10.1002/anie.201908496
Angewandte Chemie International Edition
COMMUNICATION
O
O
Pearson, M. N. Paddon-Row, M. S. Sherburn, J. Chem. Soc., Chem.
Commun. 2012, 48, 6639-6641.
H
NPh
NPh
11
O
[4]
For a review on dendralenes, see: H. Hopf, M. S. Sherburn, Angew.
Chem., Int. Ed. 2012, 51, 2298-2338.
O
H
Me
H
Me
H
[5]
(a) First synthesis: L. Skattebøl, J. L. Charlton, P. deMayo, Tetrahedron
Lett. 1966, 7, 2257–2260; (b) practical synthesis and half-life
measurement: E. J. Lindeboom, A. C. Willis, M. N. Paddon-Row, M. S.
Sherburn, Angew. Chem. Int. Ed. 2014, 53, 5440-5443.
C. G. Newton, S. L. Drew, A. L. Lawrence, A. C. Willis, M. N. Paddon-
Row, M. S. Sherburn, Nat. Chem. 2015, 7, 82-86.
neat
40 ℃
H
H
O
Me
Me
H
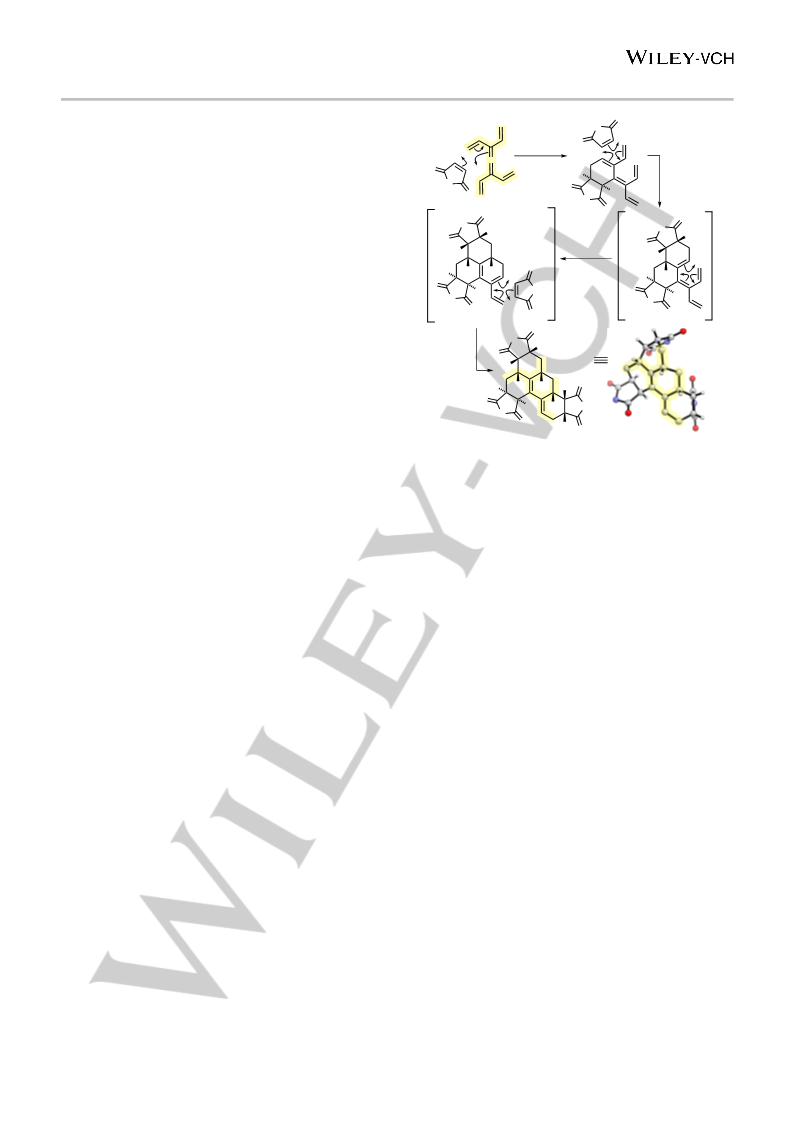
twofold
Diels-Alder
NPh
Me
Me
O
O
X-Ray
32
68%
31
[6]
Ph
N
O
H
O
Ph
[7]
[8]
C. G. Newton, M. S. Sherburn Nat. Prod. Rep. 2015, 32, 865-876.
C. G. Newton, M. S. Sherburn in Cross Conjugation: Modern Dendralene,
Radialene and Fulvene Chemistry, H. Hopf, M. S. Sherburn, eds., Wiley-
VCH 2016, pp 413-443.
N
H
O
d6-benzene
75 ℃
H
H
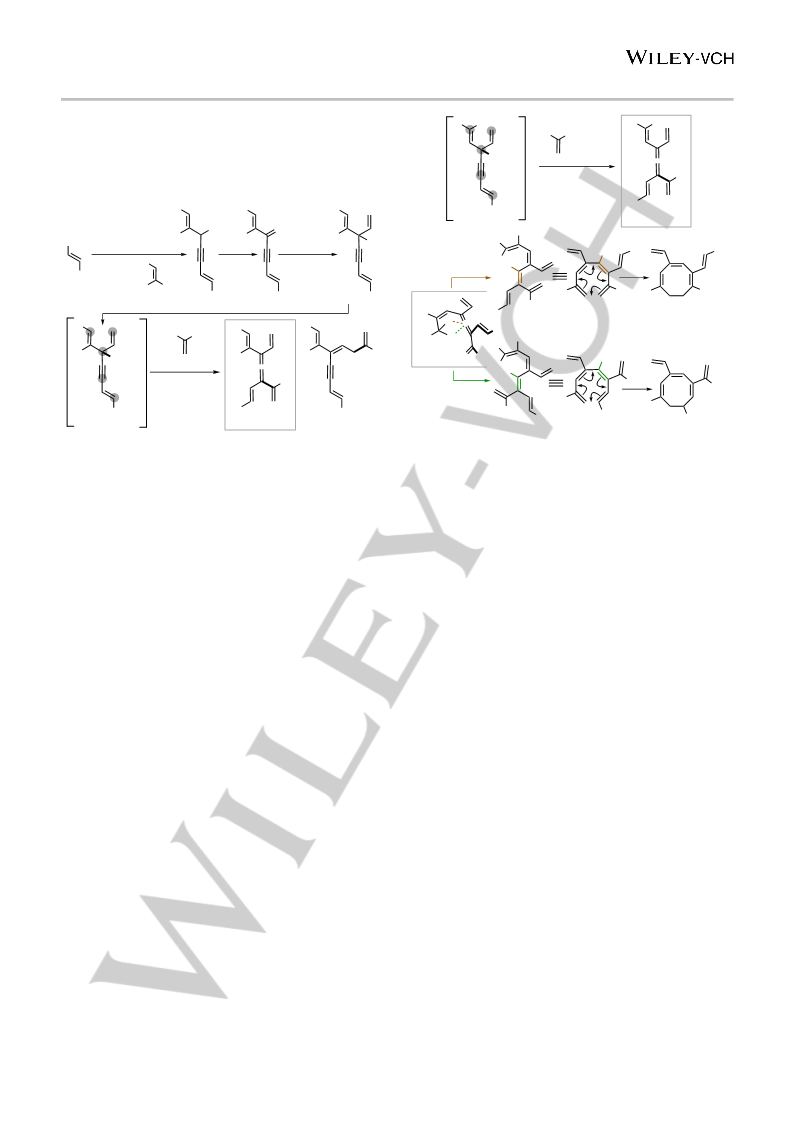
11
Diels-Alder Me
Me
Me
6π electro-
cyclization
Me
H
[9]
(a) M. Purpura, N. Krause, Eur. J. Org. Chem. 1999, 267-275 and
references cited therein. First report: (a) J. Gore, J. P. Dulcere, Chem.
Commun. 1972, 866-867.
64%
Me
Me
34
33
[10] (a) M. Ucuncu, E. Karakus, M. Kus, G. E. Akpinar, O. Aksin-Artok, N.
Krause, S. Karaca, N. Elmaci, L. Artok, J. Org. Chem. 2011, 76, 5959-
5971; (b) F. Ziyanak, M. Kus, L. Alkan-Karadeniz, L. Artok, Tetrahedron
2018, 74, 3652-3662. For an example with an alkynyl nucleophile, see
(c) D. Taç, L. Artok, Tetrahedron Lett. 2018, 59, 895-898.
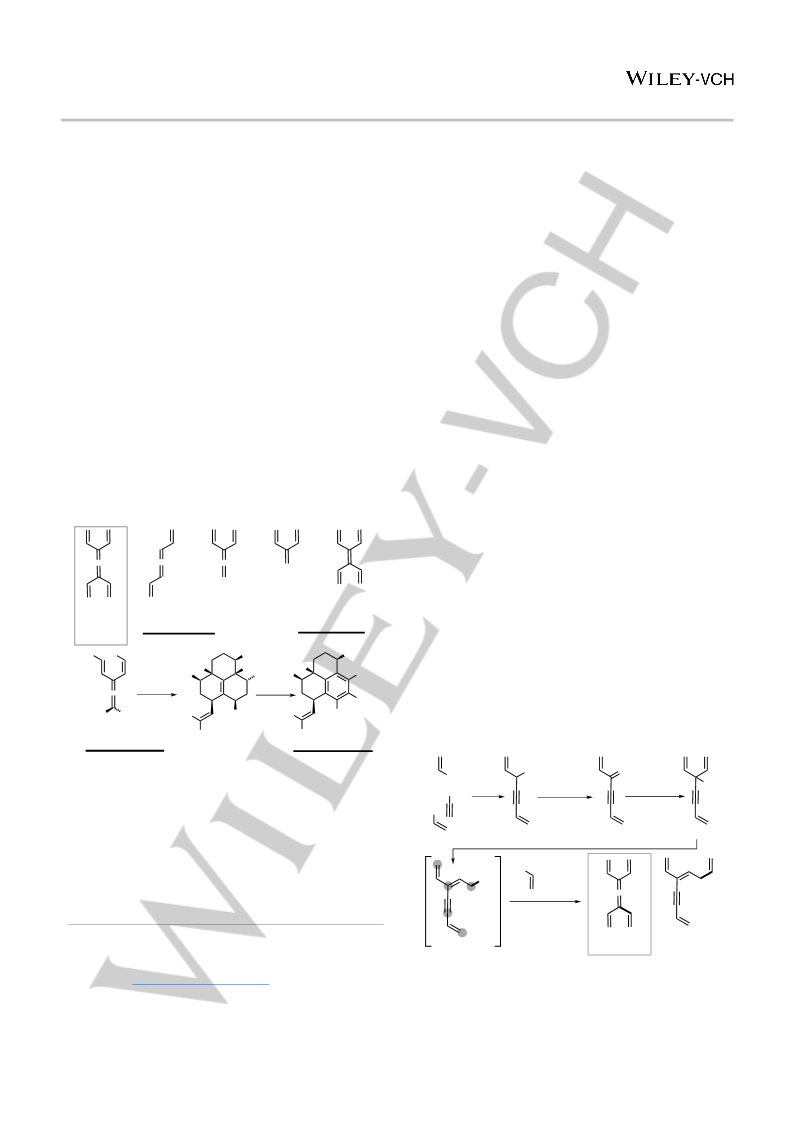
Scheme 5. Divergent, rapid structural complexity generation with cyclo-
octatriene 31. Only stereocenter hydrogens are shown in the X-ray crystal
structure and phenyl groups are omitted for clarity.
[11] I. N. Nazarov and L. B. Fisher, Zhurnal Obshchei Khimii, 1950, 20, 1107-
1113.
In summary, the first syntheses of the parent tetravinylallene
[12] For (vinyl)allene synthesis through Pd(0)-catalyzed C–C coupling
involving 1,3-propargyl to allenyl transposition, see: (a) K. Ruitenberg, H.
Kleijn, C. J. Elsevier, J. Meijer, P. Vermeer, Tetrahedron Lett. 1981, 22,
1451-1452; (b) K. Ruitenberg, H. Kleijn, H. Westmijze, J. Meijer, P.
Vermeer, Recl. Trav. Chim. Pays-Bays 1982, 101, 405-408; (c) E. Keinan,
E. Bosch, J. Org. Chem. 1986, 51, 4006-4016; (d) T. Mandai, M. Ogawa,
H. Yamoaki, T. Nakata, H. Murayama, M. Kawada, J. Tjusi, Tetrahedron
Lett. 1991, 32, 3397-3398; (e) S. Ma, A. Zhang, J. Org. Chem. 2002, 67,
2287-2294; (f) S. Ma, G. Wang, Angew. Chem. Int. Ed. 2003, 42, 4215 –
4217; Angew. Chem. 2003, 115, 4347-4349; (g) G. A. Molander, E. M.
Sommers, S. R. Baker, J. Org. Chem. 2006, 71, 1563-1568; (h) J. A.
Souto, M. Pérez, C. S. López, R. Álvarez, A. Torrado, A. R. de Lera, J.
Org. Chem. 2010, 75, 4453-4462; for Cu(I)-catalyzed reactions, see (i)
M. Yang, N. Yokokawa, H. Ohmiya, M. Sawamura, Org. Lett. 2012, 14,
816-819.
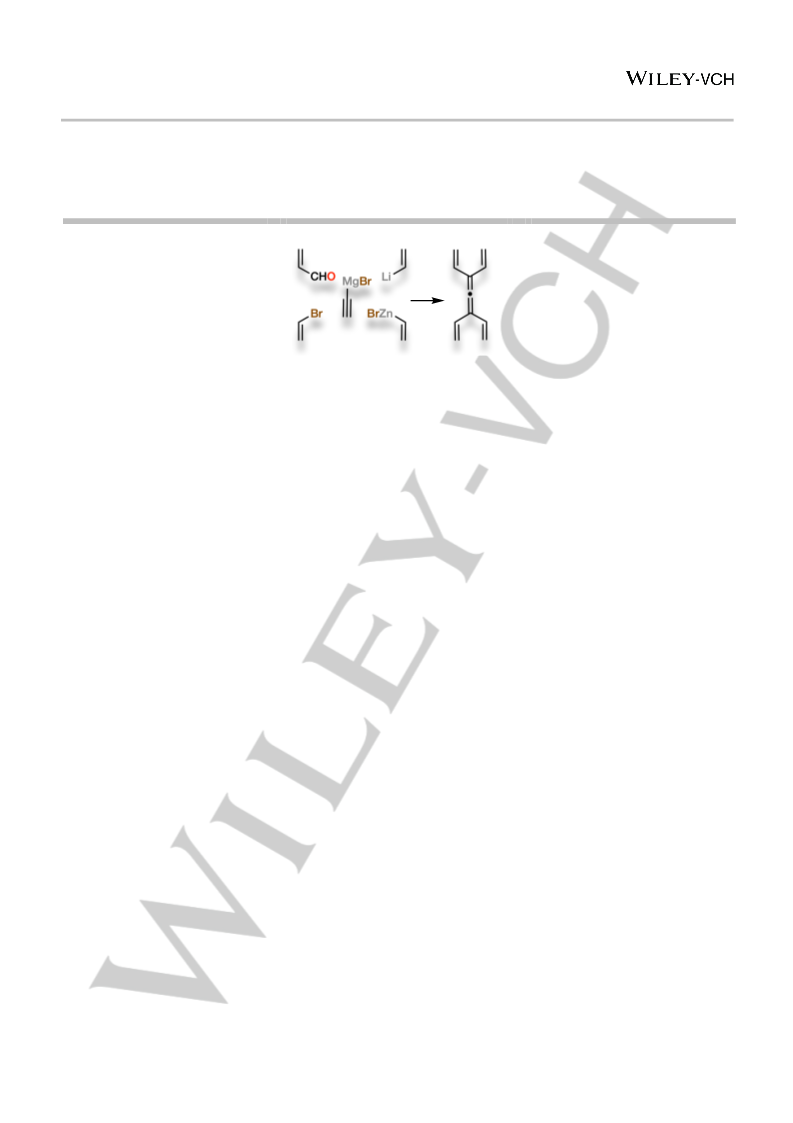
1
and substituted analogs were achieved through the
development of Negishi-type Pd(0)-catalyzed cross-coupling
processes that proceed with either 1,3-transposition or 1,5-
transposition. In principle, these couplings could generate up to
five distinct regioisomeric products. The desired allenic
regioisomer is the major isolated product in every case. In solution,
the parent, unsubstituted TVA 1 is more stable than 1,1-
divinylallene 3 but less stable than both [3]dendralene 4 and
tetravinylethylene 5. Surprisingly, the parent unsubstituted TVA is
more stable than a substituted analog, with a Z-methyl substituent
triggering decomposition by way of a pericyclic reaction cascade,
initiated by a sigmatropic [1,5]-H shift. It would appear that Z-
substituents carrying an allylic hydrogen should be avoided if
more stable TVAs are required. Finally, this work shows that TVAs
have an unequaled ability to build diverse multi-ring systems, in
the process forging many C–C bonds and creating numerous
stereocenters. In this respect, TVAs outshine their [3]dendralene
and 1,1-divinylallene relatives: compounds with a proven track
record for delivering the most step economic total syntheses.[6,22]
[13] This experimental half-life was essentially the same in both C6D6 and
CDCl3 at the same starting concentration. Increased starting
concentrations led to faster decomposition rates and decreased starting
concentrations led to slower decomposition rates.
[14] N. Radić, Z. B. Maksić, J. Org. Chem. 2019, 84, 2425−2438.
[15] C=C-based systems with both through- and cross-conjugation are poorly
represented in the literature: see Cross Conjugation: Modern Dendralene,
Radialene and Fulvene Chemistry, H. Hopf, M. S. Sherburn, eds., Wiley-
VCH 2016. 464 pages. ISBN 978-3-527-33437-7.
[16] For recent examples of torquoselective thermal 6π electrocyclizations
through remote stereocontrol, see: (a) S. Dongbang, B. Pedersen, J. A.
Ellman, Chem. Sci. 2019, 10, 535-541; (b) E. M. Phillips, T. Mesganaw,
S. Duttwyler, B. Q. Mercado, J. A. Ellman, A. Patel, K. N. Houk, Angew.
Chem. Int. Ed. 2015, 54, 12044-12048; (c) Z.-X. Ma, A. Patel, K. N. Houk,
R. P. Hsung, Org. Lett. 2015, 17, 2138–2141; (d) A. Patel, G. A. Barcan,
O. Kwon, K. N. Houk, J. Am. Chem. Soc. 2013, 135, 4878–4883; (e) G.
A. Barcan, A. Patel, K. N. Houk, O. Kwon, Org. Lett. 2012, 14, 5388-5391.
[17] Twofold Diels-Alder sequences of [3]dendralenes are diene-transmissive,
a term introduced by O. Tsuge, E. Wada, S. Kanemasa, Chem. Lett.
1983, 239-242.
Acknowledgements
[**] This work was supported by the Australian Research Council
(DP160104322). We thank Dr. Hideki Onagi and Mr. Chris Blake
(Australian National University) for assistance with HPLC separations and
NMR experiments, respectively.
Keywords: hydrocarbons • allenes • polyenes • cross-coupling •
domino reactions
[18] (a) E. W. Colvin, B. J. Hamill, J. Chem. Soc., Chem. Commun. 1973,
151-152; (b) E. W. Colvin, B. J. Hamill, J. Chem. Soc., Perkin Trans. 1
1977, 869-874; (c) E. C. Hansen, D. Lee, Org. Lett. 2004, 6, 2035-2038.
[19] Proposed intermediates 18 and 19 were not observed during the
monitoring of this transformation by 1H NMR spectroscopy.
[1]
[2]
U. Mödlhammer, H. Hopf, Angew. Chem. Int. Ed. 1975, 14, 501-502.
K. M. Cergol, C. G. Newton, A. L. Lawrence, A. C. Willis, M. N. Paddon-
Row, M. S. Sherburn, Angew. Chem. Int. Ed. 2011, 50, 10425-10428.
(a) First synthesis: A. T. Blomquist, J. A. Verdol, J. Am. Chem. Soc. 1955,
77, 81–83; (b) practical synthesis and half-life measurement in neat form:
T. A. Bradford, A. D. Payne, A. C. Willis, M. N. Paddon-Row, M. S.
Sherburn J. Org. Chem. 2010, 75, 491-494; M. F. Saglam, T. Fallon, M.
N. Paddon-Row, M. S. Sherburn, J. Am. Chem. Soc. 2016, 138, 1022-
1032; (c) half-life measurement in solution: H. Toombs-Ruane, E. L.
[3]
[20] For related 1,5-H shifts, see (a) K. J. Crowley, J. Proc. Chem. Soc.,
London 1964, 17; (b) K. L. Mikolajczak, M. O. Bagdy, R. B. Bates. I. A.
Wolff, J. Org. Chem. 1965, 30, 2983-2988; (c) L. Skattebøl, Tetrahedron
1969, 25, 4933-4938; (d) D. E. Minter, G. J. Fonken, F. T. Cook,
Tetrahedron Lett. 1979, 8, 711-714; (e) C. G. Knudsen, R. A. S.
Chandraratna, L. P. Walkeapää, Y. S. Chauhan, S. C. Carey, T. M.
This article is protected by copyright. All rights reserved.

 Elgindy, Cecile
Elgindy, Cecile
 Ward, Jas S.
Ward, Jas S.
 Sherburn, Michael S.
Sherburn, Michael S.