Products Categories
| CAS No.: | 83-34-1 |
|---|---|
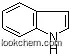
| Name: | 3-Methyl-1H-indole |
| Article Data: | 262 |
| Molecular Structure: | |
|
|
|
| Formula: | C9H9N |
| Molecular Weight: | 131.177 |
| Synonyms: | 1/C9H9N/c1-7-6-10-9-5-3-2-4-8(7)9/h2-6,10H,1H;3-Methyl-4,5-benzopyrrole;3-methyl-2,3-dihydro-1H-indole;3-MI;1H-Indole, 3-methyl-;Skatol;beta-Methylindole;Indole, 3-methyl-;1H-Indole,3-methyl-;Scatole;3-Methyl-1H-indole;FEMA No. 3019;.beta.-Methylindole;3-methyl-H-indole;3-methyl indole (Skatole);Skatole; |
| EINECS: | 201-471-7 |
| Density: | 1.11 g/cm3 |
| Melting Point: | 92-97 °C(lit.) |
| Boiling Point: | 265.1 °C at 760 mmHg |
| Flash Point: | 112.5 °C |
| Appearance: | slightly brown platelets |
| Hazard Symbols: |
 Xi Xi
|
| Risk Codes: | 36/37/38 |
| Safety: | 26-36 |
| PSA: | 15.79000 |
| LogP: | 2.47630 |
- 852475-26-4MC1568
- 958254-66-51H-Imidazo[4,5-b]pyridine-2-carboxaldehyde, 1-methyl-, hydrochloride
- 99170-93-1N-Methyl-2-oxazolamine
- 914458-26-7[5-(2-fluorophenyl)-1-pentyl-1H-pyrrol-3-yl]-1-naphthalenyl-Methanone
- 894852-01-87-BROMO-2,2-DIMETHYL-2H-PYRIDO[3,2-B][1,4]OXAZIN-3(4H)-ONE
- 90221-55-92-bromo-5-methylbenzaldehyde
- 885590-99-82,3-DIFLUORO-4-IODOBENZALDEHYDE
- 97730-31-9(S)-4'-(2-Methylbutyl)Biphenyl-4-Carbonitrile
- 926293-55-26-Bromo-2-methylpyridine-3-carboxaldehyde
- 911112-05-55-Iodo-3-(trifluoromethyl)-2-pyridinamine
- 52562-19-3
2-isopropenylaniline

- 83-34-1
3-Methylindole

| Conditions | Yield |
|---|---|
| With 1-fluoro-3,3-dimethyl-1,3-dihydro-1λ3-benzo[d][1,2]iodaoxole In acetonitrile at 20℃; Molecular sieve; | 100% |
| With dipotassium peroxodisulfate; iron(II) fluoride; triethylamine In tetrahydrofuran at 70℃; for 16h; | 54% |
| With copper diacetate In dimethyl sulfoxide at 120℃; for 12h; Wacker Oxidation; | 41% |
| Multi-step reaction with 2 steps 1: 2-Methyl-2-nitropropane; trimethylsilylazide / acetonitrile / 1 h / 0 - 20 °C 2: C58H65N5Ni2O(1+)*F6P(1-) / (2)H8-toluene / 2 h / 110 °C / Inert atmosphere; Glovebox View Scheme |

| Conditions | Yield |
|---|---|
| With palladium 10% on activated carbon; water; hydrazine hydrate In ethanol at 100℃; for 24h; Reagent/catalyst; | 99% |
| copper chromite In diethylene glycol dimethyl ether | 97% |
| In diethylene glycol dimethyl ether | 97.5% |
- 36797-43-0
3-methyl-N-(p-toluenesulfonyl)indole

- 83-34-1
3-Methylindole

| Conditions | Yield |
|---|---|
| With sodium hydroxide In ethanol for 24h; Heating; | 99% |
| With formic acid; (4,4'-di-tert-butyl-2,2'-dipyridyl)-bis-(2-phenylpyridine(-1H))-iridium(III) hexafluorophosphate; N-ethyl-N,N-diisopropylamine In acetonitrile at 20℃; for 24h; Inert atmosphere; Sealed tube; Irradiation; | 83% |
| With sodium hydride In N,N-dimethyl acetamide at 60℃; for 2h; Inert atmosphere; | 83% |

- 83-34-1
3-Methylindole

| Conditions | Yield |
|---|---|
| With lithium diisopropyl amide In tetrahydrofuran; hexane at 40 - 45℃; for 2h; | 99% |
| With n-butyllithium; diisopropylamine In tetrahydrofuran; hexanes at -78 - 45℃; for 2h; Inert atmosphere; | 99% |

| Conditions | Yield |
|---|---|
| With zinc(II) chloride In 1,4-dioxane at 100℃; for 12h; Schlenk technique; | 99% |

| Conditions | Yield |
|---|---|
| With C21H21ClIrNO2 In tetrahydrofuran; 2,2,2-trifluoroethanol for 20h; Inert atmosphere; | 98% |
| With tris(bipyridine)ruthenium(II) dichloride hexahydrate; chloropyridinecobaloxime(III) In ethanol at 30℃; for 24h; Inert atmosphere; Schlenk technique; Irradiation; | 98% |
| With aluminum oxide In N,N-dimethyl-formamide at 120℃; for 7h; Inert atmosphere; | 92% |
- 73396-87-9
2-iodo-N-allylaniline

- 83-34-1
3-Methylindole

| Conditions | Yield |
|---|---|
| With tetrabutyl-ammonium chloride; sodium carbonate; palladium diacetate In N,N-dimethyl-formamide at 25℃; for 24h; | 97% |
| With palladium diacetate; tris-(2-(8-sodium sulfonatodibenzofuranyl))phosphine; triethylamine In water; acetonitrile at 40℃; for 19h; | 97% |
| palladium diacetate In acetonitrile at 110℃; | 87% |
- 23543-66-0
N-acetyl-3-methylindole

- 83-34-1
3-Methylindole

| Conditions | Yield |
|---|---|
| With 1,8-diazabicyclo[5.4.0]undec-7-ene In methanol for 0.5h; Product distribution; Further Variations:; Solvents; reaction time; Heating; | 97% |

| Conditions | Yield |
|---|---|
| With zinc(II) chloride at 200℃; Concentration; Temperature; | 96% |
| With zinc(II) chloride at 180℃; |

| Conditions | Yield |
|---|---|
| With 4-methylbenzenesulfonic acid-based ionic liquid supported on silica gel In ethanol at 20℃; for 4h; Fischer Indole Synthesis; | 96% |
| With ammonium cerium (IV) nitrate In methanol for 4h; Reflux; |
Consensus Reports
Specification
The 3-Methylindole, with the CAS registry number 83-34-1, has the IUPAC name of 3-methyl-1H-indole. For being a kind of white or slightly brown platelets with irritating odour, it is sensitive to light, and is stable chemically while incompatible with strong oxidizing agents, strong acids, acid ahydrides, acid chlorides. combustible. Besides, its product categories are including IndoleDerivative; Pyrroles & Indoles; Indoles; Simple Indoles; Pyrroles & Indoles.
The characteristics of this chemical are as follows: (1)ACD/LogP: 2.60; (2)# of Rule of 5 Violations: 0; (3)ACD/LogD (pH 5.5): 2.6; (4)ACD/LogD (pH 7.4): 2.6; (5)ACD/BCF (pH 5.5): 55.96; (6)ACD/BCF (pH 7.4): 55.96; (7)ACD/KOC (pH 5.5): 620.53; (8)ACD/KOC (pH 7.4): 620.54; (9)#H bond acceptors: 1; (10)#H bond donors: 1; (11)#Freely Rotating Bonds: 0; (12)Polar Surface Area: 4.93; (13)Index of Refraction: 1.654; (14)Molar Refractivity: 43.35 cm3; (15)Molar Volume: 118.1 cm3; (16)Polarizability: 17.18 ×10-24 cm3; (17)Surface Tension: 46.4 dyne/cm; (18)Density: 1.11 g/cm3; (19)Flash Point: 112.5 °C; (20)Enthalpy of Vaporization: 48.28 kJ/mol; (21)Boiling Point: 265.1 °C at 760 mmHg; (22)Vapour Pressure: 0.0153 mmHg at 25°C; (23)Exact Mass: 131.073499; (24)MonoIsotopic Mass: 131.073499; (25)Topological Polar Surface Area: 15.8; (26)Heavy Atom Count: 10; (27)Complexity: 122.
The production method is as below: This is the industrial way, firstly heat the propionaldehyde and the phenylhydrazine to remove water molecules to obtain the propylal phenylhydrazone; Next heat it with zinc chloride or sulfuric acid to remove the ammonia molecular and finally you could get 3-Methylindole.
As to its usage, it is widely applied in many ways. For being a kind of good fixative, this chmeical is usually used in the floral essences and could also be used to obtain good natural animal perfume with other chemical; And it could be used in the food essence like grape, sweet fruit and nut; Beside all these, it could be used as the organic synthesis reagent and the biochemical reagents.
When you are dealing with this chemical, you should be very careful. For being a kind of irritant chemical, it is irritating to eyes, respiratory system and skin and may cause inflammation to the skin or other mucous membranes. Therefore, you should wear suitable protective clothing, and avoid contacting with skin and eyes. If in case of contact with eyes, rinse immediately with plenty of water and seek medical advice.
Additionally, you could convert the following datas into the molecular structure:
(1)Canonical SMILES: CC1=CNC2=CC=CC=C12
(2)InChI: InChI=1S/C9H9N/c1-7-6-10-9-5-3-2-4-8(7)9/h2-6,10H,1H3
(3)InChIKey: ZFRKQXVRDFCRJG-UHFFFAOYSA-N
Below are the toxicity information of this chemical:
| Organism | Test Type | Route | Reported Dose (Normalized Dose) | Effect | Source |
|---|---|---|---|---|---|
| cattle | LDLo | intravenous | 60mg/kg (60mg/kg) | LUNGS, THORAX, OR RESPIRATION: EMPHYSEMA LUNGS, THORAX, OR RESPIRATION: CHRONIC PULMONARY EDEMA | Food and Cosmetics Toxicology. Vol. 14, Pg. 863, 1976. |
| cattle | LDLo | oral | 200mg/kg (200mg/kg) | LUNGS, THORAX, OR RESPIRATION: EMPHYSEMA LUNGS, THORAX, OR RESPIRATION: CHRONIC PULMONARY EDEMA | Food and Cosmetics Toxicology. Vol. 14, Pg. 863, 1976. |
| domestic animals - goat/sheep | LDLo | oral | 300mg/kg (300mg/kg) | LUNGS, THORAX, OR RESPIRATION: CHRONIC PULMONARY EDEMA | Food and Cosmetics Toxicology. Vol. 14, Pg. 863, 1976. |
| frog | LDLo | parenteral | 435mg/kg (435mg/kg) | BEHAVIORAL: ALTERED SLEEP TIME (INCLUDING CHANGE IN RIGHTING REFLEX) LUNGS, THORAX, OR RESPIRATION: RESPIRATORY DEPRESSION | Journal of Pharmacology and Experimental Therapeutics. Vol. 19, Pg. 307, 1922. |
| frog | LDLo | subcutaneous | 1gm/kg (1000mg/kg) | "Merck Index; an Encyclopedia of Chemicals, Drugs, and Biologicals", 11th ed., Rahway, NJ 07065, Merck & Co., Inc. 1989Vol. 11, Pg. 1354, 1989. | |
| mouse | LD50 | intraperitoneal | 175mg/kg (175mg/kg) | LIVER: OTHER CHANGES KIDNEY, URETER, AND BLADDER: OTHER CHANGES BLOOD: OTHER CHANGES | Food and Cosmetics Toxicology. Vol. 14, Pg. 863, 1976. |
| mouse | LDLo | oral | 470mg/kg (470mg/kg) | Archives of Environmental Contamination and Toxicology. Vol. 14, Pg. 111, 1985. | |
| rat | LD50 | oral | 3450mg/kg (3450mg/kg) | Food and Cosmetics Toxicology. Vol. 14, Pg. 863, 1976. |