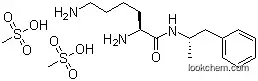
608137-33-3 Usage
Uses
Used in Pharmaceutical Industry:
(2S)-2,6-DIAMINO-N-[(1S)-1-METHYL-2-PHENYLETHYL]HEXANAMIDE DIMETHANESULFONATE is used as an active pharmaceutical ingredient for the treatment of attention deficit hyperactivity disorder (ADHD). It is believed to inhibit the reuptake of neurotransmitters dopamine and noradrenaline (norepinephrine), thereby increasing their presynaptic availability and release into the extraneuronal space.
Used in Research and Forensic Applications:
(2S)-2,6-DIAMINO-N-[(1S)-1-METHYL-2-PHENYLETHYL]HEXANAMIDE DIMETHANESULFONATE is used as an analytical reference material in the pharmaceutical industry. It is regulated as a Schedule II compound in the United States and is intended for research and forensic applications.
Brand name: Vyvanse
Originator
New River Pharmaceuticals (US)
Synthesis
The straightforward synthesis of lisdexamfetamine mesilate
was initiated by adding a solution of D-amphetamine (66) to
a solution of Boc-L-Lys(Boc)-OSu (65), N-methylmorpholine
and 1,4-dioxane. The resulting
mixture was partitioned between isopropyl acetate and an
acetic acid/brine solution, and the organic layer was washed
with aqueous sodium bicarbonate to give Boc-L-Lys(Boc)-
D-amphetamine (67) in 91% yield. The two primary amine
groups were liberated by reacting a solution of 67 in 1,4-
dioxane with methanesulfonic acid providing lisdexamfetamine
mesilate (X) in 92% yield.
Check Digit Verification of cas no
The CAS Registry Mumber 608137-33-3 includes 9 digits separated into 3 groups by hyphens. The first part of the number,starting from the left, has 6 digits, 6,0,8,1,3 and 7 respectively; the second part has 2 digits, 3 and 3 respectively.
Calculate Digit Verification of CAS Registry Number 608137-33:
(8*6)+(7*0)+(6*8)+(5*1)+(4*3)+(3*7)+(2*3)+(1*3)=143
143 % 10 = 3
So 608137-33-3 is a valid CAS Registry Number.