25086-89-9 Usage
Description
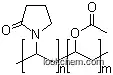
Poly(1-vinylpyrrolidone-co-vinyl acetate), also known as Copovidone, is a water-soluble polymer with a 1:2.4 mole ratio of vinylpyrrolidone (VP) to vinyl acetate (VA). It is a white to yellowish-white amorphous powder, typically spray-dried with a relatively fine particle size. Copovidone has a slight odor and a faint taste.
Uses
Used in Pharmaceutical Applications:
Poly(1-vinylpyrrolidone-co-vinyl acetate) is used as a pharmaceutical excipient for improving the uptake and drug loading of various pharmaceutical agents, including contraceptive patches. It enhances the bioavailability and efficacy of the drugs by increasing their solubility and stability in the formulation.
Used in Drug Delivery Systems:
In the drug delivery industry, Poly(1-vinylpyrrolidone-co-vinyl acetate) is utilized as a carrier material for the development of novel drug delivery systems. These systems aim to improve the targeted delivery, bioavailability, and therapeutic outcomes of various pharmaceutical agents by providing a controlled and sustained release of the drug.
Used in Cosmetic Applications:
Poly(1-vinylpyrrolidone-co-vinyl acetate) is also used in the cosmetic industry as a film-forming agent, providing a smooth and uniform texture to the products. It helps improve the skin's appearance and feel by forming a protective layer on the skin's surface, which can also enhance the stability and shelf life of cosmetic formulations.
Used in Adhesive Applications:
In the adhesive industry, Poly(1-vinylpyrrolidone-co-vinyl acetate) is employed as a component in the formulation of pressure-sensitive adhesives. It contributes to the adhesive's tackiness, shear strength, and peel resistance, making it suitable for various applications such as packaging, labeling, and medical tapes.
Used in Textile Applications:
Poly(1-vinylpyrrolidone-co-vinyl acetate) is utilized in the textile industry as a binder and sizing agent for fibers. It helps improve the adhesion and bonding of fibers, enhancing the strength and durability of the textile products. Additionally, it can be used to provide a protective coating on fibers, offering resistance to abrasion, moisture, and other environmental factors.
Production Methods
Copovidone is manufactured by free-radical polymerization of
vinylpyrrolidone and vinyl acetate in a ratio of 6 : 4. The synthesis is
conducted in an organic solvent owing to the insolubility of vinyl
acetate in water.
Pharmaceutical Applications
Copovidone is used as a tablet binder, a film-former, and as part of
the matrix material used in controlled-release formulations. In
tableting, copovidone can be used as a binder for direct compression and as a binder in wet granulation. Copovidone is often
added to coating solutions as a film-forming agent. It provides good
adhesion, elasticity, and hardness, and can be used as a moisture
barrier.
Safety Profile
Moderately toxic by ingestion.Combustible, especially in powdered form. Incompatiblewith strong oxidising agents, strong reducing agents. Whenheated to decomposition it emits toxic vapors of NOx.
Safety
Copovidone is used widely in pharmaceutical formulations and is
generally regarded as nontoxic. However, it is moderately toxic by
ingestion, producing gastric disturbances. It has no irritating or
sensitizing effects on the skin. A study was conducted to look at the
carcinogenicity and chronic toxicity of copovidone (Kollidon VA
64) in Wistar rats and Beagle dogs. The results of these studies
demonstrated the absence of any significant toxicological findings
of high dietary levels of copodivone in rats and dogs, resulting in noobserved-
adverse-effect levels of 2800 mg/kg body-weight/day in
rats and 2500 mg/kg body-weight/day in dogs, the highest doses
tested.
LD50 (rat, oral): >0.63 g/kg
storage
Copovidone is stable and should be stored in a well-closed container
in a cool, dry place.
Incompatibilities
Copovidone is compatible with most organic and inorganic
pharmaceutical ingredients. When exposed to high water levels,
copovidone may form molecular adducts with some materials; see
Crospovidone and Povidone.
Regulatory Status
Copovidone is included in the FDA Inactive Ingredients Database
(oral tablets, oral film-coated tablets, sustained action).
Check Digit Verification of cas no
The CAS Registry Mumber 25086-89-9 includes 8 digits separated into 3 groups by hyphens. The first part of the number,starting from the left, has 5 digits, 2,5,0,8 and 6 respectively; the second part has 2 digits, 8 and 9 respectively.
Calculate Digit Verification of CAS Registry Number 25086-89:
(7*2)+(6*5)+(5*0)+(4*8)+(3*6)+(2*8)+(1*9)=119
119 % 10 = 9
So 25086-89-9 is a valid CAS Registry Number.
InChI:InChI=1/C6H9NO.C4H6O2/c1-2-7-5-3-4-6(7)8;1-3-6-4(2)5/h2H,1,3-5H2;3H,1H2,2H3