Relevant articles and documents
Structure of a monoclinic polymorph of N-[4-(acetyloxy)-phenyl]acetamide, a prodrug of acetaminophen
Caira De Wet Gerber
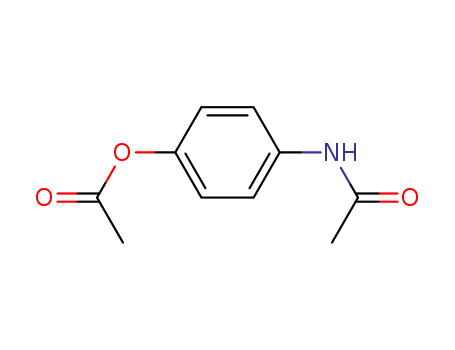
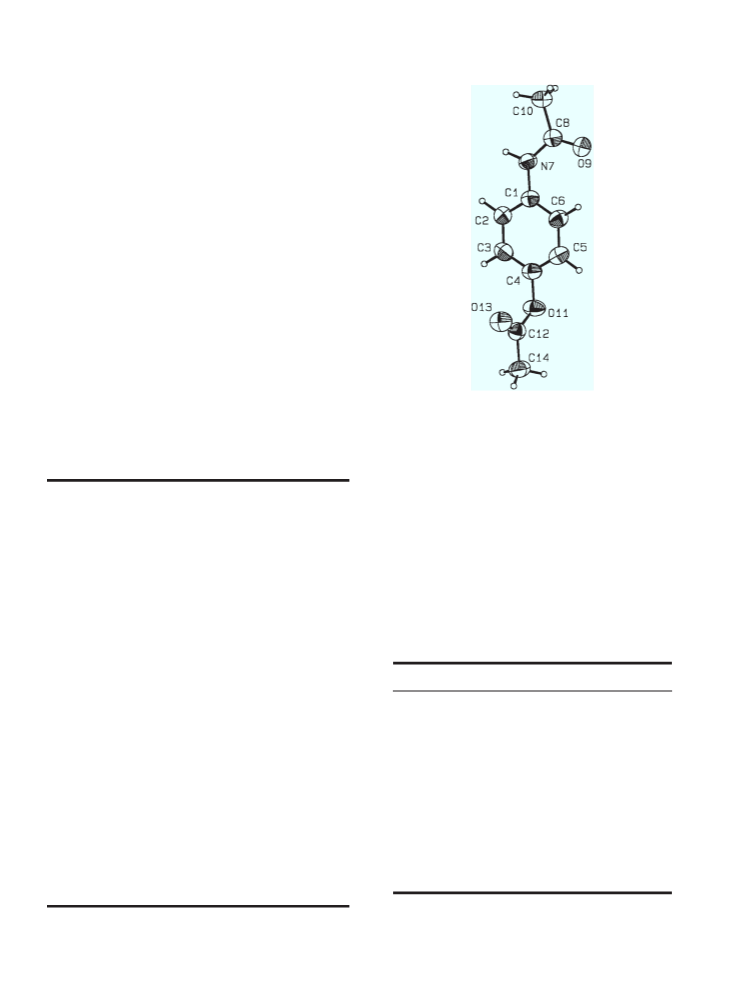
The preparation, single crystal x-ray structure and the computed powder XRD pattern of a monoclinic polymorph of N-[4-(acetyloxy)phenyl]acetamide, a prodrug of acetaminophen, are reported. The polymorph crystallizes in the space group P21/n with Z = 4 and unit cell dimensions a = 7.219(2), b = 8.015(2), c = 16.575(2) A, β = 92.07(1)°, and V = 958.4(4) A3. Infinite spiral molecular arrays result from intermolecular head-to-tail hydrogen bonding between the amidic H atom of one molecule and the acetoxy carbonyl oxygen atom of a 21-related molecule.
Synthesis and antiproliferative activity of new 1,2,3-triazole/flavone hybrid heterocycles against human cancer cell lines
Sowjanya Jayaprakash Rao Murthy
A series of new 1,2,3-triazole/flavone hybrid heterocycles were synthesized from 6-amino flavone via key intermediate N-propargyl flavone 6 by adopting the Sharpless Click reaction. Copper(I) catalyzed 1,3-dipolar cycloaddition reaction that gave products in high yields. All the synthesized compounds were screened for their in vitro antiproliferative activity against four human cancer cell lines, HeLa (cervical cancer cell line), MIA PaCa (pancreatic cancer cell line), MDA-MB-231 (breast cancer cell line), and IMR 32 (neuroblastoma cancer cell line). Compounds 7a, 7b, 7d, 7g (GI50 = 0.01–0.68 μM) demonstrated promising antiproliferative activity.