T.M. Suzuki et al. / Journal of Molecular Catalysis A: Chemical 280 (2008) 224–232
225
porate organic functional groups onto the surface of mesopores
[15].
In previous studies into the synthesis of MCM-41 meso-
2. Experimental
2.1. Chemicals and catalyst synthesis
porous silica, attempts to control the morphology of the
particles were made for MCM-41 type materials that
had been organically functionalized by the co-condensation
method. Mann et al. reported that, by changing the
organoalkoxysilane or its concentration, spherical, tube-like,
rod-like and filament-shaped particles could be obtained.
For instance, when 3-aminopropyltrimethoxysilane was used,
oblate ellipsoidal particles were obtained [16]. Furthermore,
Lin et al. carried out synthesis using lower concentra-
with 3-aminopropyltrimethoxysilane and N-(2-aminoethly)-
3-aminopropyltriethoxysilane, while spherical particles were
methoxysilane was used [17]. In addition, they found that these
spherical particles exhibited some selectivity in base catalysis
when secondary organic functional groups were incorporated
[18,19].
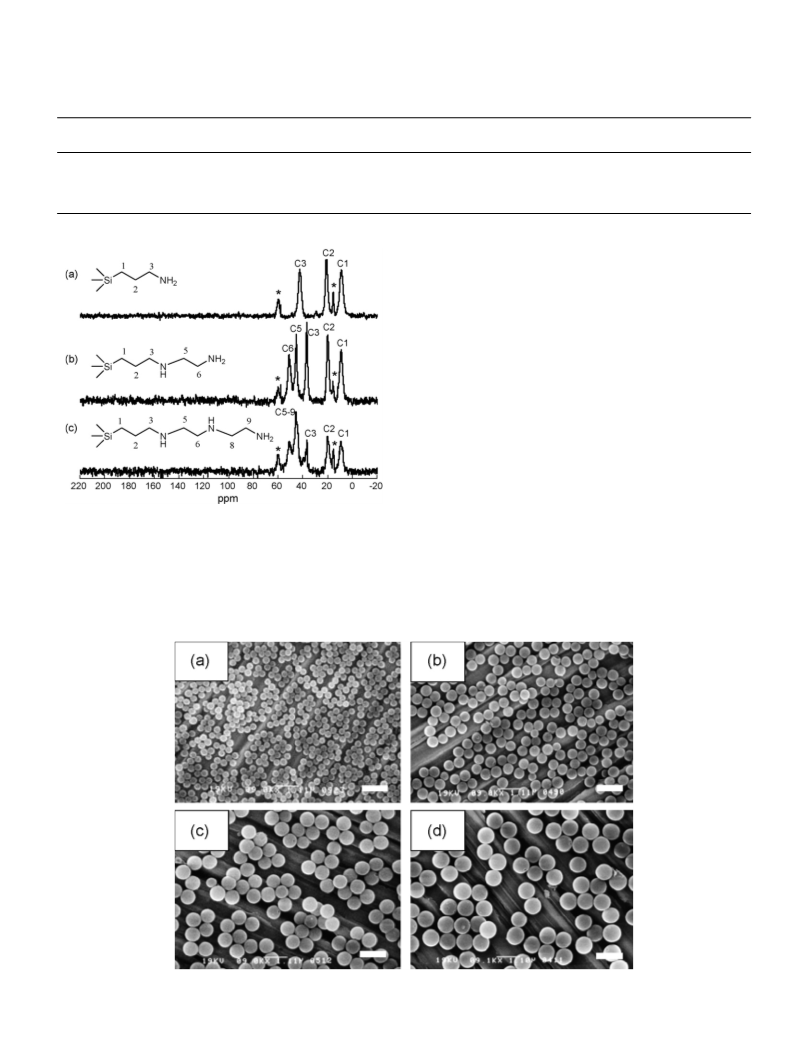
We synthesized amino-functionalized MMSS by using
hexadecyltrimethylammonium chloride (C16TMACl) as a
template for a co-condensation reaction between tetram-
ethoxysilane (TMOS) and organic trimethoxysilane (R-TMS).
Three different types of organic trimethoxysilanes were
used: 3-aminopropyltrimethoxysilane, (CH3O)3Si(CH2)3NH2
(AP-TMS), which contains just one amino group, [3-(2-
aminoethylamino)propyl]trimethoxysilane, (CH3O)3Si(CH2)3
NHCH2CH2NH2 (AEAP-TMS), which contains two amino
groups, and 3-[2-(2-aminoethylamino)ethylamino]propyltri-
methoxysilane, (CH3O)3Si(CH2)3NHCH2CH2NHCH2CH2NH2
(AEAEAPTMS) which contains three amino groups. 3-Amino-
propyltrimethoxysilane (AP-TMS), [3-(2-aminoethylamino)
propyl]trimethoxysilane (AEAP-TMS), and 3-[2-(2-amino-
ethylamino)ethylamino] propyltrimethoxysilane (AEAEAP-
TMS) were purchased from Aldrich. Hexadecyltrimethy-
lammonium chloride (C16TMACl) and tetramethoxysilane
(TMOS) were purchased from Tokyo Kasei. Monodispersed
silica spheres (500 nm in diameter) were purchased from
Nippon Shokubai Co., Ltd. All of the chemicals were used
as-received. Hereinafter in this paper, we abbreviate the
synthesized materials as MS-XY (where X is AP, AEAP or
AEAEAP, and Y is 5%, 10% or 20%). X denotes the type
of organic alkoxysilane that was used and Y denotes the
molar ratio of R-TMS in the total silane monomer used in
the co-condensation reaction. The co-condensation ratio (Y) is
expressed as R-TMS/(TMOS + R-TMS), which was calculated
by the added amount of silanes for the syntheses. In case of
MS-AP5% with different particle diameter, we abbreviate the
synthesized material as MS-AP5%-Z. Z denotes the particle
diameter (310–780 nm).
However, these spherical mesoporous silicas were found to
feature lower monodispersity and the array-direction of the
mesopores was irregular.
of hexagonally ordered and well-defined highly monodis-
persed mesoporous silica spheres (hereafter abbreviated as
MMSS)fromtetramethoxysilane(TMOS)andn-alkyltrimethyl-
ammonium halide (CnTMAX, X = Cl, Br) [20–22]. By changing
the surfactant that is used for the template and also the syn-
thetic conditions, the particle diameters and the pore sizes
of MMSS can be controlled flexibly. For fundamental stud-
ies of catalytic reactions, the diameters of the particles should
be monodispersed. We have been studied the effect of pore
size on base catalysis using amino-functionalized MMSS by
grafting method. It was found for the first time that the reac-
tion mostly proceeded inside the radially aligned mesopores
(the effective factor: 0.63) and that the optimum pore size
for amino-functionalized MMSS was affected by changing
the type and the number of the substituent groups on the
reactants. In addition, amino-functionalized MMSS was found
to be an excellent catalyst due to the radial alignment of
the mesopores compared to the other types of mesoporous
silica [23].
In this study, we attempted to synthesize functionalized
MMSS using a co-condensation method to incorporate various
amino groups. Compared with the grafting method, it can be
expected that the distribution of amino groups becomes homoge-
nously and the catalytic activity improves.
As a result, we have succeeded in obtaining amino-
functionalized monodispersed spherical particles that contain
radially aligned mesopores. We also confirmed that they show
high base catalytic activities. By controlling the conditions used
for the syntheses, MMSS particles of different sizes but with the
same diameter of pores were synthesized.
The synthesis reactions were carried out under basic condi-
tions, as follows.
For example, in the case of MS-AP5%, 3.52 g of C16TMACl
and 2.28 ml of 1 M sodium hydroxide solution were dis-
solved in 800 g of a methanol/water (50/50, w/w) solution
(methanol ratio: 0.5). A mixture of 1.25 g (8.24 mmol) of TMOS
and 0.08 g (0.43 mmol) of AP-TMS (AP-TMS/(TMOS + AP-
TMS) = 5 mol%) were then added to the solution with vigorous
stirring at 298 K. After the addition of the TMOS and the AP-
TMS, the clear solution suddenly turned opaque and resulted in
a white precipitate.
In the case of MS-AP5%-680 nm, the same amount of
TMOS/AP-TMS mixture was again added to the solution
0.5 h later. After 8 h of continuous stirring, the mixture
was aged overnight. The white powder was then filtered
and washed three times with distilled water, after which
it was dried at 318 K for 72 h. The powder that was
obtained was heated in 60 ml of ethanol solution contain-
ing 1 ml of concentrated hydrochloric acid (0.6 ml) at 333 K
for 3 h to remove the templates. Then, the powder was
filtered, washed several times with ethanol, and dried at
318 K.
It was found that the effectiveness factor improved
greatly compared with the particle prepared by grafting
method.

 Suzuki, Tomiko M.
Suzuki, Tomiko M.
 Nakamura, Tadashi
Nakamura, Tadashi
 Fukumoto, Keiko
Fukumoto, Keiko
 Yamamoto, Masami
Yamamoto, Masami
 Akimoto, Yusuke
Akimoto, Yusuke
 Yano, Kazuhisa
Yano, Kazuhisa