
Journal of Organic Chemistry p. 10306 - 10320 (2019)
Update date:2022-08-15
Topics:
 Bulandr, Jacob J.
Bulandr, Jacob J.
 Grabowski, Jacob P.
Grabowski, Jacob P.
 Law, Chunyin M.
Law, Chunyin M.
 Shaw, Jessica L.
Shaw, Jessica L.
 Goodell, John R.
Goodell, John R.
 Mitchell, T. Andrew
Mitchell, T. Andrew
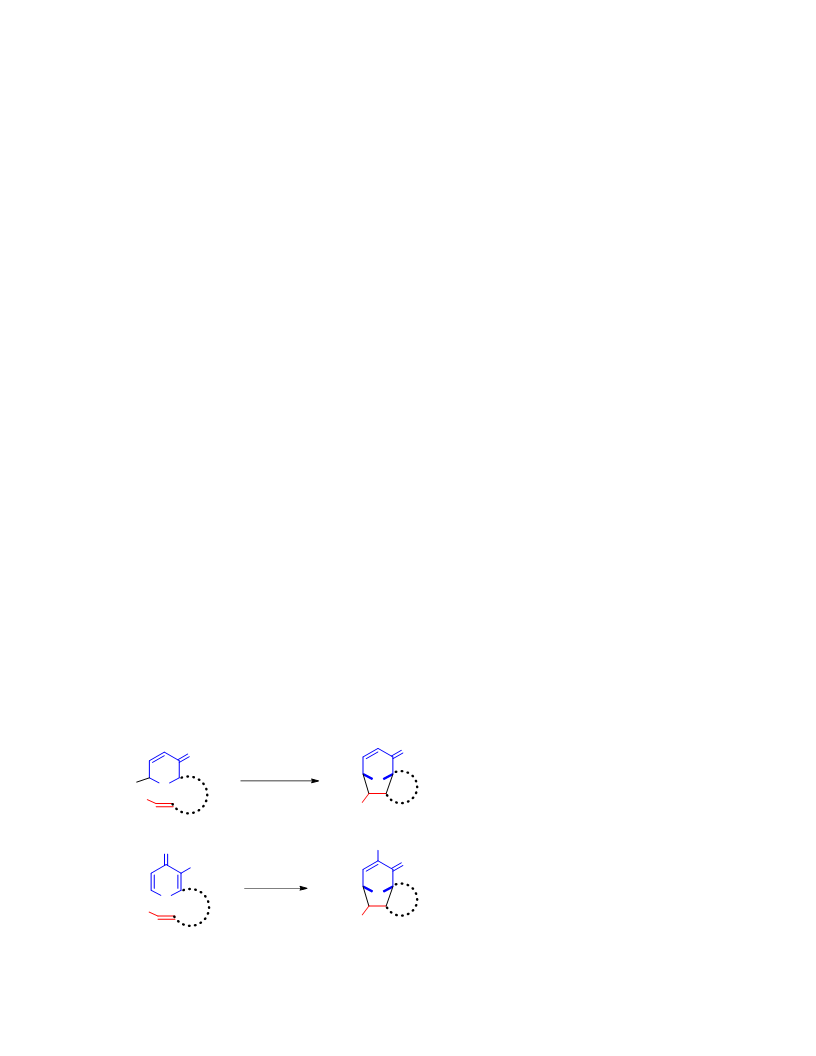
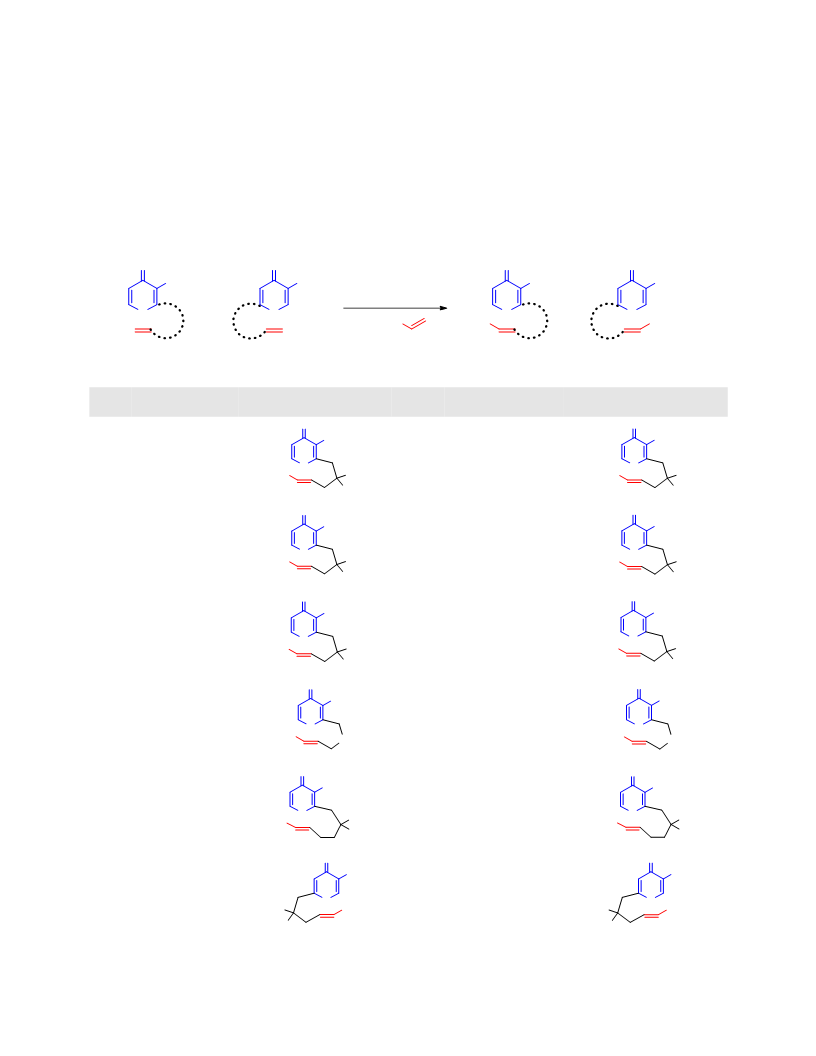
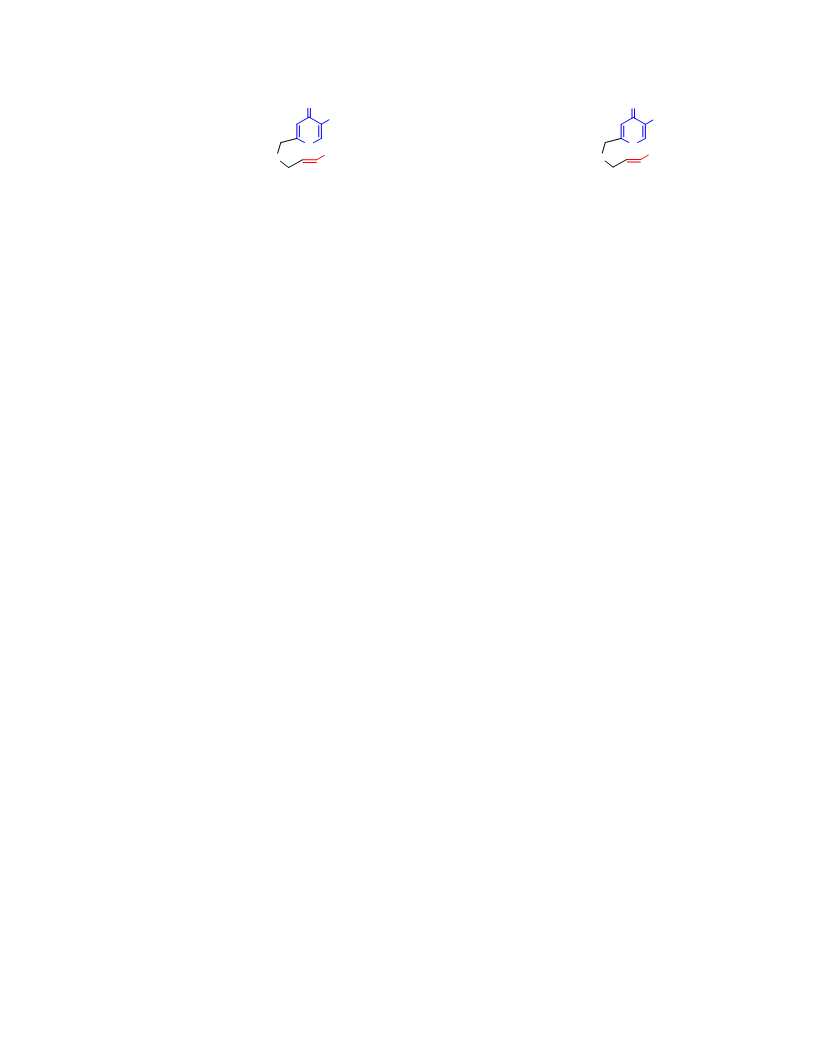
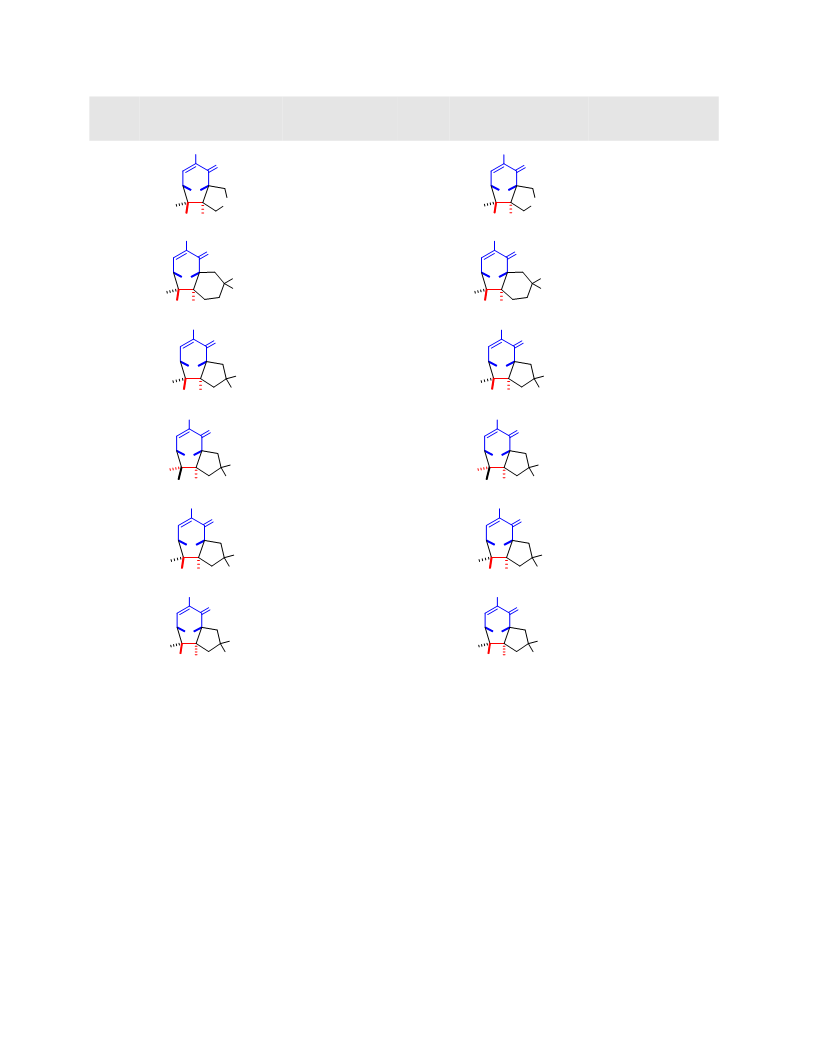
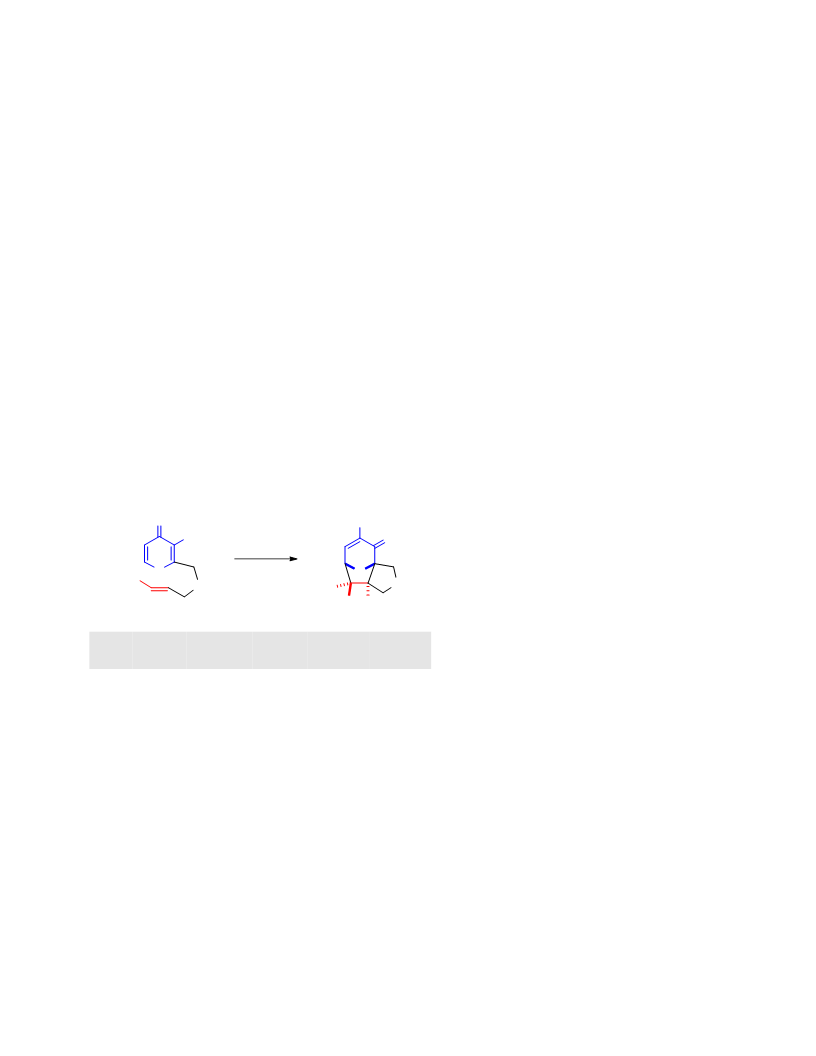
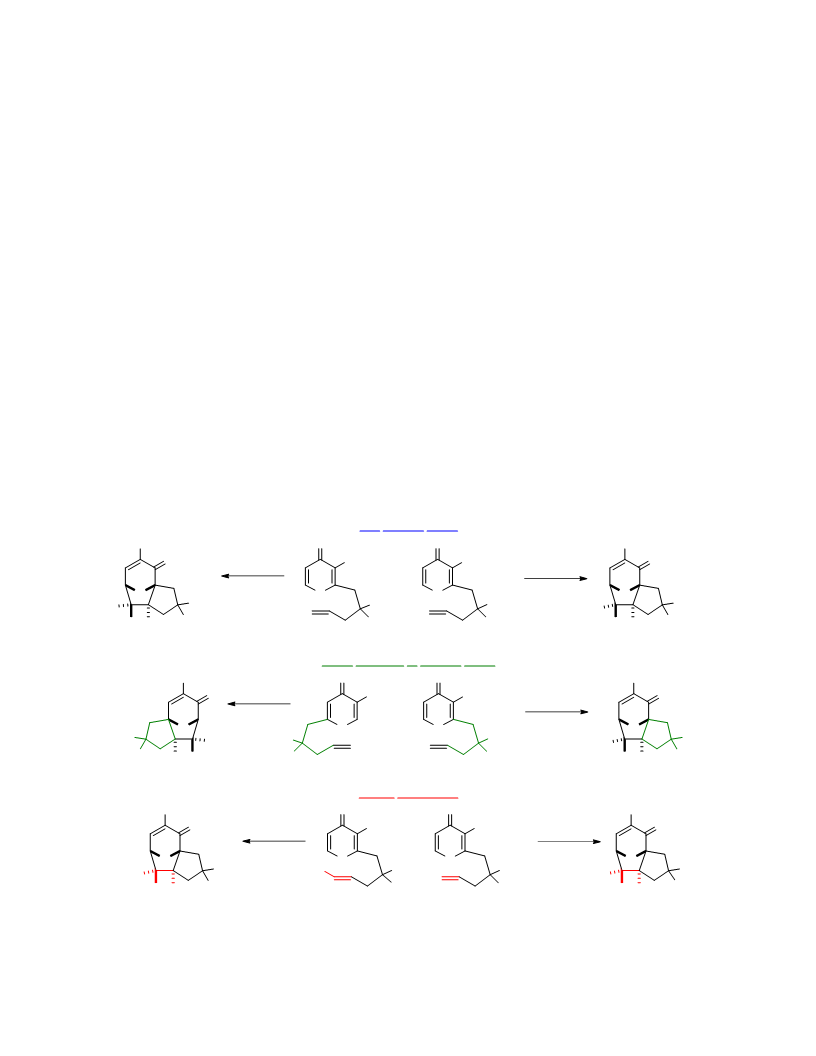
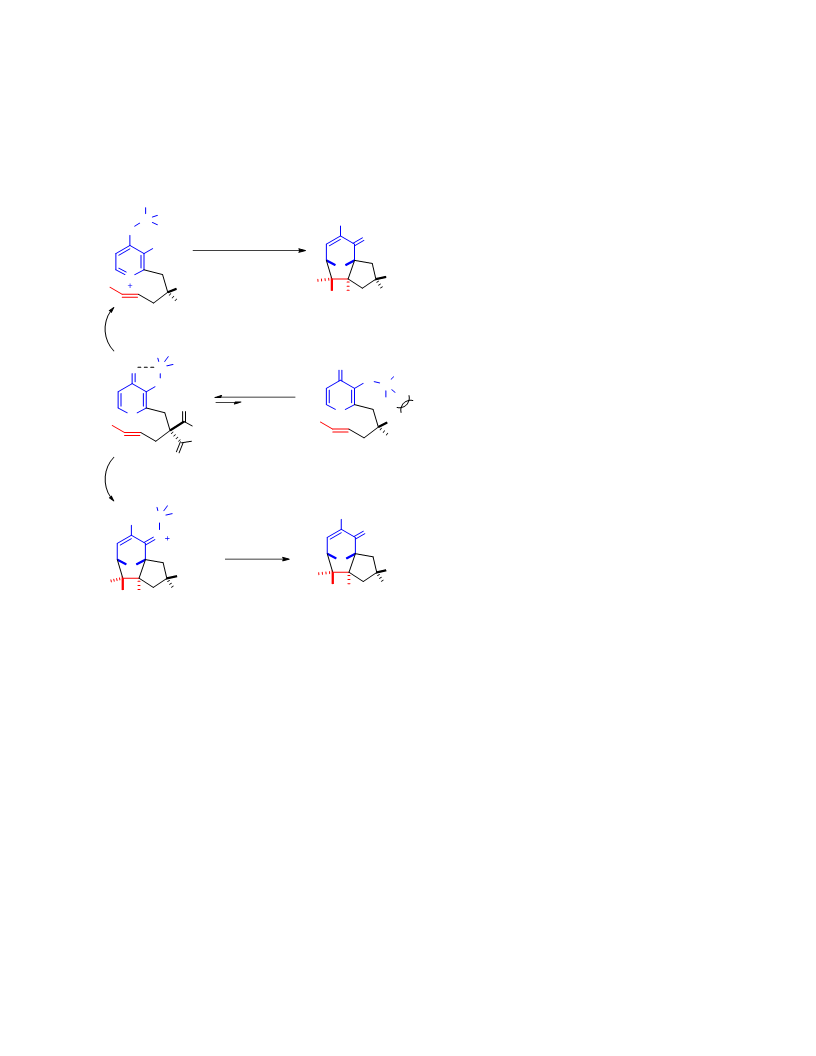
Systematic investigation of intramolecular silyloxypyrone-based [5 + 2] cycloadditions revealed three significant factors impacting conversion to cycloadduct: (1) the silyl transfer group has a substantial influence on the rate of reaction, and the robust t-butyldiphenylsilyl group was found to be more effective overall than the conventional t-butyldimethylsilyl group; (2) α,β-unsaturated esters were generally more reactive than terminal olefins and afforded appreciable quantity of cycloadduct even at room temperature; and (3) the proximity of the tether to the silyl transfer group revealed a critical alignment trend between the pyrone and the alkene. Taken together, these investigations provided insight regarding the steric and electronic parameters that impact the scope and limitation of these reactions.
View More



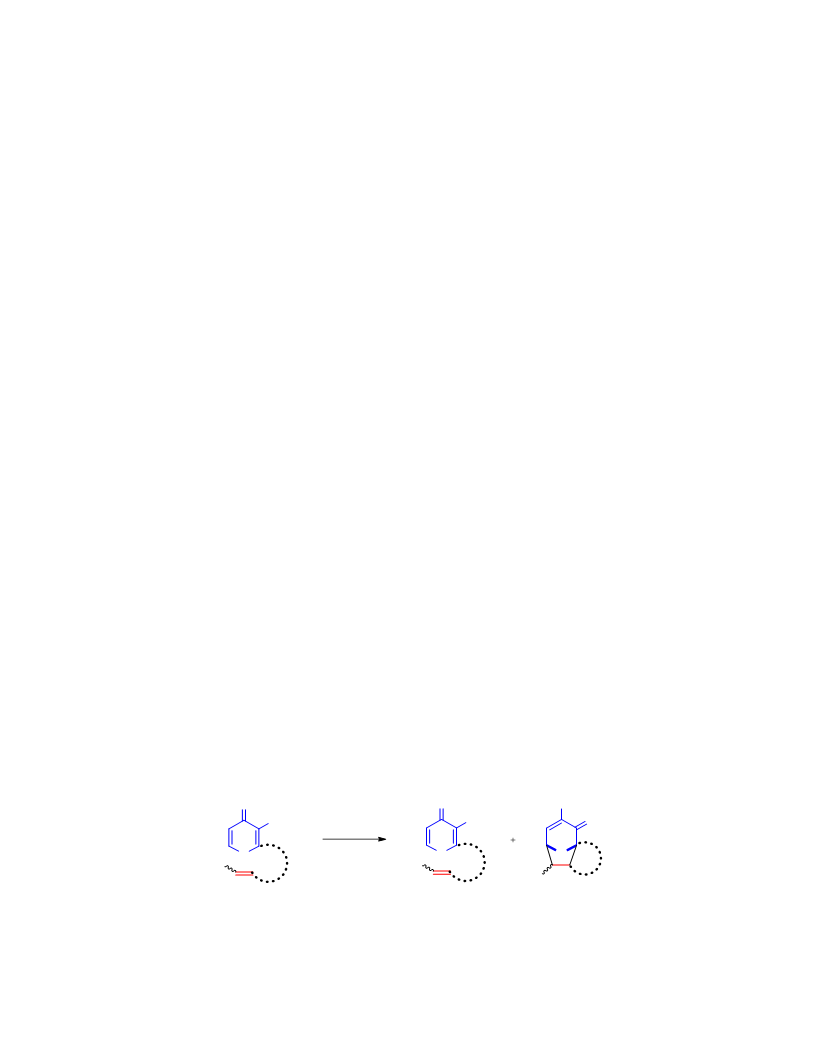


Zhejiang Allied Chemical Co.,Ltd
Contact:18967038207
Address:Area A-30, High-tech Industrial Park, Quzhou, Zhejiang, China.
Zhushan County Tianxin Pharmaceutical & Chemical Co., Ltd.
Contact:0086-719-4224892
Address:Tutang Road, Chengguan Town, Zhushan County, Hubei Province
Wuhan Yitongtai Science and Technology Co.,Ltd.
Contact:+86-27-88933550
Address:27th Fl. Bldg. 1, Shuian International Mansion, Heping Ave, Wuhan, Hubei, China
website:http://www.hybio.com.cn
Contact:+86 755 26588093
Address:No.9 Linkong West Street, Hengdian Street, Huangpi District, Wuhan City, China.
Quzhou Aokai Chemical Co., Ltd.
Contact:86-570-3032832
Address:NO.16 , Laodong Road,Quzhou City, Zhejiang Province,China
Doi:10.1039/b610088k
(2006)Doi:10.1016/j.dyepig.2016.09.013
(2017)Doi:10.1021/acs.inorgchem.9b01383
(2019)Doi:10.1248/cpb.34.1184
(1986)Doi:10.1248/cpb.59.692
(2011)Doi:10.1016/j.mcat.2021.111495
(2021)