Products Categories
| CAS No.: | 6576-51-8 |
|---|---|
| Name: | distamycin A hydrochloride from*streptomyces dist |
| Article Data: | 3 |
| Molecular Structure: | |
|
|
|
| Formula: | C22H27 N9 O4 . Cl H |
| Molecular Weight: | 517.975 |
| Synonyms: | 1H-Pyrrole-2-carboxamide,N-[5-[[(3-amino-3-iminopropyl)amino]carbonyl]-1-methyl-1H-pyrrol-3-yl]-4-[[[4-(formylamino)-1-methyl-1H-pyrrol-2-yl]carbonyl]amino]-1-methyl-,monohydrochloride (9CI); N,4':N',4''-Ter[pyrrole-2-carboxamide],N''-(2-amidinoethyl)-4-formamido-1,1',1''-trimethyl-, hydrochloride (7CI);N,4':N',4''-Ter[pyrrole-2-carboxamide],N''-(2-amidinoethyl)-4-formamido-1,1',1''-trimethyl-, monohydrochloride (8CI);Distamycin A hydrochloride; Distamycin hydrochloride; MEN 10399; Stallimycinhydrochloride |
| EINECS: | 229-505-6 |
| Melting Point: | 184-187° (Arcamone, 1967). Also reported as 189-193° from ethanol-ethyl acetate (Bialer) |
| Boiling Point: | 715.4 °C at 760 mmHg |
| Flash Point: | 386.4 °C |
| Solubility: | CLEAR YELLOW TO GREEN SOLUTION AT 20MG/ML IN ETHANOL |
| Appearance: | YELLOW TO YELLOW WITH A BROWN CAST POWDER |
| Hazard Symbols: |
 Xi Xi
|
| Risk Codes: | 43 |
| Safety: | Poison by intraperitoneal and intravenous routes. Mutation data reported. When heated to decomposition it emits very toxic fumes of NOx and HCl. |
| PSA: | 181.06000 |
| LogP: | 3.67870 |
- 81281-59-67-Benzylideneaminotheophylline
- 82993-81-5D-threo-Ritalinic acid hydrochloride
- 73441-42-6METHYL-5-CHLORO-2,2-DIMETHYLVALERATE
- 68439-39-4Poly(oxy-1,2-ethanediyl), alpha-(2-ethylhexyl)-omega-hydroxy-,
- 852475-26-4MC1568
- 958254-66-51H-Imidazo[4,5-b]pyridine-2-carboxaldehyde, 1-methyl-, hydrochloride
- 99170-93-1N-Methyl-2-oxazolamine
- 717878-06-31-(4-fluorophenyl)-4-nitro-1H-imidazole
- 914458-26-7[5-(2-fluorophenyl)-1-pentyl-1H-pyrrol-3-yl]-1-naphthalenyl-Methanone
- 894852-01-87-BROMO-2,2-DIMETHYL-2H-PYRIDO[3,2-B][1,4]OXAZIN-3(4H)-ONE
- 77727-43-6
N-Succinimidyl 4-<<<4-<<<4-(Formylamino)-1-methylpyrrol-2-yl>carbonyl>amino>-1-methylpyrrol-2-yl>carbonyl>amino>-1-methylpyrrole-2-carboxylate

- 77152-88-6
(1-amino-3-ammoniopropylidene)ammonium dihydrobromide

- 6576-51-8
distamycin A hydrochloride

| Conditions | Yield |
|---|---|
| With hydrogenchloride; sodium hydrogencarbonate In 1,4-dioxane; water 1.) 15 min, ice-cooled, 2.) room temperature, overnight; Yield given; |
- 3786-88-7
1-methyl-4-<1-methyl-4-(1-methyl-4-aminopyrrole-2-carboxamido)pyrrole-2-carboxamido>pyrrole-2-carboxamidopropionamidine hydrochloride

- 3197-61-3
N-formylimidazole

- 6576-51-8
distamycin A hydrochloride

| Conditions | Yield |
|---|---|
| In tetrahydrofuran; methanol at -40℃; for 0.5h; Yield given; |

| Conditions | Yield |
|---|---|
| Multi-step reaction with 8 steps 1: 43 percent / 1.) 70percent nitric acid, acetic anhydride / 1.) a) -15 deg C, 0.5 h, b) RT, 20 min 2: SOCl2 / tetrahydrofuran / 0.08 h / Heating 3: Hunig's base / tetrahydrofuran / -20 - 20 °C 4: 1.) H2, 2.) HCl / 10percent Pd/C / 1.) methanol, 40 deg C, 1 atm, 2.) isopropanol, -30 deg C 5: 64 percent / 1,8-bis(dimethylamino)naphthalene, dicyclohexylcarbodiimide / tetrahydrofuran; dimethylformamide / 3 h / Ambient temperature 6: 1.) HCl, 2.) NH3 / 1.) EtOH, RT, 1.5 h, 2.) EtOH, 1 h, RT 7: 70 percent / H2 / 10percent Pd/C / methanol / 40 °C 8: methanol; tetrahydrofuran / 0.5 h / -40 °C View Scheme | |
| Multi-step reaction with 8 steps 1: 43 percent / 1.) 70percent nitric acid, acetic anhydride / 1.) a) -15 deg C, 0.5 h, b) RT, 20 min 2: SOCl2 / tetrahydrofuran / 0.08 h / Heating 3: 1.) H2, 2.) Hunig's base / 10percent Pd/C / 1.) MeOH, 1 atm, 2.) THF, 3) THF, RT, 30 min 4: 95 percent / NaOH / H2O; ethanol / Heating 5: 64 percent / 1,8-bis(dimethylamino)naphthalene, dicyclohexylcarbodiimide / tetrahydrofuran; dimethylformamide / 3 h / Ambient temperature 6: 1.) HCl, 2.) NH3 / 1.) EtOH, RT, 1.5 h, 2.) EtOH, 1 h, RT 7: 70 percent / H2 / 10percent Pd/C / methanol / 40 °C 8: methanol; tetrahydrofuran / 0.5 h / -40 °C View Scheme |
- 13138-78-8
1-methyl-4-nitro-pyrrol-2-carboxylic acid

- 6576-51-8
distamycin A hydrochloride

| Conditions | Yield |
|---|---|
| Multi-step reaction with 7 steps 1: SOCl2 / tetrahydrofuran / 0.08 h / Heating 2: Hunig's base / tetrahydrofuran / -20 - 20 °C 3: 1.) H2, 2.) HCl / 10percent Pd/C / 1.) methanol, 40 deg C, 1 atm, 2.) isopropanol, -30 deg C 4: 64 percent / 1,8-bis(dimethylamino)naphthalene, dicyclohexylcarbodiimide / tetrahydrofuran; dimethylformamide / 3 h / Ambient temperature 5: 1.) HCl, 2.) NH3 / 1.) EtOH, RT, 1.5 h, 2.) EtOH, 1 h, RT 6: 70 percent / H2 / 10percent Pd/C / methanol / 40 °C 7: methanol; tetrahydrofuran / 0.5 h / -40 °C View Scheme | |
| Multi-step reaction with 7 steps 1: SOCl2 / tetrahydrofuran / 0.08 h / Heating 2: 1.) H2, 2.) Hunig's base / 10percent Pd/C / 1.) MeOH, 1 atm, 2.) THF, 3) THF, RT, 30 min 3: 95 percent / NaOH / H2O; ethanol / Heating 4: 64 percent / 1,8-bis(dimethylamino)naphthalene, dicyclohexylcarbodiimide / tetrahydrofuran; dimethylformamide / 3 h / Ambient temperature 5: 1.) HCl, 2.) NH3 / 1.) EtOH, RT, 1.5 h, 2.) EtOH, 1 h, RT 6: 70 percent / H2 / 10percent Pd/C / methanol / 40 °C 7: methanol; tetrahydrofuran / 0.5 h / -40 °C View Scheme | |
| Multi-step reaction with 8 steps 1: H2, 1.0 M Na2CO3(aq.) / 5percent Pd/C / 3.5 h / 3102.9 Torr / Ambient temperature 2: tetrahydrofuran; diethyl ether / 1.) 1 h, ice cooled, 2.) 2 h, ambient temperature 3: 1-ethyl-3-<3-(dimethylamino)propyl>carbodiimide hydrochloride / dimethylformamide / 40 °C 4: TFA / CH2Cl2 5: EDC / dimethylformamide / 40 °C 6: H2 / 5percent Pd/C / dimethylformamide / 2 h / 3102.9 Torr / Ambient temperature 7: 88 percent / DCC / dimethylformamide / 1.) 70 min, 0 deg C, 2.) 40 deg C, overnight 8: 1.) NaHCO3, 2.) HCl / H2O; dioxane / 1.) 15 min, ice-cooled, 2.) room temperature, overnight View Scheme |
- 97950-76-0
1-methyl-4-(1-methyl-4-nitropyrrole-2-carboxamido)pyrrole-2-carboxylic acid

- 6576-51-8
distamycin A hydrochloride

| Conditions | Yield |
|---|---|
| Multi-step reaction with 4 steps 1: 64 percent / 1,8-bis(dimethylamino)naphthalene, dicyclohexylcarbodiimide / tetrahydrofuran; dimethylformamide / 3 h / Ambient temperature 2: 1.) HCl, 2.) NH3 / 1.) EtOH, RT, 1.5 h, 2.) EtOH, 1 h, RT 3: 70 percent / H2 / 10percent Pd/C / methanol / 40 °C 4: methanol; tetrahydrofuran / 0.5 h / -40 °C View Scheme |
- 69910-20-9
methyl 1-methyl-4-(1-methyl-4-nitropyrrole-2-carboxamido)pyrrole-2-carbonate

- 6576-51-8
distamycin A hydrochloride

| Conditions | Yield |
|---|---|
| Multi-step reaction with 5 steps 1: 95 percent / NaOH / H2O; ethanol / Heating 2: 64 percent / 1,8-bis(dimethylamino)naphthalene, dicyclohexylcarbodiimide / tetrahydrofuran; dimethylformamide / 3 h / Ambient temperature 3: 1.) HCl, 2.) NH3 / 1.) EtOH, RT, 1.5 h, 2.) EtOH, 1 h, RT 4: 70 percent / H2 / 10percent Pd/C / methanol / 40 °C 5: methanol; tetrahydrofuran / 0.5 h / -40 °C View Scheme |
- 28494-51-1
1-methyl-4-nitro-1H-pyrrole-2-carbonyl chloride

- 6576-51-8
distamycin A hydrochloride

| Conditions | Yield |
|---|---|
| Multi-step reaction with 6 steps 1: Hunig's base / tetrahydrofuran / -20 - 20 °C 2: 1.) H2, 2.) HCl / 10percent Pd/C / 1.) methanol, 40 deg C, 1 atm, 2.) isopropanol, -30 deg C 3: 64 percent / 1,8-bis(dimethylamino)naphthalene, dicyclohexylcarbodiimide / tetrahydrofuran; dimethylformamide / 3 h / Ambient temperature 4: 1.) HCl, 2.) NH3 / 1.) EtOH, RT, 1.5 h, 2.) EtOH, 1 h, RT 5: 70 percent / H2 / 10percent Pd/C / methanol / 40 °C 6: methanol; tetrahydrofuran / 0.5 h / -40 °C View Scheme | |
| Multi-step reaction with 6 steps 1: 1.) H2, 2.) Hunig's base / 10percent Pd/C / 1.) MeOH, 1 atm, 2.) THF, 3) THF, RT, 30 min 2: 95 percent / NaOH / H2O; ethanol / Heating 3: 64 percent / 1,8-bis(dimethylamino)naphthalene, dicyclohexylcarbodiimide / tetrahydrofuran; dimethylformamide / 3 h / Ambient temperature 4: 1.) HCl, 2.) NH3 / 1.) EtOH, RT, 1.5 h, 2.) EtOH, 1 h, RT 5: 70 percent / H2 / 10percent Pd/C / methanol / 40 °C 6: methanol; tetrahydrofuran / 0.5 h / -40 °C View Scheme |
- 13138-76-6
1-methyl-4-nitropyrrole-2-carboxylic acid methyl ester

- 6576-51-8
distamycin A hydrochloride

| Conditions | Yield |
|---|---|
| Multi-step reaction with 8 steps 1: 92 percent / aq. NaOH / Heating 2: SOCl2 / tetrahydrofuran / 0.08 h / Heating 3: Hunig's base / tetrahydrofuran / -20 - 20 °C 4: 1.) H2, 2.) HCl / 10percent Pd/C / 1.) methanol, 40 deg C, 1 atm, 2.) isopropanol, -30 deg C 5: 64 percent / 1,8-bis(dimethylamino)naphthalene, dicyclohexylcarbodiimide / tetrahydrofuran; dimethylformamide / 3 h / Ambient temperature 6: 1.) HCl, 2.) NH3 / 1.) EtOH, RT, 1.5 h, 2.) EtOH, 1 h, RT 7: 70 percent / H2 / 10percent Pd/C / methanol / 40 °C 8: methanol; tetrahydrofuran / 0.5 h / -40 °C View Scheme | |
| Multi-step reaction with 8 steps 1: 92 percent / aq. NaOH / Heating 2: SOCl2 / tetrahydrofuran / 0.08 h / Heating 3: 1.) H2, 2.) Hunig's base / 10percent Pd/C / 1.) MeOH, 1 atm, 2.) THF, 3) THF, RT, 30 min 4: 95 percent / NaOH / H2O; ethanol / Heating 5: 64 percent / 1,8-bis(dimethylamino)naphthalene, dicyclohexylcarbodiimide / tetrahydrofuran; dimethylformamide / 3 h / Ambient temperature 6: 1.) HCl, 2.) NH3 / 1.) EtOH, RT, 1.5 h, 2.) EtOH, 1 h, RT 7: 70 percent / H2 / 10percent Pd/C / methanol / 40 °C 8: methanol; tetrahydrofuran / 0.5 h / -40 °C View Scheme |
- 3185-95-3
1-methyl-4-nitro-1H-pyrrole-2-carboxylic acid (2-cyanoethyl)amide

- 6576-51-8
distamycin A hydrochloride

| Conditions | Yield |
|---|---|
| Multi-step reaction with 5 steps 1: 1.) H2, 2.) HCl / 10percent Pd/C / 1.) methanol, 40 deg C, 1 atm, 2.) isopropanol, -30 deg C 2: 64 percent / 1,8-bis(dimethylamino)naphthalene, dicyclohexylcarbodiimide / tetrahydrofuran; dimethylformamide / 3 h / Ambient temperature 3: 1.) HCl, 2.) NH3 / 1.) EtOH, RT, 1.5 h, 2.) EtOH, 1 h, RT 4: 70 percent / H2 / 10percent Pd/C / methanol / 40 °C 5: methanol; tetrahydrofuran / 0.5 h / -40 °C View Scheme |
- 2522-28-3
3-<1-methyl-4-<1-methyl-4-(1-methyl-4-nitropyrrole-2-carboxamido)pyrrole-2-carboxamido>pyrrole-2-carboxamido>propionitrile

- 6576-51-8
distamycin A hydrochloride

| Conditions | Yield |
|---|---|
| Multi-step reaction with 3 steps 1: 1.) HCl, 2.) NH3 / 1.) EtOH, RT, 1.5 h, 2.) EtOH, 1 h, RT 2: 70 percent / H2 / 10percent Pd/C / methanol / 40 °C 3: methanol; tetrahydrofuran / 0.5 h / -40 °C View Scheme |

What can I do for you?
Get Best Price
Chemistry
MW: 517.97
EINECS: 229-505-6
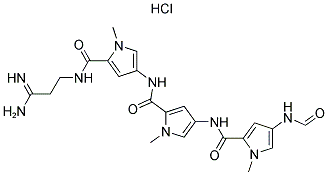
Following is the molecular structure of DISTAMYCIN A HYDROCHLORIDE:
Toxicity Data With Reference
| 1. | dni-mus:lym 1900 nmol/L | CBINA8 Chemico-Biological Interactions. 8 (1974),183. | ||
| 2. | oms-mus:lym 3200 nmol/L | CBINA8 Chemico-Biological Interactions. 8 (1974),183. | ||
| 3. | dnd-mam:lym 200 µmol/ | EJBCAI European Journal of Biochemistry. 26 (1972),81. | ||
| 4. | ipr-rat LD50:169 mg/kg | MDACAP Medicamentos de Actualidad. 13 (1977),319. | ||
| 5. | ipr-mus LD50:160 mg/kg | MDACAP Medicamentos de Actualidad. 13 (1977),319. | ||
| 6. | ivn-mus LD50:75 mg/kg | MEIEDD Merck Index. 11 (1989),1383. |
RTECS :WZ7110000
Safety Profile
Hazard Codes : Xi (Irritant)
Risk Statements: 43 (May cause sensitization by skin contact)
Safety Statements : 36/37 (Wear suitable protective clothing and gloves)
WGK Germany: 3
