- Cupric acetate
- Cupric acetate monohydrate
- Cupric acetylacetonate
- Cupric bromide
- Cupric carbonate
- Cupric carbonate basic
- Cupric chloride
- Cupric chloride dihydrate
- Cupric citrate
- Cupric fluoride
- 589-38-83-Hexanone
- 589-41-3Carbamic acid,N-hydroxy-, ethyl ester
- 589-43-5Hexane, 2,4-dimethyl-
- 58944-75-5Selenopentathionic acid([(HO)S(O)2S]2Se)
- 5894-59-7D-Galacturonic acid, 4-O-α-D-galactopyranuronosyl-
- 58-94-62H-1,2,4-Benzothiadiazine-7-sulfonamide,6-chloro-, 1,1-dioxide
- 5894-60-0Silane,trichlorohexadecyl-
- 5894-65-5Benzamide,N-(1,1-dimethylethyl)-
- 5894-79-1Benzeneacetic acid, alpha-formyl-, methyl ester
- 5895-35-22-Oxetanone,4-(trichloromethyl)-
Hot Products
- 104987-11-3Tacrolimus
- 141-53-7Sodium formate
- 8001-54-5Quaternary ammonium compounds, alkylbenzyldimethyl, chlorides
- 9003-39-8Povidone
- 10161-34-9Trenbolone acetate
- 402957-28-2Telaprevir
- 68-19-9Cyanocobalamin
- 7631-86-9Silicon dioxide
- 302-79-4Tretinoin
- 77-92-9Citric acid

|
Basic Information |

|
Post buying leads |

|
Suppliers |

| Name |
Cupric oxalate |
EINECS | 212-411-4 |
| CAS No. | 5893-66-3 | Density | 1.28g/cm3 |
| PSA | 52.60000 | LogP | -1.00110 |
| Solubility | N/A | Melting Point |
310°C |
| Formula | CuC2O4 | Boiling Point | 365.1 °C at 760 mmHg |
| Molecular Weight | 151.56 | Flash Point | 188.8 °C |
| Transport Information | 3288 | Appearance | Blue fine powder |
| Safety | 36 | Risk Codes | 20/21/22 |
| Molecular Structure |
|
Hazard Symbols | N/A |
| Synonyms |
Cupric oxalate |
Cupric oxalate Chemical Properties
IUPAC Name: Copper oxalate
Molecular Formula: C2CuO4
Molecular Weight: 151.57 g/mol
Molecular Structure of Copper oxalate (CAS NO.814-91-5):
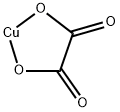
Flash Point: 188.8 °C
Boiling Point: 365.1 °C at 760 mmHg
Enthalpy of Vaporization: 67.15 kJ/mol
Appearance: Odorless bluish-white solid.
Vapour Pressure: 2.51E-06 mmHg at 25 °C
Water Solubility: insoluble
H-Bond Acceptor: 4
Exact Mass: 150.90926
MonoIsotopic Mass: 150.90926
Topological Polar Surface Area: 80.3
Heavy Atom Count: 7
Canonical SMILES: C(=O)(C(=O)[O-])[O-].[Cu+2]
InChI: InChI=1S/C2H2O4.Cu/c3-1(4)2(5)6;/h(H,3,4)(H,5,6);/q;+2/p-2
InChIKey: QYCVHILLJSYYBD-UHFFFAOYSA-L
EINECS: 212-411-4
Product Categories: organic-metal salt.
Cupric oxalate Uses
Copper oxalate (CAS NO.814-91-5) is used as a catalysts for organic reactions.
Cupric oxalate Safety Profile
Risk Statements: 20/21/22
R20/21/22:Harmful by inhalation, in contact with skin and if swallowed.
Safety Statements: 36
S36:Wear suitable protective clothing.
RIDADR: UN 2449
HazardClass: 6.1(b)
PackingGroup: III
Explodes weakly when heated slightly. See also OXALATES and COPPER COMPOUNDS.
Cupric oxalate Standards and Recommendations
DOT Classification: 6.1; Label: KEEP AWAY FROM FOOD
Cupric oxalate Specification
Copper oxalate (CAS NO.814-91-5) is also named as Caswell No. 248A ; Copper oxalate (CuC2O4) ; Copper(II) oxalate ; Cupric oxalate ; Cupric oxalate (1:1) ; EPA Pesticide Chemical Code 023305 ; Ethanedioic acid copper salt ; Ethanedioic acid, copper(2+) salt (1:1) ; HSDB 265 ; NSC 112246 ; NSC 86015 ; Oxalic acid copper(2+) salt (1:1) ; Copper, (ethanedioato(2-)-kappaO1,kappaO2)- . Copper oxalate (CAS NO.814-91-5) is odorless bluish-white solid. Cupric oxalate dissolves in aqueous ammonia and reacts as an acid to neutralize other bases as well. It can serve as a reducing agent in reactions that generate carbon dioxide. Inhalation causes irritation of nose and throat. Ingestion of very large amounts may produce symptoms of oxalate poisoning; watch for edema of the glottis and delayed constriction of esophagus. Contact with eyes causes irritation. It may form toxic carbon monoxide gas in fire.

