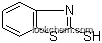Products Categories
| CAS No.: | 6295-57-4 |
|---|---|
| Name: | (2-BENZOTHIAZOLYLTHIO)ACETIC ACID |
| Article Data: | 31 |
| Molecular Structure: | |
|
|
|
| Formula: | C9H7NO2S2 |
| Molecular Weight: | 225.292 |
| Synonyms: | Aceticacid, (2-benzothiazolylthio)- (6CI,7CI,8CI,9CI);(2-Benzothiazolylthio)aceticacid;2-(2-Benzothiazolylthio)acetic acid;2-Carboxymethylthiobenzothiazole;NSC 11891;NSC 4443;S-2-Benzothiazolylthioglycolic acid;Sanbit ABT; |
| EINECS: | 228-565-0 |
| Density: | 1.51 g/cm3 |
| Melting Point: | 159°C |
| Boiling Point: | 423.4 °C at 760 mmHg |
| Flash Point: | 209.9 °C |
| Solubility: | 133mg/L at 20℃ |
| Hazard Symbols: |
 Xi Xi
|
| Risk Codes: | 36/37/38 |
| Safety: | 26-36/37/39 |
| PSA: | 103.73000 |
| LogP: | 2.47300 |
- 81281-59-67-Benzylideneaminotheophylline
- 82993-81-5D-threo-Ritalinic acid hydrochloride
- 73441-42-6METHYL-5-CHLORO-2,2-DIMETHYLVALERATE
- 68439-39-4Poly(oxy-1,2-ethanediyl), alpha-(2-ethylhexyl)-omega-hydroxy-,
- 852475-26-4MC1568
- 958254-66-51H-Imidazo[4,5-b]pyridine-2-carboxaldehyde, 1-methyl-, hydrochloride
- 99170-93-1N-Methyl-2-oxazolamine
- 717878-06-31-(4-fluorophenyl)-4-nitro-1H-imidazole
- 914458-26-7[5-(2-fluorophenyl)-1-pentyl-1H-pyrrol-3-yl]-1-naphthalenyl-Methanone
- 894852-01-87-BROMO-2,2-DIMETHYL-2H-PYRIDO[3,2-B][1,4]OXAZIN-3(4H)-ONE
- 24044-88-0
ethyl 2-(benzo[d]thiazol-2-ylthio)acetate

- 6295-57-4
2-benzthiazole thioacetic acid

| Conditions | Yield |
|---|---|
| With water; sodium hydroxide In methanol at 20℃; for 19h; | 94% |
| With sodium hydroxide | |
| With hydroxide |

| Conditions | Yield |
|---|---|
| With potassium hydroxide In ethanol at 20℃; for 3h; | 89% |
| With triethylamine In N,N-dimethyl-formamide | |
| In methanol at 20℃; pH=Ca.8; |

| Conditions | Yield |
|---|---|
| With sodium hydroxide In water at 90℃; for 2h; Alkylation; | 85% |
| Stage #1: 2-Mercaptobenzothiazole; chloroacetic acid With potassium hydroxide In methanol; water for 5h; Reflux; Stage #2: With hydrogenchloride In methanol; water | 85% |
| Stage #1: 2-Mercaptobenzothiazole With potassium hydroxide In methanol for 1h; Stage #2: chloroacetic acid In methanol for 8h; Reflux; | 83% |
- 149-30-4
2-thioxo-3H-1,3-benzothiazole

- 79-11-8
chloroacetic acid

- 6295-57-4
2-benzthiazole thioacetic acid

| Conditions | Yield |
|---|---|
| With sodium hydroxide | |
| With sodium monochloroacetic acid; sodium hydrogencarbonate | |
| With sodium hydroxide | |
| With sodium carbonate | |
| With sodium hydroxide |
- 7778-70-3
potassium 2-mercaptobenzothiazolate

- 7748-25-6
potassium chloroacetate

- 6295-57-4
2-benzthiazole thioacetic acid

| Conditions | Yield |
|---|---|
| With ethanol |

| Conditions | Yield |
|---|---|
| With aluminum oxide In acetone microwave irradiation; |

| Conditions | Yield |
|---|---|
| Multi-step reaction with 2 steps 1: dimethylformamide / 3 h / Heating 2: OH- View Scheme |

| Conditions | Yield |
|---|---|
| Multi-step reaction with 2 steps 1: aqueous (?) KOH 2: aqueous NaOH View Scheme |

| Conditions | Yield |
|---|---|
| Multi-step reaction with 2 steps 1.1: potassium carbonate / N,N-dimethyl-formamide / 0.5 h / 20 °C 1.2: 15 h / 20 °C 2.1: water; lithium hydroxide / methanol; tetrahydrofuran / 7 h / 0 - 20 °C View Scheme | |
| Multi-step reaction with 2 steps 1.1: potassium carbonate / N,N-dimethyl-formamide / 0.5 h / 20 °C 1.2: 15 h / 20 °C 2.1: lithium hydroxide; water / methanol; tetrahydrofuran / 7 h / 0 - 20 °C View Scheme | |
| Multi-step reaction with 2 steps 1: potassium hydroxide / ethanol / 30 °C 2: water; sodium hydroxide / methanol; tetrahydrofuran / 20 °C View Scheme |

| Conditions | Yield |
|---|---|
| Multi-step reaction with 2 steps 1: potassium hydroxide / ethanol / 1 h 2: potassium carbonate / N,N-dimethyl-formamide / 1 h View Scheme |

What can I do for you?
Get Best Price
Consensus Reports
Reported in EPA TSCA Inventory.
Specification
The 2-Carboxymethylthiobenzothiazole is an organic compound with the formula C9H7NO2S2. The IUPAC name of this chemical is 2-(1,3-benzothiazol-2-ylsulfanyl)acetic acid. With the CAS registry number 6295-57-4, it is also named as (1,3-benzothiazol-2-ylsulfanyl)acetic acid. The product's categories are Benzothiazole; Aliphatics; Carboxylic Acids.
Physical properties about 2-Carboxymethylthiobenzothiazole are: (1)ACD/LogP: 2.74; (2)ACD/LogD (pH 5.5): 0.39; (3)ACD/LogD (pH 7.4): -0.9; (4)ACD/BCF (pH 5.5): 1; (5)ACD/BCF (pH 7.4): 1; (6)ACD/KOC (pH 5.5): 3.27; (7)ACD/KOC (pH 7.4): 1; (8)#H bond acceptors: 3; (9)#H bond donors: 1; (10)#Freely Rotating Bonds: 3; (11)Polar Surface Area: 92.73 Å2; (12)Index of Refraction: 1.727; (13)Molar Refractivity: 59.17 cm3; (14)Molar Volume: 148.6 cm3; (15)Polarizability: 23.45×10-24cm3; (16)Surface Tension: 79.3 dyne/cm; (17)Density: 1.51 g/cm3; (18)Flash Point: 209.9 °C; (19)Enthalpy of Vaporization: 71.45 kJ/mol; (20)Boiling Point: 423.4 °C at 760 mmHg; (21)Vapour Pressure: 6.34E-08 mmHg at 25°C.
Preparation: this chemical can be prepared by 3H-benzothiazole-2-thione and chloroacetic acid. This reaction will need reagent alkaline medium.
.gif)
Uses of 2-Carboxymethylthiobenzothiazole: it can be used to produce (benzothiazol-2-ylsulfanyl)-acetic acid; compound with morpholine. This reaction is a kind of salt formation. It will need reagent diethyl ether. The yield is about 90%.
.gif)
When you are using this chemical, please be cautious about it as the following:
This chemical is irritating to eyes, respiratory system and skin. When you are using it, wear suitable gloves and eye/face protection. In case of contact with eyes, rinse immediately with plenty of water and seek medical advice.
You can still convert the following datas into molecular structure:
(1)SMILES: O=C(O)CSc1nc2ccccc2s1
(2)InChI: InChI=1/C9H7NO2S2/c11-8(12)5-13-9-10-6-3-1-2-4-7(6)14-9/h1-4H,5H2,(H,11,12)
(3)InChIKey: ZZUQWNYNSKJLPI-UHFFFAOYAI
(4)Std. InChI: InChI=1S/C9H7NO2S2/c11-8(12)5-13-9-10-6-3-1-2-4-7(6)14-9/h1-4H,5H2,(H,11,12)
(5)Std. InChIKey: ZZUQWNYNSKJLPI-UHFFFAOYSA-N