12069-32-8 Usage
Description
Boron carbide, also known as B4C, is a black, hard, crystalline compound with a density of 2.50 g/cm3, a hardness of 9.3 on the Mohs scale, and a melting point of 2,350°C. It is insoluble in water and acid, inert to most chemicals at ordinary temperatures, and rapidly attacked by hot alkalies. Boron carbide is manufactured by the reduction of boric oxide with carbon at high temperatures.
Uses
1. Nuclear Reaction Industry: Boron carbide is used as a thermal-neutron absorber for controlling nuclear fission. It absorbs a large number of neutrons without forming any radioactive isotopes.
2. Wear Applications: In the manufacturing of hard and chemical-resistant ceramics or wear-resistant tools, boron carbide is used as an abrasive material. The molded article can be used for wear-resistant material in grinding, milling, drilling, and polishing of gemstones, cemented carbide, and other hard materials.
3. Nozzles: Boron carbide is used in high-pressure water jet cutter nozzles and grit blasting nozzles due to its high hardness and good pressure, temperature, and wear resistance.
4. Medicine: It is used in boron neutron capture therapy for brain cancer as a neutron absorber, which absorbs neutrons without forming radioactive isotopes.
5. Coatings: Boron carbide is used as a coating material for ceramic coating of warships and helicopters, providing light weight and good resistance to piercing shells. It is also used in scratch and wear-resistant coatings due to its high hardness and wear resistance.
6. Chemical Manufacture: It serves as a source of boron in the manufacturing of metal boron compounds.
7. Others: Boron carbide is used in cutting tools and dies, brake linings of vehicles, and as an additive to increase the hardness and wear resistance of linings. It is also used in the inner plates of ballistic vests for its light weight and good resistance to bullets.
Physical and chemical properties
Boron carbide is black crystal with metallic luster, hardness ranks only second to diamond, higher than silicon carbide, Mohs hardness is 9.3. Chemical property is stable, it does not react with the acid solution, the formula is B4C, relative density is 2.52, melting point is 2350℃, boiling point is higher than 3500℃. The melt boron carbide can dissolve in a large amount of boron carbide graphitic carbon. Boron carbide is stable in dilute acid solution, the mixed acid of sulfuric acid and hydrofluoric acid, the mixed acid of sulfuric acid and nitric acid can decompose boron carbide. When heated to 1000℃, it is slowly oxidized to carbon dioxide and boron oxide in oxygen. Boron carbide has high thermal neutron capture capability, it is wear-resisting, it has semiconducting properties. In most cases, because boron carbide (of B4C) is used as control materials, it can meet the requirements of high-temperature reactors. Increasing the concentration of B10 in the boron carbide can improve the control efficiency of boron material. Boron carbide has a density of 2.51 × 103kg/m3, melting point is 2450 ℃, the thermal expansion coefficient (20~800 ℃) is 4.5 × 10-6/℃.
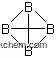
Figure 1 is the molecular structure of boron carbide.
Industrial boron carbide is mainly produced by methods of melting boron oxide in an electric arc furnace, and reacts with carbon can generat boron carbide. It can also obtained by carbon magnesium reduction method or hydrocarbon reduction method. Boron carbide is mainly used as abrasive, for grinding and polishing of industrial use.
Boron carbide is usually prepared by boric oxide and carbon in an electric furnace by high temperature heating. Reaction is as follows: 2B2O3 + 7C → B4C + 6CO.
Product quality general requires for grains with B4C is not less than 94%, milling class is not less than 90%.
Boron carbide is mainly used for grinding, milling, drilling and polishing of cemented carbide, precious stones and other hard materials. Moldings can be used for wear-resistant material, refractory material, used in the manufacture of hard resistant and corrosion-resistant ceramic water-resistant bearings, it is used as neutron control rods of nuclear reactor, it is also used for smelting boron steel, boron alloys and special welding. Alloy of boron carbide and aluminum (containing up to 50% B4C) is used for neutron shielding, reacting furnace screen cover and so on.
Boron carbide is packed in plastic bags, stored in a dry, clean warehouse.
More information is edited by lookchem Xiaonan (2016-12-03).
Boron carbide ceramics
Boron carbide ceramics is a class of ceramics which the main chemical ingredient is boron carbide. The chemical formula of boron carbide is B4C, it belongs to hexagonal diamond crystal, there are 12 boron atoms in the unit cell, lattice parameter co = 1.212nm, ao = 0. 56nm. Crystal structure exists atom which can accommodate up to 0.18nm diameter, so it can remain the lithium or helium atoms within the crystal structure. Synthesis of boron carbide powder is mainly used carbon thermal reduction method, except the direct reduction with boron anhydride, it can also obtained in the presence of carbon (C), boron anhydride is reduced by Mg, the reaction is: 2B2O3 + 6Mg + C → B4C + 6MgO, the reaction temperature is 1000~1200 ℃. This reaction is highly exothermic, the final product requires H2SO4 or HCl acid pickling, then be washed with hot water to obtain more pure and fine grain size (0.1~5μm), no C boron carbide powder. Boron carbide ceramic is main hot pressing, hot isostatic pressing and non-pressure sintering is also used. Hot press sintering temperature is 2000~2100 ℃, generally Mg, Al, Cr, Si, Ti or a metal such as Al2O3, MgO, etc. or an oxide glass is added to use as sintering aid. The melting point of boron carbide is 2450 ℃, the theoretical density is 2.519g/cm3, elastic modulus is 360~460GPa, the thermal expansion coefficient is 5.73 × 10-6/K (300~1970K), the thermal conductivity at 200 ℃ is 24W/( m ? K), hardness is 4950kg/mm2, just below the diamond and cubic boron nitride, it has a large thermal neutron capture cross-section. It is used to process precious stones, ceramics, molds, tools and bearings. It is also used as nozzles, bulletproof materials and nuclear reactor neutron absorber.
References
1.https://www.azonano.com/article.aspx?ArticleID=3328
2.http://www.dynacer.com/materials/boron-carbide/
3.http://www.sciencedirect.com/science/article/pii/S0969804305003118
4.https://en.wikipedia.org/wiki/Boron_carbide#Uses
5.https://www.lookchem.com/ProductChemicalPropertiesCB6315643.htm
6.https://baike.baidu.com/item/%E7%A2%B3%E5%8C%96%E7%A1%BC/2363107?fr=aladdin
Preparation
Boron carbide is prepared by reduction of boric oxide either with carbon or with magnesium in presence of carbon in an electric furnace at a temperature above 1,400°C. When magnesium is used, the reaction may be carried out in a graphite furnace and the magnesium byproducts are removed by treatment with acid.
Check Digit Verification of cas no
The CAS Registry Mumber 12069-32-8 includes 8 digits separated into 3 groups by hyphens. The first part of the number,starting from the left, has 5 digits, 1,2,0,6 and 9 respectively; the second part has 2 digits, 3 and 2 respectively.
Calculate Digit Verification of CAS Registry Number 12069-32:
(7*1)+(6*2)+(5*0)+(4*6)+(3*9)+(2*3)+(1*2)=78
78 % 10 = 8
So 12069-32-8 is a valid CAS Registry Number.
InChI:InChI=1/CB4/c2-1-3(2)5(1)4(1)2