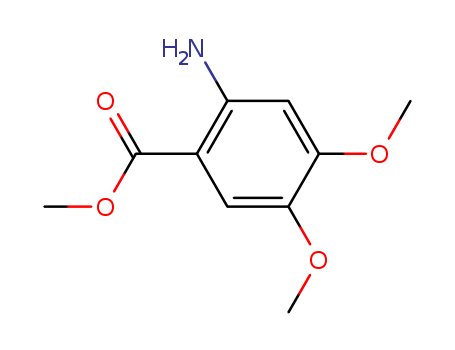
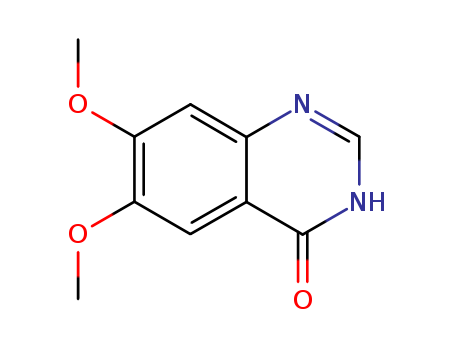
Relevant articles and documents
Novel 3,4-dihydro-4-oxoquinazoline-based acetohydrazides: Design, synthesis and evaluation of antitumor cytotoxicity and caspase activation activity
Huan, Le Cong Tran, Phuong-Thao Phuong, Cao Viet Duc, Phan Huy Anh, Duong Tien Hai, Pham The Huong, Le Thi Thu Thuan, Nguyen Thi Lee, Hye Jin Park, Eun Jae Kang, Jong Soon Linh, Nguyen Phuong Hieu, Tran Trung Oanh, Dao Thi Kim Han, Sang-Bae Nam, Nguyen-Hai
In search for novel small molecules with antitumor cytotoxicity via activating procaspase-3, we have designed and synthesized three series of novel (E)-N′-benzylidene-4-oxoquinazolin-3(4H)-yl)acetohydrazides (5a-j, 6a-h, and 7a-h). On the phenyl ring ò the benzylidene part, three different substituents, including 2-OH-4-OCH3, 4-OCH3, and 4-N(CH3)2, were introduced, respectively. Biological evaluation showed that the acetohydrazides in series 5a-j, in which the phenyl ring of the benzylidene part was substituted by 2-OH-4-OCH3 substituent, exhibited potent cytotoxicity against three human cancer cell lines (SW620, colon; PC-3, prostate; NCI-H23, lung). Most of the compounds, in this series, especially compounds 5c, 5b and 5h, also significantly activated caspase-3 activity. Among these, compound 5c displayed 1.61-fold more potent than PAC-1 as caspase-3 activator. Cell cycle analysis showed that compounds 5b, 5c, and 5h significantly arrested the cell cycle in G1 phase. Further apoptotic studies also demonstrated compounds 5b, 5c, and 5h as strong apoptotic cell death inducers. The docking simulation studies showed that these compounds could activate procaspase via chelating Zn2+ ion bound to the allosteric site of the zymogen.
Visible-Light-Induced Radical Difluoromethylation/Cyclization of Unactivated Alkenes: Access to CF2H-Substituted Quinazolinones
Chen, Xiaoyu Liu, Bo Pei, Congcong Li, Jingya Zou, Dapeng Wu, Yangjie Wu, Yusheng
A mild and efficient visible-light-induced radical difluoromethylation/cyclization of unactivated alkenes toward the synthesis of substituted quinazolinones with easily accessible difluoromethyltriphenylphosphonium bromide has been developed. The transformation has the advantages of wide functional group compatibility, a broad substrate scope, and operational simplicity. The benign protocol offers a facile access to pharmaceutically valuable difluoromethylated polycyclic quinazolinones.