Relevant articles and documents
Engineering cyclohexanone monooxygenase for the production of methyl propanoate
Van Beek, Hugo L. Romero, Elvira Fraaije, Marco W.
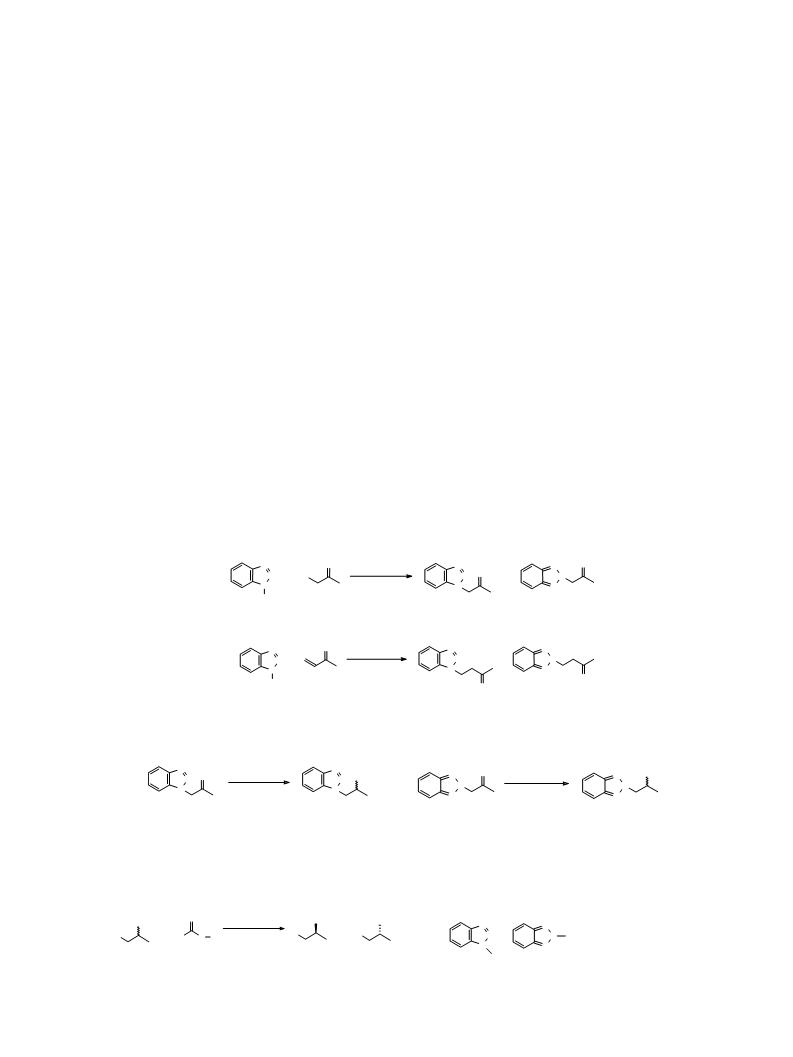
A previous study showed that cyclohexanone monooxygenase from Acinetobacter calcoaceticus (AcCHMO) catalyzes the Baeyer-Villiger oxidation of 2-butanone, yielding ethyl acetate and methyl propanoate as products. Methyl propanoate is of industrial interest as a precursor of acrylic plastic. Here, various residues near the substrate and NADP+ binding sites in AcCHMO were subjected to saturation mutagenesis to enhance both the activity on 2-butanone and the regioselectivity toward methyl propanoate. The resulting libraries were screened using whole cell biotransfor-mations, and headspace gas chromatography-mass spectrometry was used to identify improved AcCHMO variants. This revealed that the I491A AcCHMO mutant exhibits a significant improvement over the wild type enzyme in the desired regioselectivity using 2-butanone as a substrate (40% vs 26% methyl propanoate, respectively). Another interesting mutant is the T56S AcCHMO mutant, which exhibits a higher conversion yield (92%) and kcat (0.5 s-1) than wild type AcCHMO (52% and 0.3 s-1, respectively). Interestingly, the uncoupling rate for the T56S AcCHMO mutant is also significantly lower than that for the wild type enzyme. The T56S/I491A double mutant combined the beneficial effects of both mutations leading to higher conversion and improved regioselectivity. This study shows that even for a relatively small aliphatic substrate (2-butanone), catalytic efficiency and regioselectivity can be tuned by structure-inspired enzyme engineering.
Preparation of various enantiomerically pure (benzotriazol-1-yl)- and (benzotriazol-2-yl)-alkan-2-ols
Pchelka, Beata K. Loupy, Andre Petit, Alain
(S)-(-)-(Benzotriazol-1-yl)- and (S)-(-)-(benzotriazol-2-yl)-alkan-2-ols 7a-9a, 7b-9b and their (R)-(+)-acetates 10a-12a and 10b-12b were prepared in high enantiomeric excess via lipase from Pseudomonas fluorescens (Amano AK) catalyzed enantioselective acetylation of racemic alcohols 4a-6a and 4b-6b with vinyl acetate in tert-butyl methyl ether or toluene at 23 °C. The enantioselectivity of this transformation was dependent on the length of the alkyl chain with E-values ranging from 30 to 57. Several benzotriazole substituted ketones 1a-3a and 1b-3b were synthesized from 1H-benzotriazole and corresponding haloketones. These compounds were stereoselectively reduced with Baker's yeast in water or in organic solvent containing 5% v/v of water at 30 °C to give the (S)-(-)-alcohol. Better stereoselectivity was observed in the kinetic resolution of racemic alcohols 4a-6a and 4b-6b (ee = 69-92% at 44-52% conversion) compared to reduction of corresponding prochiral ketones 1a-3a and 1b-3b with Baker's yeast (ee = 40-67% at 39-89% conversion). Enhanced enantioselectivities were observed at lower temperatures.

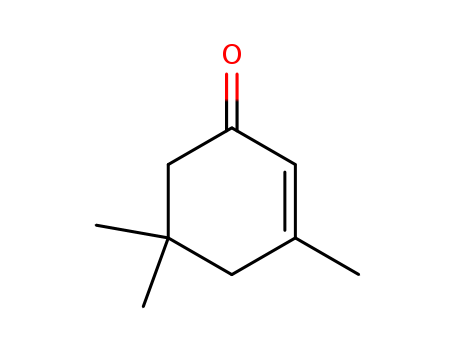

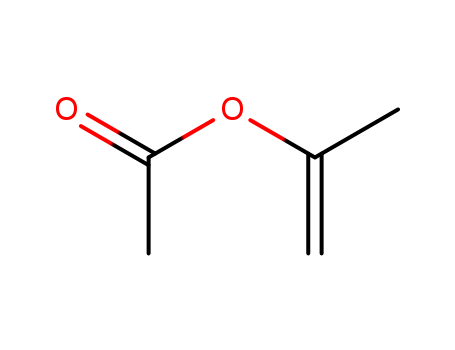

![7-Acetoxy-4,4,6,7-tetramethylbicyclo[4,2,0]octan-2-one](http://www.lookchem.com/300w/casimage/2011-02-22-00/66016-89-5.gif)