Relevant articles and documents
Transformations of myo-inositol hexa-0-nitrate under the action of amines
Kuznetsov Sukhov Pichugin Ershov
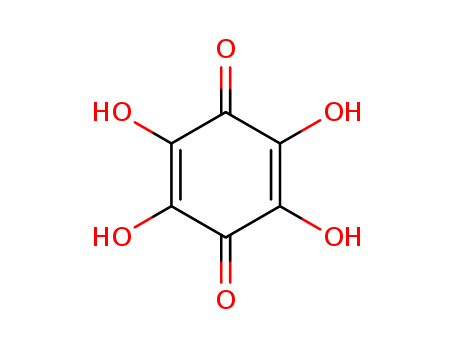
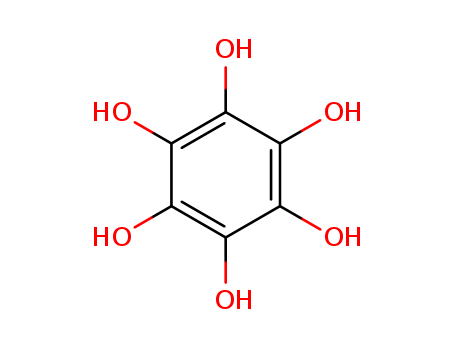
Reactions of myo-inositiol hexa-0-nitrate with ammonia and primary amines yield tetrahydroxy-1,4-benzoquinone derivatives, viz., its tetraammonium salt and its diimines, respectively. Reactions with secondary and tertiary amines give salts of rhodizonic acid, which are converted into salts of croconic acid under certain conditions. The reactions with secondary amines involve intermediate formation of radical species, which were dectected by ESR spectroscopy. A scheme for the chemical transformations of myo-inositol hexa-0-nitrate under the action of amines was proposed.
Synthesis and self-assembly of a heteroarm star amphiphile with 12 alternating arms and a well-defined core
Teng, Jing Zubarev, Eugene R.
We report on a stepwise synthesis of a heteroarm starlike amphiphile containing 12 alternating arms (six polystyrene and six poly(acrylic acid)) connected to a hexabiphenyl aromatic core. The synthesis does not involve polymerization, and only commercially available precursors are used. Most importantly, this amphiphile undergoes self-assembly into spherical and wormlike cylindrical micelles in aqueous and methanol solutions, and forms reverse 1D micellar structures in chloroform. This remarkable morphological diversity of the reported amphiphile 1 is believed to be a direct consequence of its well-defined molecular architecture. Copyright