Relevant articles and documents
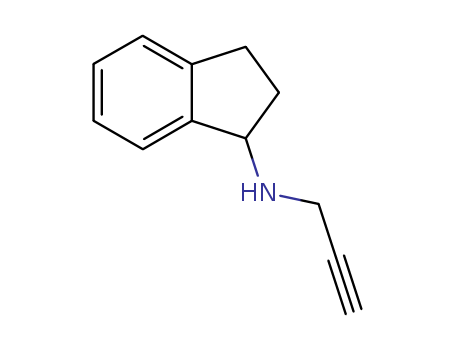
In silico and in vitro antioxidant and anticancer activity profiles of urea and thiourea derivatives of 2,3-dihydro-1H-inden-1-amine
Chandrasekhar, Mandala Rajendra, Wudayagiri Raju, Chamarthi Naga Seshaiah, Kalluru Syam Prasad, Gandavaram Umapriya, Kollu Venkataramaiah, Chintha
Synthesis of a series of new urea and thiourea compounds have been accomplished by the reaction of 2,3-dihydro-1H-inden-1-amine with various phenyl isocyanates and isothiocyanates. These compounds were evaluated for their antioxidant activity by using 1,1-diphenyl-2-picrylhydrazyl (DPPH) radical and nitric oxide (NO) radical scavenging assay methods including IC50 values. Some of the compounds exhibited potential activity in the two tested methods. Among the series of compounds, urea derivative linked with 4-bromo phenyl ring (4b), and thiourea derivatives bonded with phenyl ring (4e), 4-fluoro phenyl ring (4f) and 4-nitro pheyl ring (4h) were found to exhibit promising anti oxidant activity with low IC50 values. Where four of the title comounds exhibited higher bindig energies than the reference compound (Imatinib) in in silico molecular docking studies with Aromatase. All the synthesized compounds were characterized by IR, 1H, 13C NMR and mass spectral data.
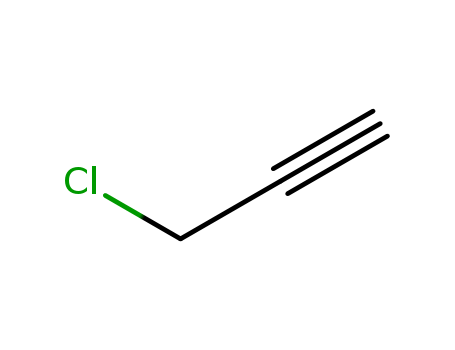
Isomerization and decomposition of chloromethylacetylene
Kumaran, Soundararajan S. Lim, Kee P. Michael, Joe V. Tilson, Jeffrey L. Suslensky, Aya Lifshitz, Assa
The isomerization and thermal decomposition of chloromethylacetylene (CMA) has been studied with two shock tube techniques. The first experiment (Jerusalem) utilizes single-pulse shock tube methods to measure the isomerization rate of CMA to chloroallene. In addition, equilibrium constants can be estimated at ~1200 K. The second experiment (Argonne) monitors Cl-atom formation at temperatures above ~1150 K. Absolute yield measurements have been performed over the 1200-1700 K range and indicate that two decomposition channels contribute to CMA destruction, namely, Cl fission and HCl elimination. The results show that the branching fraction between processes is temperature dependent. Therefore, direct Cl-atom fission is accompanied by molecular elimination, undoubtedly giving HCl and one or more isomers of C3H2. MP2 6-31G(d,p) ab initio electronic structure calculations have been used to determine vibration frequencies and moments of inertia for three C3H3Cl isomers. Using these quantities, the experimental equilibrium constants required that ΔH00(CH2Cl-C≡CH ? CHCl=C=CH2) = -0.24 kcal mole-1. A potential energy surface pertinent to the present system has been constructed, and RRKM calculations have been carried out in order to explain the isomerization rates. The isomerization data can be explained with E0 = 52.3 kcal mole-1 and 〈ΔEdown〉 = 225 cm-1. Subsequent semi-empirical Troe and RRKM-Gorin modeling of the Cl atom rate data require E0 = (67.5 ± 0.5) kcal mole-1 with a (ΔEdown) = (365 ± 90) cm-1. This suggests a heat of formation for propargyl radicals of (79.0 ± 2 5) kcal mole-1.