |
|---|---|
| With sodium phosphate buffer; adenosine deaminase In water at 27℃; pH=7.5; Enzyme kinetics; | |
| With adenosine deaminase Kinetics; Concentration; Enzymatic reaction; | |
| Multi-step reaction with 2 steps 1: purine nucleoside phosphorylase from aeromonas hydrophila II / aq. phosphate buffer / 20 °C / pH 7.5 / Enzymatic reaction 2: aq. phosphate buffer / 20 °C / pH 7.5 View Scheme | |
| With adenine deaminase from E. coli; water In aq. phosphate buffer at 25℃; pH=7.0; Catalytic behavior; Reagent/catalyst; Enzymatic reaction; |
- 131-99-7, 21138-48-7, 21214-07-3, 54656-49-4, 56553-00-5
inosine-5'-monophosphate

- 58-63-9
inosine

| Conditions | Yield |
|---|---|
| With recombinant nucleoside acid phosphatase/phosphotransferase from Escherichia blattae at 30℃; for 0.166667h; pH=5; Kinetics; Time; pH-value; Temperature; Reagent/catalyst; aq. acetate buffer; |

| Conditions | Yield |
|---|---|
| Multi-step reaction with 2 steps 1.1: potassium phosphate; recombinant Escherichia coli uridine phosphorylase / water / 72 h / 50 °C / pH 7 / Enzymatic reaction 1.2: 24 h / 4 °C 2.1: recombinant Escherichia coli purine nucleoside phosphorylase / 20 °C / pH 7 / Enzymatic reaction View Scheme |

| Conditions | Yield |
|---|---|
| Multi-step reaction with 2 steps 1: purine nucleoside phosphorylase from aeromonas hydrophila II / aq. phosphate buffer / 20 °C / pH 7.5 / Enzymatic reaction 2: aq. phosphate buffer / 20 °C / pH 7.5 View Scheme |
- 99790-49-5
α-D-ribose-1-phosphate

- 58-63-9
inosine

| Conditions | Yield |
|---|---|
| Multi-step reaction with 3 steps 1: aq. phosphate buffer / 20 °C / pH 7.5 2: purine nucleoside phosphorylase from aeromonas hydrophila II / aq. phosphate buffer / 20 °C / pH 7.5 / Enzymatic reaction 3: aq. phosphate buffer / 20 °C / pH 7.5 View Scheme | |
| Multi-step reaction with 3 steps 1: aq. phosphate buffer / 20 °C / pH 7.5 2: purine nucleoside phosphorylase from aeromonas hydrophila II / aq. phosphate buffer / 20 °C / pH 7.5 / Enzymatic reaction 3: aq. phosphate buffer / 20 °C / pH 7.5 View Scheme |

| Conditions | Yield |
|---|---|
| In aq. phosphate buffer at 20℃; pH=7.5; |

| Conditions | Yield |
|---|---|
| With recombinant Escherichia coli purine nucleoside phosphorylase at 20℃; pH=7; Kinetics; Reagent/catalyst; Enzymatic reaction; |

| Conditions | Yield |
|---|---|
| With recombinant Escherichia coli purine nucleoside phosphorylase; recombinant Escherichia coli uridine phosphorylase at 20℃; pH=7; Enzymatic reaction; |

| Conditions | Yield |
|---|---|
| With Clostridium perfringens uridine phosphorylase; Aeromonas hydrophila purine nucleosidephosphorylase co-immobilized on glyoxyl-agarose In aq. phosphate buffer at 28℃; under 1034.32 Torr; pH=7.5; Flow reactor; Green chemistry; Enzymatic reaction; |
- 149-73-5Trimethoxymethane
- 72287-26-4[1,1'-Bis(diphenylphosphino)ferrocene]dichloropalladium(II)
- 5934-29-2L-Histidine hydrochloride monohydrate
- 2627-86-3Benzenemethanamine, a-methyl-, (aS)-
- 107-91-52-Cyanoacetamide
- 432-60-0Allylestrenol
- 50910-55-92-Amino-3,5-dibromobenzaldehyde
- 491878-06-9Doripenem side-chain
- 104987-11-3Tacrolimus
- 141-53-7Sodium formate
- 8001-54-5Quaternary ammonium compounds, alkylbenzyldimethyl, chlorides
- 9003-39-8Povidone
- 10161-34-9Trenbolone acetate
- 402957-28-2Telaprevir
- 68-19-9Cyanocobalamin
Consensus Reports
Specification
The Inosine, with the CAS registry number 58-63-9 and EINECS registry number 200-390-4, has the IUPAC name of 9-[(2R,3R,4S,5R)-3,4-dihydroxy-5-(hydroxymethyl)oxolan-2-yl]-3H-purin-6-one. It is a kind of white crystalline powder, and belongs to the following product categories: Nucleotides and Nucleosides; Antivirals for Research and Experimental Use; Biochemistry; Nucleosides, Nucleotides & Related Reagents; Nucleic acids; Bases & Related Reagents. And the molecular formula of the chemical is C10H12N4O5.
The Inosine can be found in tRNAs and is essential for proper translation of the genetic code in wobble base pairs. It is an intermediate in a chain of purine nucleotides reactions required for muscle movements. And it has neuroprotective properties. Besides, it is classified as an antiviral in the Anatomical Therapeutic Chemical Classification System. What's more, it can be produced by Straight-Dough Method.
The physical properties of Inosine are as followings: (1)# of Rule of 5 Violations: 0; (2)ACD/BCF (pH 5.5): 1; (3)ACD/BCF (pH 7.4): 1; (4)ACD/KOC (pH 5.5): 2.004; (5)ACD/KOC (pH 7.4): 2.02; (6)#H bond acceptors: 9; (7)#H bond donors: 4; (8)#Freely Rotating Bonds: 5; (9)Polar Surface Area: 129.2 Å2; (10)Index of Refraction: 1.879; (11)Molar Refractivity: 58.896 cm3; (12)Molar Volume: 128.666 cm3; (13)Polarizability: 23.348×10-24cm3; (14)Surface Tension: 104.426 dyne/cm; (15)Density: 2.085 g/cm3; (16)Flash Point: 396.993 °C; (17)Enthalpy of Vaporization: 112.251 kJ/mol; (18)Boiling Point: 732.815 °C at 760 mmHg; (19)Vapour Pressure: 0 mmHg at 25°C.
You should be cautious while dealing with this chemical. It irritates to eyes, respiratory system and skin. Therefore, you had better take the following instructions: Avoid contact with skin and eyes. Wear suitable protective clothing, and if in case of contacting with eyes, rinse immediately with plenty of water and seek medical advice.
You can still convert the following datas into molecular structure:
(1)SMILES: c1[nH]c2c(c(=O)n1)ncn2[C@H]3[C@@H]([C@@H]([C@H](O3)CO)O)O
(2)InChI: InChI=1/C10H12N4O5/c15-1-4-6(16)7(17)10(19-4)14-3-13-5-8(14)11-2-12-9(5)18/h2-4,6-7,10,15-17H,1H2,(H,11,12,18)/t4-,6-,7-,10-/m1/s1
(3)InChIKey: UGQMRVRMYYASKQ-KQYNXXCUBA
The toxicity data is as follows:
| Organism | Test Type | Route | Reported Dose (Normalized Dose) | Effect | Source |
|---|---|---|---|---|---|
| mouse | LD50 | intraperitoneal | 3175mg/kg (3175mg/kg) | Pharmaceutical Chemistry Journal Vol. 20, Pg. 160, 1986. | |
| mouse | LD50 | intravenous | > 2800mg/kg (2800mg/kg) | Drugs in Japan Vol. -, Pg. 156, 1995. | |
| mouse | LD50 | oral | > 20gm/kg (20000mg/kg) | Drugs in Japan Vol. 6, Pg. 77, 1982. | |
| mouse | LD50 | subcutaneous | 5gm/kg (5000mg/kg) | Drugs in Japan Vol. 6, Pg. 77, 1982. | |
| rat | LD50 | intraperitoneal | 2900mg/kg (2900mg/kg) | Drugs in Japan Vol. 6, Pg. 77, 1982. | |
| rat | LD50 | intravenous | > 2gm/kg (2000mg/kg) | Drugs in Japan Vol. 6, Pg. 77, 1982. | |
| rat | LD50 | oral | > 10gm/kg (10000mg/kg) | Drugs in Japan Vol. -, Pg. 156, 1995. |