Relevant articles and documents
Fundamental Insights into Photoelectrocatalytic Hydrogen Production with a Hole-Transport Bismuth Metal-Organic Framework
García-Sánchez, Alba Gomez-Mendoza, Miguel Barawi, Mariam Villar-Garcia, Ignacio J. Liras, Marta Gándara, Felipe De La Pe?a O'Shea, Víctor A.
Solar fuels production is a cornerstone in the development of emerging sustainable energy conversion and storage technologies. Light-induced H2 production from water represents one of the most crucial challenges to produce renewable fuel. Metal-organic frameworks (MOFs) are being investigated in this process, due to the ability to assemble new structures with the use of suitable photoactive building blocks. However, the identification of the reaction intermediates remains elusive, having negative impacts in the design of more efficient materials. Here, we report the synthesis and characterization of a new MOF prepared with the use of bismuth and dithieno[3,2-b:2′,3′-d]thiophene-2,6-dicarboxylic acid (DTTDC), an electron-rich linker with hole transport ability. By combining theoretical studies and time-resolved spectroscopies, such as core hole clock and laser flash photolysis measurements, we have completed a comprehensive study at different time scales (fs to ms) to determine the effect of competitive reactions on the overall H2 production. We detect the formation of an intermediate radical anion upon reaction of photogenerated holes with an electron donor, which plays a key role in the photoelectrocatalytic processes. These results shed new light on the use of MOFs for solar fuel production.
Dithienothiophene (DTT)-based dyes for dye-sensitized solar cells: Synthesis of 2,6-dibromo-DTT
Kwon, Tae-Hyuk Armel, Vanessa Nattestad, Andrew MacFarlane, Douglas R. Bach, Udo Lind, Samuel J. Gordon, Keith C. Tang, Weihua Jones, David J. Holmes, Andrew B.
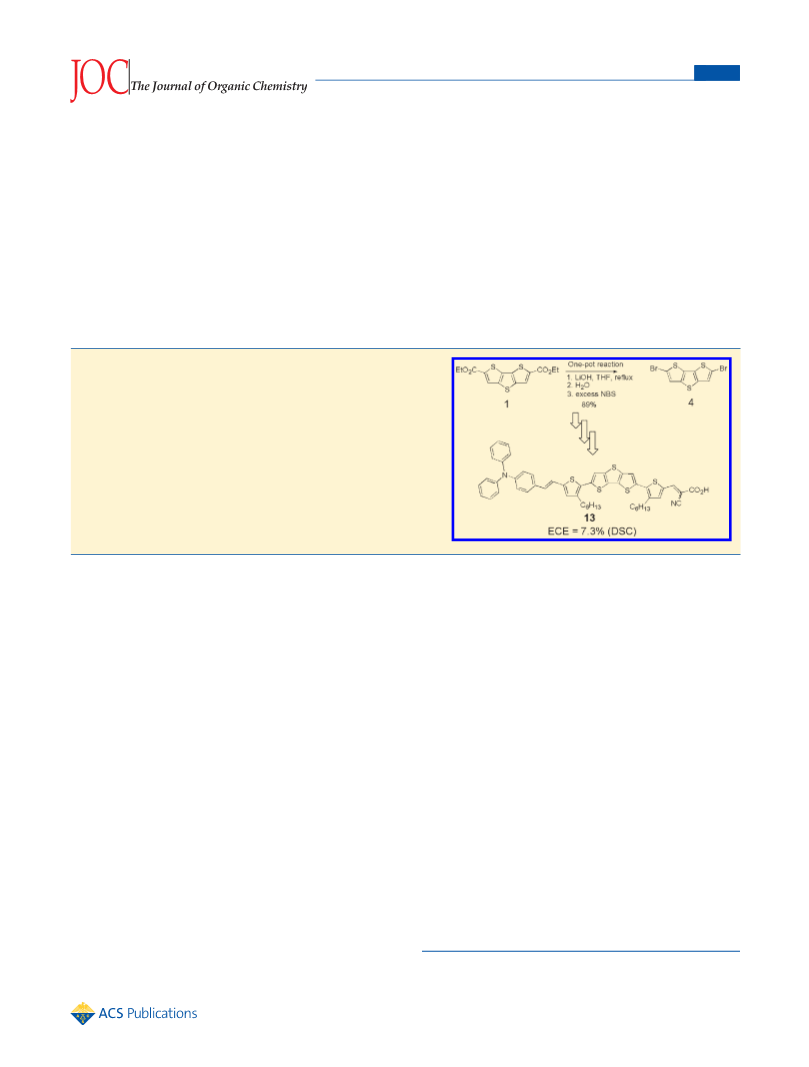
A one-pot synthesis of 2,6-dibromodithieno[3,2-b;2′,3′-d] thiophene (dibromo-DTT, 4) was developed. A key step was bromodecarboxylation of DTT-2,6-dicarboxylic acid, obtained by saponification of the diester 1. The donor-acceptor dye DAHTDTT (13), based o

![2',3'-d]thiophene-2,5-dicarboxylic acid diethyl ester](http://file1.lookchem.com/300w/substances/2022-03-29-06/d9a46968-95f4-4bbf-bf70-cdc70146d177.png)

![Dithieno[3,2-b:2',3'-d]Thiophene-2,6-Dicarboxylic Acid](http://file1.lookchem.com/300w/substances/2022-03-19-01/91c216f4-31fd-4a54-abe3-54f3f08a2d87.png)