Organic Letters
Letter
(f) Wang, Z.; Zhu, L.; Yin, F.; Su, Z.; Li, Z.; Li, C. J. Am. Chem. Soc.
2012, 134, 4258.
(5) Pan, F.; Lei, Z.-Q.; Wang, H.; Li, H.; Sun, J.; Shi, Z.-J. Angew.
Chem., Int. Ed. 2013, 52, 2063.
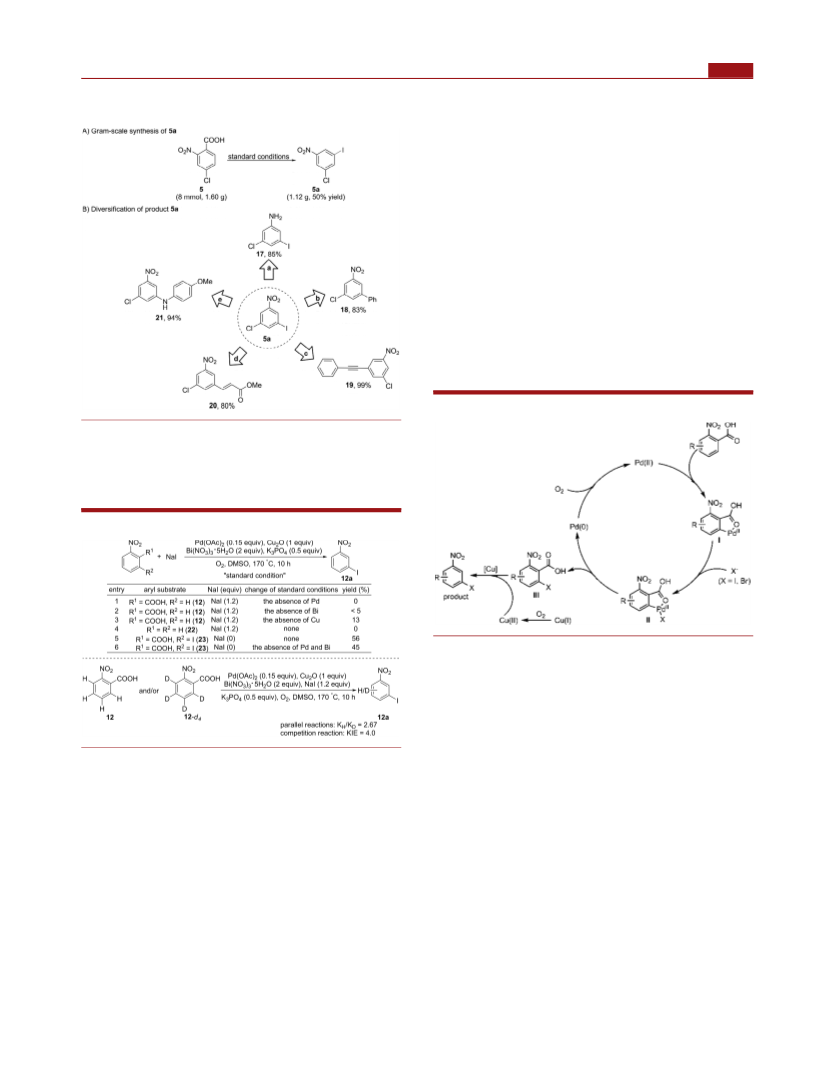
an intermediate in the transformation. Efforts are currently
underway to improve reaction efficiency, and the result will be
reported in due course.
(6) (a) Li, C.; Wang, J.; Barton, L. M.; Yu, S.; Tian, M.; Peters, D. S.;
Kumar, M.; Yu, A. W.; Johnson, K. A.; Chatterjee, A. K.; Yan, M.;
Baran, P. S. Science 2017, 356, 1045. (b) Edwards, J. T.; Merchant, R.
R.; McClymont, K. S.; Knouse, K. W.; Qin, T.; Malins, L. R.; Vokits,
B.; Shaw, S. A.; Bao, D.-H.; Wei, F.-L.; Zhou, T.; Eastgate, M. D.;
Baran, P. S. Nature 2017, 545, 213. (c) Huang, L.; Olivares, A. M.;
Weix, D. J. Angew. Chem., Int. Ed. 2017, 56, 11901. (d) Huihui, K. M.
M.; Caputo, J. A.; Melchor, Z.; Olivares, A. M.; Spiewak, A. M. J. Am.
Chem. Soc. 2016, 138, 5016. (e) Li, G.; Wang, T.; Fei, F.; Su, Y.-M.;
Li, Y.; Lan, Q.; Wang, X.-S. Angew. Chem., Int. Ed. 2016, 55, 3491.
(7) Qian, B.; Chen, S.; Wang, T.; Zhang, X.; Bao, H. J. Am. Chem.
Soc. 2017, 139, 13076.
ASSOCIATED CONTENT
* Supporting Information
The Supporting Information is available free of charge on the
■
S
Experimental procedures and spectral data for all
AUTHOR INFORMATION
■
(8) Huang, X.; Liu, W.; Hooker, J. M.; Groves, J. T. Angew. Chem.,
Int. Ed. 2015, 54, 5241.
Corresponding Author
ORCID
(9) (a) Pichette Drapeau, M.; Goossen, L. J. Chem. - Eur. J. 2016, 22,
18654. (b) Shi, G.; Zhang, Y. Adv. Synth. Catal. 2014, 356, 1419.
(c) Davies, D. L.; Macgregor, S. A.; McMullin, C. L. Chem. Rev. 2017,
117, 8649. (d) Giri, R.; Maugel, N.; Li, J.-J.; Wang, D.-H.; Breazzano,
S. P.; Saunders, L. B.; Yu, J. Q. J. Am. Chem. Soc. 2007, 129, 3510.
(10) (a) Cornella, J.; Righi, M.; Larrosa, I. Angew. Chem., Int. Ed.
2011, 50, 9429. (b) Luo, J.; Preciado, S.; Larrosa, I. Chem. Commun.
2015, 51, 3127.
Author Contributions
†Y.J. and S.W. contributed equally.
(11) Luo, J.; Preciado, S.; Larrosa, I. J. Am. Chem. Soc. 2014, 136,
4109.
Notes
(12) (a) Tang, J.; Hackenberger, D.; Goossen, L. J. Angew. Chem.,
Int. Ed. 2016, 55, 11296. (b) Kumar, N. Y. P.; Bechtoldt, A.;
Raghuvanshi, K.; Ackermann, L. Angew. Chem., Int. Ed. 2016, 55,
6929. (c) Zhang, J.; Shrestha, R.; Hartwig, J. F.; Zhao, P. Nat. Chem.
2016, 8, 1144. (d) Kim, K.; Vasu, D.; Im, H.; Hong, S. Angew. Chem.,
Int. Ed. 2016, 55, 8652. (e) Zou, J.-P.; Wu, D.-D.; Luo, J.; Xing, Q.-J.;
Luo, X.-B.; Dong, W.-H.; Luo, S.-L.; Du, H.-M.; Suib, S. L. ACS Catal.
2016, 6, 6861. (f) Maehara, A.; Tsurugi, H.; Satoh, T.; Miura, M. Org.
Lett. 2008, 10, 1159.
(13) (a) Zhang, Y.; Zhao, H.; Zhang, M.; Su, W. Angew. Chem., Int.
Ed. 2015, 54, 3817. (b) Qin, X.; Sun, D.; You, Q.; Cheng, Y.; Lan, J.;
You, J. Org. Lett. 2015, 17, 1762.
(14) Mandal, A.; Sahoo, H.; Dana, S.; Baidya, M. Org. Lett. 2017, 19,
4138.
(15) Shi, X.-Y.; Liu, K.-Y.; Fan, J.; Dong, X.-F.; Wei, J.-F.; Li, C.-J.
Chem. - Eur. J. 2015, 21, 1900.
(16) Bhadra, S.; Dzik, W. I.; Goossen, L. J. Angew. Chem., Int. Ed.
2013, 52, 2959.
(17) (a) Fu, Z.; Li, Z.; Xiong, Q.; Cai, H. Eur. J. Org. Chem. 2014,
2014, 7798. (b) Fu, Z.; Li, Z.; Xiong, Q.; Cai, H. RSC Adv. 2015, 5,
52101. (c) Li, Z.; Fu, Z.; Zhang, H.; Long, J.; Song, Y.; Cai, H. New J.
Chem. 2016, 40, 3014. (d) Fu, Z.; Li, Z.; Xiong, Q.; Cai, H. Youji
Huaxue 2015, 35, 984. (e) Fu, Z.; Li, Z.; Song, Y.; Yang, R.; Liu, Y.;
Cai, H. J. Org. Chem. 2016, 81, 2794. (f) Fu, Z.; Jiang, L.; Zuo, Q.; Li,
Z.; Liu, Y.; Wei, Z.; Cai, H. Org. Biomol. Chem. 2018, 16, 5416.
(g) Fu, Z.; Jiang, Y.; Jiang, L.; Li, Z.; Guo, S.; Cai, H. Tetrahedron Lett.
2018, 59, 4458.
The authors declare no competing financial interest.
ACKNOWLEDGMENTS
■
Financial support from the National Natural Science
Foundation of China (NSFC) (21761021 and 21571094),
Natural Science Foundation of Jiangxi Province
(20171BAB203002), and Sci & Tech Project of Education
Department of Jiangxi Province (60007) is gratefully acknowl-
edged.
REFERENCES
■
(1) (a) Davies, D. L.; Macgregor, S. A.; McMullin, C. L. Chem. Rev.
2017, 117, 8649. (b) Wei, Y.; Hu, P.; Zhang, M.; Su, W. Chem. Rev.
2017, 117, 8864.
(2) (a) Goossen, L. J.; Deng, G.; Levy, L. M. Science 2006, 313, 662.
(b) Goossen, L. J.; Rodríguez, N.; Melzer, B.; Linder, C.; Deng, G.;
Levy, L. M. J. Am. Chem. Soc. 2007, 129, 4824. (c) Goossen, L. J.;
Rodríguez, N.; Linder, C. J. Am. Chem. Soc. 2008, 130, 15248.
(d) Song, B.; Knauber, T.; Goossen, L. Angew. Chem., Int. Ed. 2013,
52, 2954. (e) Tang, J.; Biafora, A.; Goossen, L. J. Angew. Chem., Int.
Ed. 2015, 54, 13130. (f) Wang, C.; Piel, I.; Glorius, F. J. Am. Chem.
Soc. 2009, 131, 4194. (g) Zhang, F.; Greaney, M. F. Angew. Chem., Int.
Ed. 2010, 49, 2768. (h) Fang, P.; Li, M.; Ge, H. J. Am. Chem. Soc.
2010, 132, 11898. (i) Shang, R.; Ji, D.-S.; Chu, L.; Fu, Y.; Liu, L.
Angew. Chem., Int. Ed. 2011, 50, 4470. (j) Myers, A. G.; Tanaka, D.;
Mannion, M. R. J. Am. Chem. Soc. 2002, 124, 11250. (k) Pichette
Drapeau, M.; Bahri, J.; Lichte, D.; Goossen, L. J. Angew. Chem., Int.
Ed. 2019, 58, 892.
(18) Lyons, T. W.; Sanford, M. S. Chem. Rev. 2010, 110, 1147.
(19) Baur, A.; Bustin, K. A.; Aguilera, E.; Petersen, J. L.; Hoover, J.
M. Org. Chem. Front. 2017, 4, 519.
(20) For selected examples of Cu(I)-mediated decarboxylation
reactions, see: (a) Goossen, L. J.; Rudolphi, F.; Oppel, C.; Rodríguez,
N. Angew. Chem., Int. Ed. 2008, 47, 3043. (b) Goossen, L. J.;
Rodríguez, N.; Lange, P. P.; Linder, C. Angew. Chem., Int. Ed. 2010,
49, 1111. (c) Shang, R.; Fu, Y.; Wang, Y.; Xu, Q.; Yu, H.-Z.; Liu, L.
Angew. Chem., Int. Ed. 2009, 48, 9350. (d) Hu, G.; Gao, Y.; Zhao, Y.
Org. Lett. 2014, 16, 4464. (e) Hossian, A.; Manna, K.; Das, P.; Jana, R.
ChemistrySelect 2018, 3, 4315.
(3) (a) Moon, P. J.; Fahandej-Sadi, A.; Qian, W.; Lundgren, R. J.
Angew. Chem., Int. Ed. 2018, 57, 4612. (b) Liu, Z.-J.; Lu, X.; Wang, G.;
Li, L.; Jiang, W.-T.; Wang, Y.-D.; Xiao, B.; Fu, Y. J. Am. Chem. Soc.
2016, 138, 9714. (c) Zhang, L.; Hang, Z.; Liu, Z.-Q. Angew. Chem.,
Int. Ed. 2016, 55, 236.
(4) (a) Hu, F.; Shao, X.; Zhu, D.; Lu, L.; Shen, Q. Angew. Chem., Int.
Ed. 2014, 53, 6105. (b) Liu, C.; Wang, X.; Li, Z.; Cui, L.; Li, C. J. Am.
Chem. Soc. 2015, 137, 9820. (c) Kan, J.; Huang, S.; Lin, J.; Zhang, M.;
Su, W. Angew. Chem., Int. Ed. 2015, 54, 2199. (d) Liu, X.; Wang, Z.;
Cheng, X.; Li, C. J. Am. Chem. Soc. 2012, 134, 14330. (e) Bhadra, S.;
Dzik, W. I.; Goossen, L. J. J. Am. Chem. Soc. 2012, 134, 9938.
(21) For selected examples of Cu(II)-mediated decarboxylation
reactions, see: (a) Goossen, L. J.; Zimmermann, B.; Knauber, T.
Beilstein J. Org. Chem. 2010, 6, 43. (b) Kumar, N.; Ansari, M. Y.; Kant,
D
Org. Lett. XXXX, XXX, XXX−XXX

 Fu, Zhengjiang
Fu, Zhengjiang
 Jiang, Yongqing
Jiang, Yongqing
 Wang, Shuiliang
Wang, Shuiliang
 Song, Yuanyuan
Song, Yuanyuan
 Guo, Shengmei
Guo, Shengmei
 Cai, Hu
Cai, Hu