Relevant articles and documents
Synthesis and in-vitro antibacterial activity of 7-(3-aminopyrrolo[3,4-c] pyrazol-5(2H,4H,6H)-yl)-6-fluoro-4-oxo-1,4-dihydroquinoline-3-carboxylic acid derivatives
Guo, Xin Bai, Xiao-Guang Li, Yi-Liang An, Zhi-Jiao Xu, Le-Xing Han, Li-You Liu, Ming-Liang Guo, Hui-Yuan Wang, Yu-Cheng
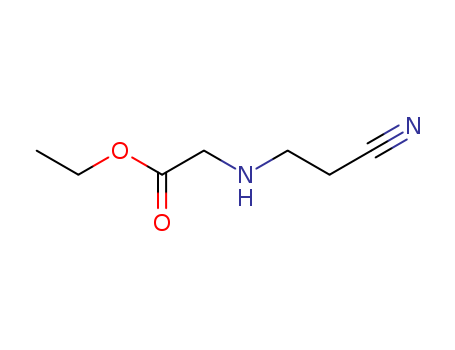
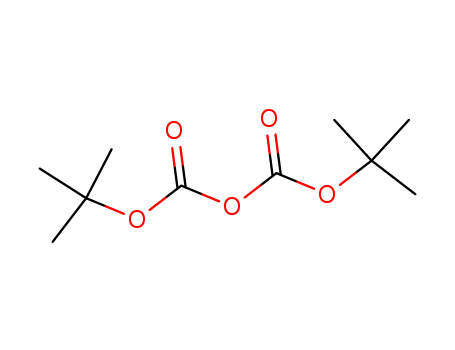
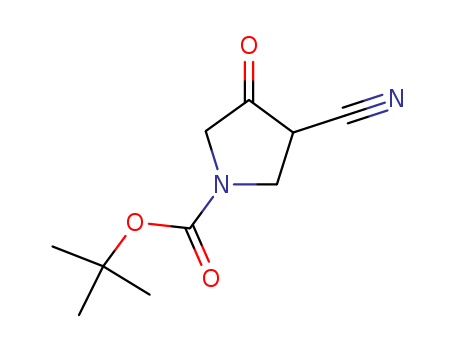
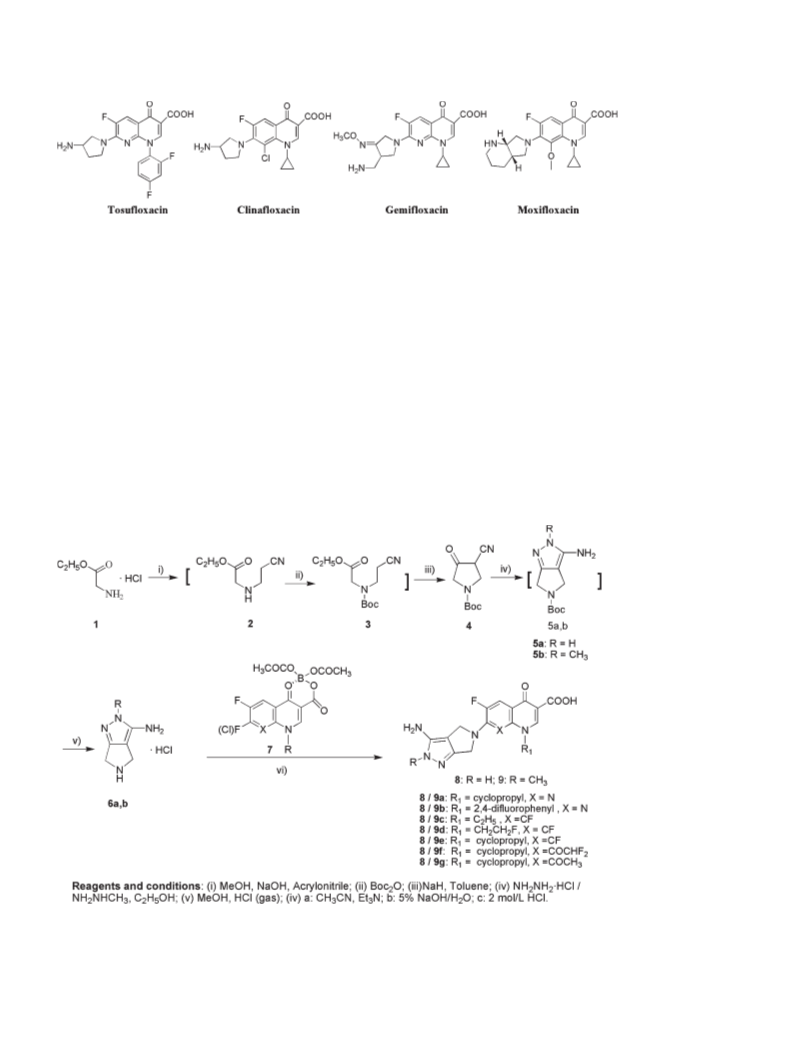
A series of novel 7-(3-aminopyrrolo[3,4-c]pyrazol-5(2H,4H,6H)-yl)-6-fluoro- 4-oxo-1,4-dihydroquinoline-3-carboxylic acid derivatives was designed, synthesized and characterized by 1H-NMR, MS and HRMS. These fluoroquinolones were evaluated for their in-vitro antibacterial activity against representative Gram-positive and Gram-negative strains. Generally, all of the target compounds display rather weak potency against the tested Gram-negative strains, but most of them exhibit good potency in inhibiting the growth of S. aureus including methicillin-resistant Staphylococcus aureus (MRSA) and Staphylococcus epidermidis including methicillin-resistant S. epidermidis (MRSE) (MIC: 0.125-8 μg/mL). In particular, the compound 9g is 2 to 32 fold more potent than gemifloxacin (GM), moxifloxacin (MX), gatifloxacin (GT), and levofloxacin (LV) against S. pneumoniae 08-3, K. pneumoniae 09-23, and P. aeruginosa ATCC27853, 4 to 32 fold more potent than MX, GM, and LV against K. pneumoniae 09-21, and more active than or comparable to the four reference drugs against P. aeruginosa 09-32. A series of novel fluoroquinolone derivatives was designed and synthesized. All target compounds display rather weak potency against the tested Gram-negative strains, but most of them exhibit good potency in inhibiting the growth of Staphylococcus aureus and Staphylococcus epidermidis. In particular, the compound 9g is the most promising drug.
Discovery of gemifloxacin (Factive, LB20304a): A quinolone of new a generation
Hong, Chang Yong
Novel quinolone antibacterials, which bear an alkyloxime substituent in the 4-position and an aminomethyl substituent in the 3-position of the pyrrolidine ring, have been designed and synthesized. These fluoroquinolones were found to possess extremely potent antimicrobial activity against Gram-positive organisms including resistant strains such as methicillin-resistant Staphylococcus aureus (MRSA). Among these compounds our development candidate, Gemifloxacin (Factive, LB20304a), showed the best in vivo efficacy and pharmacokinetic profile in animals, as well as good safety pharmacological properties. Gemifloxacin was found to be especially effective against respiratory tract infections that account for over 70% of all infections. With once-a-day dosage, potency against respiratory tract infections such as chronic bronchitis and pneumonia was ensured without any significant side effect. In December 1999, Gemifloxacin filed a NDA for marketing approval to the US Food and Drug Administration. Copyright