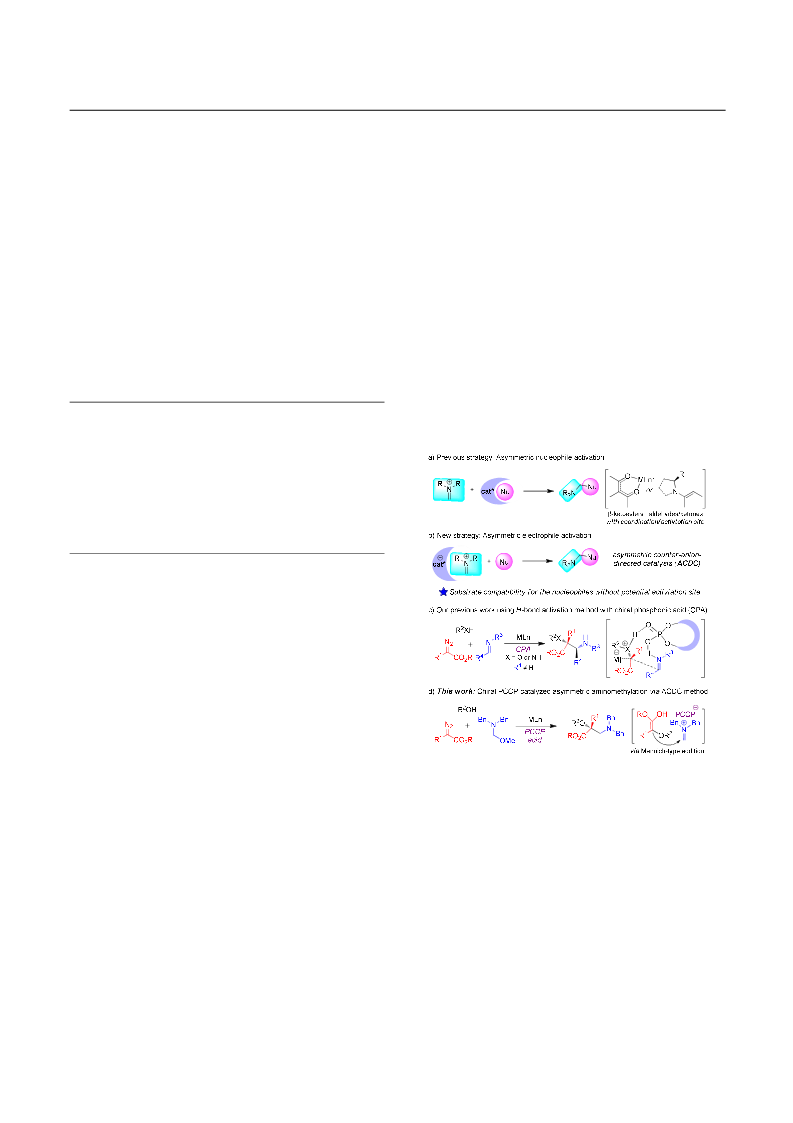Relevant articles and documents
Asymmetric Counter-Anion-Directed Aminomethylation: Synthesis of Chiral β-Amino Acids via Trapping of an Enol Intermediate
Kang, Zhenghui Wang, Yongheng Zhang, Dan Wu, Ruibo Xu, Xinfang Hu, Wenhao
A novel enantioselective aminomethylation reaction of diazo compound, alcohol and α-aminomethyl ether enabled by asymmetric counteranion-directed catalysis is disclosed that offers an efficient and convenient access to furnish optically active α-hydroxyl-β-amino acids in high yield with high to excellent enantioselectivities. Control experiments and DFT calculations indicate that the transformation proceeds through trapping the in situ generated enol intermediate with methylene iminium ion, and the asymmetric induction was enabled by chiral pentacarboxycyclopentadiene anion via H-bonding and electrostatic interaction.
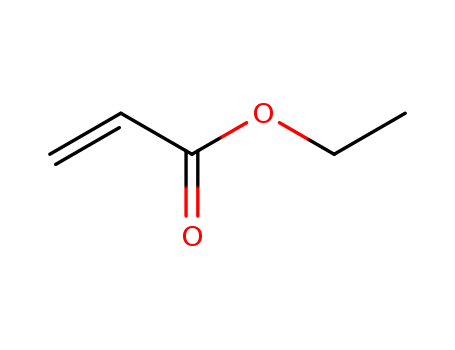
Liberation of acrylates from nickelalactones via Ni─O ring opening with alkyl iodides
Zhang, Zhizhi Guo, Fangjie Kühn, Fritz E. Sun, Jing Zhou, Mingdong Fang, Xiangchen
The utilization of carbon dioxide as a feedstock for the production of raw chemicals is of high current industrial interest. One attractive reaction is the transformation of carbon dioxide into acrylic acid or acrylates. The cleavage of the Ni─O bond of nickelalactones may result in the formation of acrylates. In this work, C2H5I, CF3CH2I and CF3I are studied as alkylation reagents for the Ni─O ring opening of nickelalactones. The results indicate that both C2H5I and CF3CH2I are able to release acrylates from nickelalactones. Based on the experimental evidence and literature precedents, a mechanism – proceeding via Ni─O ring opening of nickelalactone, β-H elimination to release the acrylate and reductive elimination for recovery of the Ni(0) species – is proposed.