Relevant articles and documents
Biphasic Bioelectrocatalytic Synthesis of Chiral β-Hydroxy Nitriles
Dong, Fangyuan Chen, Hui Malapit, Christian A. Prater, Matthew B. Li, Min Yuan, Mengwei Lim, Koun Minteer, Shelley D.
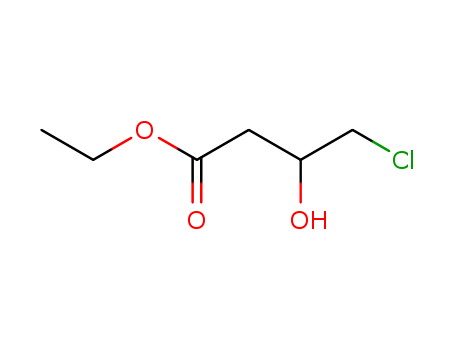
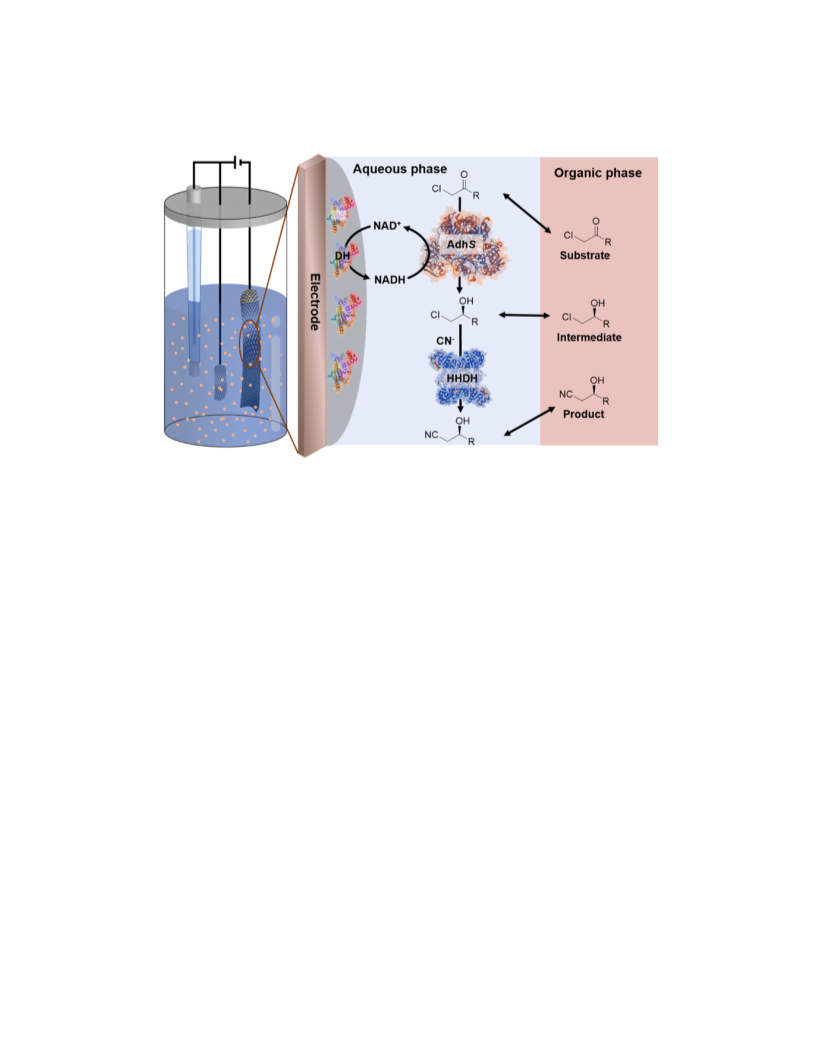
Two obstacles limit the application of oxidoreductase-based asymmetric synthesis. One is the consumption of high stoichiometric amounts of reduced cofactor. The other is the low solubility of organic substrates, intermediates, and products in the aqueous phase. In order to address these two obstacles to oxidoreductase-based asymmetric synthesis, a biphasic bioelectrocatalytic system was constructed and applied. In this study, the preparation of chiral β-hydroxy nitriles catalyzed by alcohol dehydrogenase (AdhS) and halohydrin dehalogenase (HHDH) was investigated as a model bioelectrosynthesis, since they are high-value intermediates in statin synthesis. Diaphorase (DH) was immobilized by a cobaltocene-modified poly(allylamine) redox polymer on the electrode surface (DH/Cc-PAA bioelectrode) to achieve effective bioelectrocatalytic NADH regeneration. Since AdhS is a NAD-dependent dehydrogenase, the diaphorase-modified biocathode was used to regenerate NADH to support the conversion from ethyl 4-chloroacetoacetate (COBE) to ethyl (S)-4-chloro-3-hydroxybutanoate ((S)-CHBE) catalyzed by AdhS. The addition of methyl tert-butyl ether (MTBE) as an organic phase not only increased the uploading of COBE but also prevented the spontaneous hydrolysis of COBE, extended the lifetime of DH/Cc-PAA bioelectrode, and increased the Faradaic efficiency and the concentration of generated (R)-ethyl-4-cyano-3-hydroxybutyrate ((R)-CHCN). After 10 h of reaction, the highest concentration of (R)-CHCN in the biphasic bioelectrocatalytic system was 25.5 mM with 81.2% enantiomeric excess (eep). The conversion ratio of COBE achieved 85%, which was 8.8 times higher than that achieved with the single-phase system. Besides COBE, two other substrates with aromatic ring structures were also used in this biphasic bioelectrocatalytic system to prepare the corresponding chiral β-hydroxy nitriles. The results indicate that the biphasic bioelectrocatalytic system has the potential to produce a variety of β-hydroxy nitriles with different structures.
Efficient biosynthesis of ethyl (R)-3-hydroxyglutarate through a one-pot bienzymatic cascade of halohydrin dehalogenase and nitrilase
Yao, Peiyuan Wang, Lei Yuan, Jing Cheng, Lihua Jia, Rongrong Xie, Meixian Feng, Jinhui Wang, Min Wu, Qiaqing Zhu, Dunming
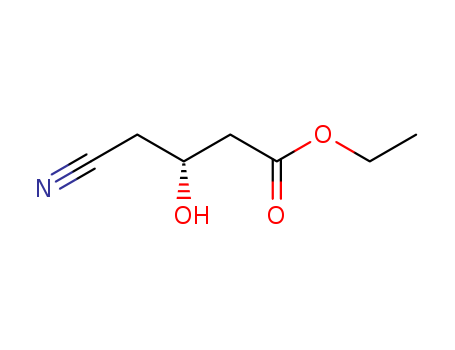
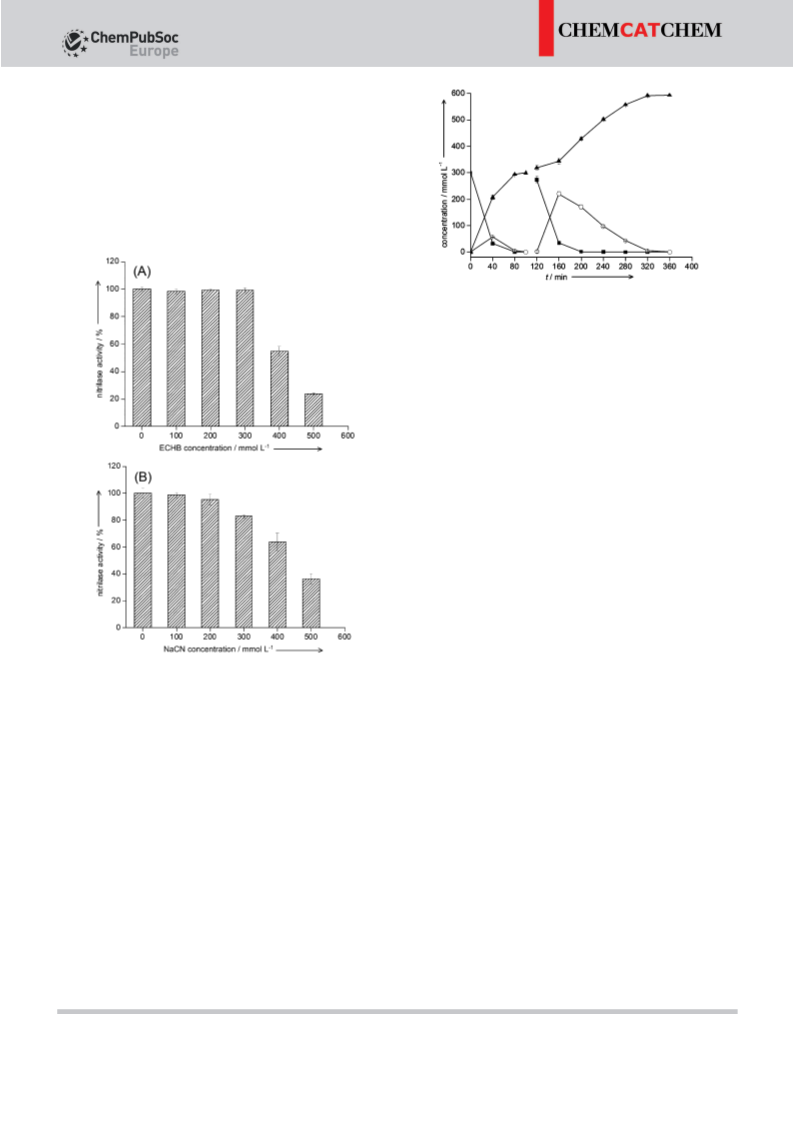
An effective one-pot bienzymatic synthesis of ethyl (R)-3-hydroxyglutarate (EHG) from ethyl (S)-4-chloro-3-hydroxybutyrate (ECHB) was achieved by using recombinant Escherichia coli cells expressing separately or co-expressing a mutant halohydrin dehalogenase gene from Agrobacterium radiobacter AD1 and a nitrilase gene from Arabidopsis thaliana. The activity of nitrilase was inhibited by high concentration of ECHB and NaCN. Consequently, the one-pot one-step process was implemented by fed-batch of ECHB and NaCN with high accumulative product concentration (up to 0.9 mol L-1). The biotransformation of ECHB to EHG was successfully achieved at 1.2 mol L-1 substrate concentration by a one-pot two-step process. As such, this one-pot bienzymatic transformation should be useful in synthesizing these important optical pure β-hydroxycarboxylic acids.