9004-53-9 Usage
Identification test
Take about 1 g sample and suspend it in 20ml of water. Add a few drops of iodine test solution (TS-124), which should then appear dark blue color to reddish brown color. The infrared absorption spectrum should be consistent with Figure 10107.
Toxicity
ADI does not make special provisions (FAO/WHO, 2001).
GRAS (FDA, § 184.1277, 2000);
Usage limit
FAO/WHO (1984, g/kg): for broth, soup, the value should be according to GMI; cheese (in the cream mixture): 5; the sweetened yogurt and its products after being subject to the heat treatment: 10; mackerel and fish canned: 60 (Only in the packaging material); cold drink: 30.
Chemical properties
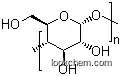
It appears as white, yellow or brown flowing powder. Dextrin of relatively small molecular weight is completely soluble in water to form viscous slurry; dextrin with relatively large molecular mass is not completely soluble in water. ADI is not subject to special provisions (FAO/WHO, 1994).
Application
It can be used as emulsifying stabilizers and thickeners, adhesives and surface decorators.
It can be applied to the pharmaceutical industry, protective adhesives and suspending agents, adhesives. For example, it can be applied to the sizing treatment of paper, textile sizing and ink preparation; it can also be used as a drug excipient and a substitute of the Arabic gum.
Production
It is obtained from using natural starch originated from cereals or rhizome plants (E.g., corn, sorghum, potato, bamboo, wheat, rice, cassava, sago, etc.) as raw materials which are heated in the presence of suitable food-grade acids and buffers, leading to partial hydrolysis to obtain it. Drying method: apply hydrochloric acid (≤ 0.15%) or orthophosphoric acid (≤ 0.17%) treatment.
Enzymatic method: take the natural starch of corn, wheat and dried potato as raw material, use 0.15% hydrochloric acid or 0.17% phosphoric acid for heat treatment, and apply starch partial degradation to derive the products.
Chemical Properties
Different sources of media describe the Chemical Properties of 9004-53-9 differently. You can refer to the following data:
1. White powder
2. Dextrin is partially hydrolyzed maize (corn), potato or cassava starch. It is a white, pale yellow or brown-colored powder with a slight characteristic odor.
Uses
Different sources of media describe the Uses of 9004-53-9 differently. You can refer to the following data:
1. Dextrin is a partially hydrolyzed starch formed from the treatment of
starch by dry heat, acid, or enzymes. it can be formed from amylose
and amylopectin-type starches. they are white or yellow (canary) in
color. as compared to unmodified starch, s have increased
water solubility, viscosity stability, and reduced paste viscosity. uses
include dough improvement and binding.
2. Pharmaceutic aid (suspending agent); pharmaceutic aid
(viscosity-increasing agent); pharmaceutic aid (tablet
binder); pharmaceutic aid (tablet and capsule diluent).
3. dextrin (British gum; starch gum) absorbs moisture. It is also used as a binder to control product viscosity and reduce the density of a cosmetic. This powder is produced from corn starch and modified by means of a bacterial process. It may cause an allergic reaction.
Production Methods
Dextrin is prepared by the incomplete hydrolysis of starch by heating in the dry state with or without the aid ofsuitable acids and buffers; moisture may be added during heating. The PhEur 6.4 specifies that dextrin is derived from maize(corn),potatoorcassava starch. A specification for cassava is included in the USP32–NF27.
Definition
Different sources of media describe the Definition of 9004-53-9 differently. You can refer to the following data:
1. A polysaccharide sugar
produced by the action of amylase enzymes
on or the chemical hydrolysis of starch.
Dextrins are used as adhesives.
2. dextrin: An intermediate polysaccharidecompound resulting fromthe hydrolysis of starch to maltose byamylase enzymes.
General Description
Dextrin is basically a sweet sugar extracted from potato starch. Dextrin color range is from white to tan; it has low to high solubilities in cold water, and gives pastes that vary widely in viscosity. It is produced from all commercial grain and tuber starches. During its manufacture, the factors taken into consideration are type of starch and moisture content, roasting time and temperature, and the type and amount of catalyst used.
Agricultural Uses
Dextrin is a group of colloidal products, formed by the hydrolysis of starches with dilute acids or by heating dry starch. The yellow or white powder or granules obtained are soluble in boiling water and insoluble in ether or alcohol. Dextrin is used in adhesives, as thickening agents and in penicillin manufacture.
Pharmaceutical Applications
Dextrin is a dextrose polymer used as an adhesive and stiffening agent for surgical dressings. It is also used as a tablet and capsule diluent; as a binder for tablet granulation; as a sugar-coating ingredient that serves as a plasticizer and adhesive; and as a thickening agent for suspensions.Additionally, dextrin has been used as a source of carbohydrate by people with special dietary requirements because it has a low electrolyte content and is free of lactose and sucrose.Dextrin is also used in cosmetics.
Safety Profile
Mdly toxic by
intravenous route. When heated to
decomposition it emits acrid smoke and
irritating fumes.
Safety
Dextrin is generally regarded as a nontoxic and nonirritant material at the levels employed as an excipient. Larger quantities are used as a dietary supplement without adverse effects, although ingestion of very large quantities may be harmful.
LD50 (mouse, IV): 0.35g/kg
storage
Physical characteristics of dextrin may vary slightly depending on the method of manufacture and on the source material. In aqueous solutions, dextrin molecules tend to aggregate as density, temperature, pH, or other characteristics change. An increase in viscosity is caused by gel at ion or retro gradation as dextrin solutions age,and is particularly noticeable in the less-soluble maize starch dextrins. Dextrin solutions are thixotropic, becoming less viscous when sheared but changing to a soft paste or gel when allowed to stand. However, acids that are present in dextrin as residues from manufacturing can cause further hydrolysis, which results in a gradual thinning of solutions. Residual acid, often found in lesssoluble dextrins such as pyrodextrin, will also cause a reduction in viscosity during dry storage. To eliminate these problems, dextrin manufacturers neutralize dextrins of low solubility with ammonia or sodium carbonate in the cooling vessel.
The bulk material should be stored in a well-closed container in a cool, dry place.
Regulatory Status
GRAS listed.Included in the FDA Inactive Ingredients Database(IV injections, oral tablets and topical preparations). Included in nonparenteral medicines licensed in the UK. Included in the Canadian List of Acceptable Non-medicinal Ingredients.
Check Digit Verification of cas no
The CAS Registry Mumber 9004-53-9 includes 7 digits separated into 3 groups by hyphens. The first part of the number,starting from the left, has 4 digits, 9,0,0 and 4 respectively; the second part has 2 digits, 5 and 3 respectively.
Calculate Digit Verification of CAS Registry Number 9004-53:
(6*9)+(5*0)+(4*0)+(3*4)+(2*5)+(1*3)=79
79 % 10 = 9
So 9004-53-9 is a valid CAS Registry Number.
InChI:InChI=1/C18H32O16/c19-1-4-7(22)8(23)12(27)17(31-4)34-15-6(3-21)32-18(13(28)10(15)25)33-14-5(2-20)30-16(29)11(26)9(14)24/h4-29H,1-3H2/t4-,5-,6-,7-,8+,9-,10-,11-,12-,13-,14-,15-,16+,17?,18?/m1/s1