Med Chem Res (2014) 23:1581–1590
1585
water at 0–5 °C and stirred for 1 h. The resulting crystals
were isolated by filtration and dried to provide 3 as a white
powder. mp: 112 °C. Yield = 80.1 %. 1H NMR
(300 MHz, DMSO-d6): d 1.71 (2H, t), 2.36 (3H, s), 2.63
(2H, t), 3.48 (2H, s), 3.76 (H, s), 6.90–7.76 (8H, m). IR
transducer was connected to a MP150 data acquisition
system from BIOPAC Systems Inc. in the following order:
transducer, DA100C amplifier, universal interface module
100C, and a computerized system with the AcqKnowledge
3.8.1 program. The aortic rings were put into 10 mL iso-
lated organ baths containing K–H solution at 37 °C and
gassed with 95 % O2 and 5 % CO2. The initial tension was
set at 4 g, and the rings were allowed to stabilize for at
least 30 min to reach equilibrium. After the stabilization
period, the aortic rings were contracted with AVP (0.1 lM)
to prove the viability of the rings. Subsequently, an
observation time was given to record the maximum con-
traction of the ring, which was then washed three times
with K–H solution, and a new stabilization period was
allowed before testing the drugs.
(KBr): 3440, 2935, 2241, 1732 cm-1 13C NMR (75 MHz,
.
DMSO-d6): d 14.23, 21.67, 24.86, 50.04, 52.81, 120.99,
127.88, 129.04, 129.19, 130.47, 131.27, 133.14, 133.36,
135.28, 137.83, 144.36, 167.03.
1-[(4-methylphenyl)sulfonyl]-5-oxo-2,3,4,5-tetrahydro-
1H-1-benzazepine-4-carbonitrile (C9)
Potassium t-butoxide (1.76 g, 15.7 mmol) was added to a
solution of 3 (2.91 g, 7.81 mmol) in dry DMF (8.74 mL) at
an internal temperature of -10 °C. The mixture was then
warmed to 0–5 °C and stirred for 2 h. Water was poured
into the mixture at 0–5 °C, 2.2 mL of 37 % hydrochloric
acid was added dropwise, and the mixture was stirred at
5 °C for 1 h. The resulting solid was isolated by filtration
to give crude C9. A mixture of crude C9 in methanol was
stirred at reflux for 30 min, then cooled to 5 °C and stirred
for 1 h. The resulting crystals were isolated by filtration
and dried to afford C9 as a white powder. mp: 152–153 °C.
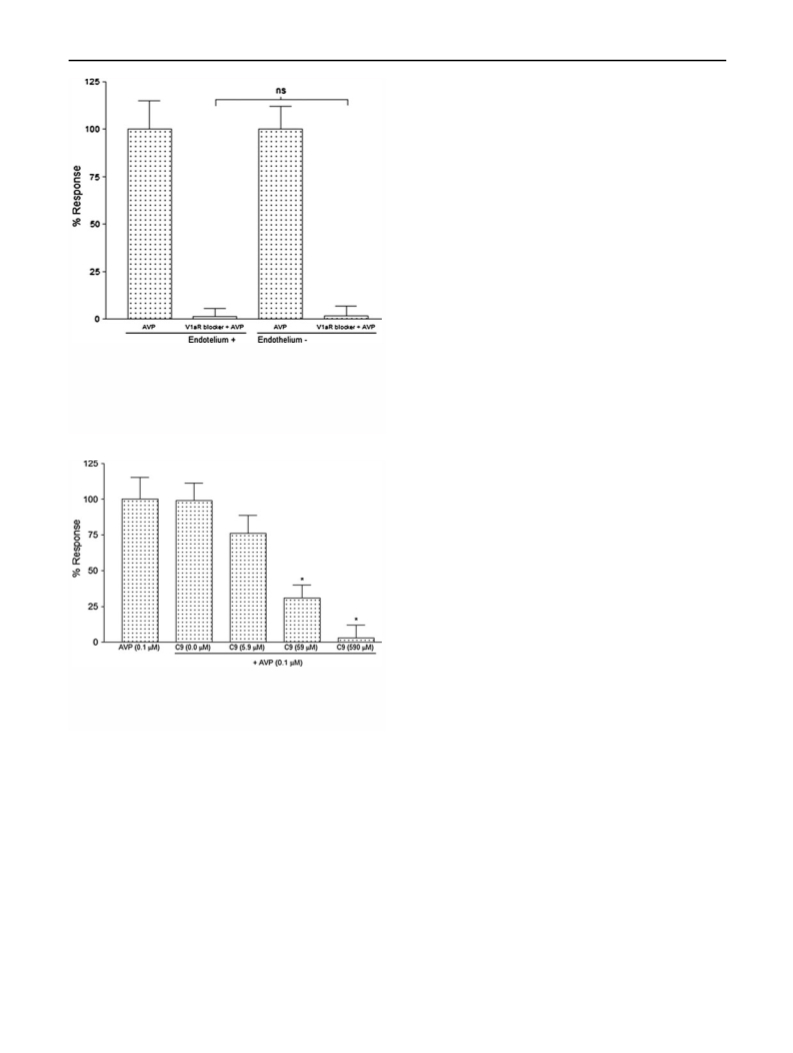
Influence of vascular endothelium on the maximum AVP-
induced contraction
To discriminate between the contractile effects of AVP on
V1aR of the VSM from the V2R-mediated vasodilator
effects from the vascular endothelium, two studies were
performed: (1) aorta rings with endothelium and (2) aorta
rings without endothelium. For these experiments, the
well-known V1aR peptide antagonist [(deamino-Pen1,
O-Me-Tyr2, Arg8)-Vasopressin, Sigma V1880] was used.
Assays started after the aortic ring viability test, and after
the third H–K wash, the V1aR antagonist (10 lM) was
added to the bath and was followed by a single pulse of
AVP (0.1 lM). The percentage of AVP-induced maximum
contraction was measured in the presence of V1aR antag-
onist. Finally, after three H–K washes, adrenaline (0.1 lM)
was added to assess the rings’ viability. Those rings that
did not respond to an adrenaline pulse were considered
non-viable and excluded from the study. These experi-
ments allowed us to determine that in our conditions,
vascular endothelium did not affect AVP-induced con-
tractions (Fig. 5).
1
Yield: 77.2 %. H NMR (300 MHz, DMSO-d6): d 2.09
(2H, m), 2.41 (3H, s), 3.39 (2H, br s), 7.31–7.58 (8H, m),
11.1 (1H, br s). IR (KBr): 3440, 2934, 2241, 1732 cm-1
.
13C NMR (75 MHz, DMSO-d6): d 21.79, 26.59, 56.64,
82.16, 119.10, 127.46, 129.27, 130.60, 132.00, 132.20,
135.22, 136.92, 137.54, 143.94, 166.04.
Biological effects
Blocking effects of C9 on AVP-induced aortic ring
contraction
Animals were anesthetized with sodium pentobarbital
´
(40 mg/kg of body weight; Holland de Mexico) and
underwent thoracolaparotomies. For each animal, the
descending thoracic aorta was removed and placed in a
Petri dish with Krebs–Henseleit (K–H) solution (NaCl
118 mM, KCl 4.7 mM, KH2PO4 1.2 mM, MgSO4Á2H2O
1.2 mM, CaCl2Á2H2O 2.5 mM, NaHCO3 25 mM, dextrose
11.7 mM and EDTA 0.026 mM). The adhering perivas-
cular fat and connective tissue were carefully removed, and
the segments were cut into rings of 3–4 mm in length. The
endothelium of the rings was or not mechanically removed
by inserting a rough surface wire into the vessel lumen. To
measure tension changes, two triangle-shaped parallel steel
strings were gently inserted into the ring, with one of the
steel strings used to fix the ring in the bottom of the iso-
lated organ chamber and the other connected to a tension
transducer (TSD125C from BIOPAC). In turn, the
The AVP-induced (0.1 lM) VSM contraction in aortic
rings with and without endothelium (Fig. 4) was blocked
with the V1a antagonist (deamino-Pen1, O-Me-Tyr2,
Arg8) Vasopressin, indicating that the vascular endothe-
lium did not affect the vasopressor effect of AVP, whereas
the V2-mediated endothelium vasodilator effect was not
evident.
Effect of C9 on the contractility of vascular smooth muscle
(VSM) from aortic rings
Having established that the endothelium did not signifi-
cantly affect the rings’ contraction, the blocking effect of
C9 on AVP-induced contraction was assayed by testing
several doses of C9 (vide infra). Five experimental groups
123

 Contreras-Romo, M. Citlalli
Contreras-Romo, M. Citlalli
 Correa-Basurto, Jose
Correa-Basurto, Jose
 Padilla-Martinez, Itzia
Padilla-Martinez, Itzia
 Martinez-Archundia, Marlet
Martinez-Archundia, Marlet
 Martinez-Ramos, Federico
Martinez-Ramos, Federico
 Slusarz, Magdalena J.
Slusarz, Magdalena J.
 Lopez-Perez, Gilberto
Lopez-Perez, Gilberto
 Quintanar-Stephano, Andres
Quintanar-Stephano, Andres