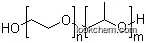9003-11-6 Usage
Chemical Properties
Polyethylene-polypropylene glycol generally occur as white, waxy, free-flowing prilled granules, or as cast solids. They are practically odorless and tasteless. At room temperature, poloxamer 124 occurs as a colorless liquid.
Originator
Polykol ,Upjohn, US ,1958
Uses
Different sources of media describe the Uses of 9003-11-6 differently. You can refer to the following data:
1. Polyethylene-polypropylene glycol is a liquid surfactant polymer.
2. pyrethroid-based pesticide for use in mosquito control
Production Methods
Poloxamer polymers are prepared by reacting propylene oxide with
propylene glycol to form polyoxypropylene glycol. Ethylene oxide
is then added to form the block copolymer.
Manufacturing Process
(A) In a 1-liter 3-necked round bottom flask equipped with a mechanical stirrer, reflux condenser, thermometer and propylene oxide feed inlet, there were placed 57 g (0.75 mol) of propylene glycol and 7.5 g of anhydrous sodium hydroxide. The flask was purged with nitrogen to remove air and heated to 120°C with stirring and until the sodium hydroxide was dissolved. Then sufficient propylene oxide was introduced into the mixture as fast as it would react until the product possessed a calculated molecular weight of 2,380. The product was cooled under nitrogen, the NaOH catalyst neutralized with sulfuric acid and the product filtered. The final product was a waterinsoluble polyoxypropylene glycol having an average molecular weight of 1,620 as determined by hydroxyl number or acetylation analytical test procedures.(B) The foregoing polyoxypropylene glycol having an average 1,620 molecular weight was placed in the same apparatus as described in procedure (A), in the amount of 500 g (0.308 mol), to which there was added 5 g of anhydrous sodium hydroxide. 105 g of ethylene oxide was added at an average temperature of 120°C, using the same technique as employed in (A). The amount of added ethylene oxide corresponded to 17.4% of the total weight of the polyoxypropylene glycol base plus the weight of added ethylene oxide.
Brand name
Lutrol F (BASF); Pluracare (BASF);
Pluronic (BASF).
Therapeutic Function
Pharmaceutic aid (surfactant)
General Description
Pluronic? L-81 is a lipoprotein secretion inhibitor.
Pharmaceutical Applications
Polyethylene-polypropylene glycol is nonionic polyoxyethylene–polyoxypropylene copolymers used primarily in pharmaceutical formulations as emulsifying or solubilizing agents.The polyoxyethylene segment is hydrophilic while the polyoxypropylene segment is hydrophobic. All of the Polyethylene-polypropylene glycol is chemically similar in composition, differing only in the relative amounts of propylene and ethylene oxides added during manufacture. Their physical and surface-active properties vary over a wide range and a number of different types are commercially available;
Polyethylene-polypropylene glycol is used as emulsifying agents in intravenous fat emulsions, and as solubilizing and stabilizing agents to maintain the clarity of elixirs and syrups. Polyethylene-polypropylene glycol may also be used as wetting agents; in ointments, suppository bases, and gels; and as tablet binders and coatings.
Poloxamer 188 has also been used as an emulsifying agent for fluorocarbons used as artificial blood substitutes, and in the preparation of solid-dispersion systems. More recently,Polyethylene-polypropylene glycol has found use in drug-delivery systems.
Therapeutically, poloxamer 188 is administered orally as a wetting agent and stool lubricant in the treatment of constipation; it is usually used in combination with a laxative such as danthron. Polyethylene-polypropylene glycol may also be used therapeutically as wetting agents in eye-drop formulations, in the treatment of kidney stones, and as skin-wound cleansers.
Poloxamer 338 and 407 are used in solutions for contact lens care.
Safety Profile
When heated to
decomposition it emits acrid smoke and
irritating fumes.
Safety
Polyethylene-polypropylene glycol is used in a variety of oral, parenteral, and topical pharmaceutical formulations, and are generally regarded as nontoxic and nonirritant materials.Polyethylene-polypropylene glycol is not metabolized in the body.
Animal toxicity studies, with dogs and rabbits, have shown Polyethylene-polypropylene glycol to be nonirritating and nonsensitizing when applied in 5% w/v and 10% w/v concentration to the eyes, gums, and skin.
In a 14-day study of intravenous administration at concentrations up to 0.5 g/kg/day to rabbits, no overt adverse effects were noted. A similar study with dogs also showed no adverse effects at dosage levels up to 0.5 g/kg/day. In a longer-term study, rats fed 3% w/w or 5% w/w of poloxamer in food for up to 2 years did not exhibit any significant symptoms of toxicity. However, rats receiving 7.5% w/w of poloxamer in their diet showed some decrease in growth rate.
No hemolysis of human blood cells was observed over 18 hours at 25°C, with 0.001–10% w/v poloxamer solutions.
(mouse, IV): 1 g/kg
(mouse, oral): 15 g/kg
(mouse, SC): 5.5 g/kg
(rat, IV): 7.5 g/kg
(rat, oral): 9.4 g/kg
storage
Polyethylene-polypropylene glycol is stable materials. Aqueous solutions are stable in the presence of acids, alkalis, and metal ions. However, aqueous solutions support mold growth.
The bulk material should be stored in a well-closed container in a cool, dry place.
Incompatibilities
Depending on the relative concentrations, poloxamer 188 is
incompatible with phenols and parabens.
Regulatory Status
Included in the FDA Inactive Ingredients Database (IV injections;
inhalations, ophthalmic preparations; oral powders, solutions,
suspensions, and syrups; topical preparations). Included in nonparenteral
medicines licensed in the UK. Included in the Canadian
List of Acceptable Non-medicinal Ingredients.
Check Digit Verification of cas no
The CAS Registry Mumber 9003-11-6 includes 7 digits separated into 3 groups by hyphens. The first part of the number,starting from the left, has 4 digits, 9,0,0 and 3 respectively; the second part has 2 digits, 1 and 1 respectively.
Calculate Digit Verification of CAS Registry Number 9003-11:
(6*9)+(5*0)+(4*0)+(3*3)+(2*1)+(1*1)=66
66 % 10 = 6
So 9003-11-6 is a valid CAS Registry Number.
InChI:InChI=1/C3H6O.C2H4O/c1-3-2-4-3;1-2-3-1/h3H,2H2,1H3;1-2H2