8
54
H. Kunkely - A Vogler • Photooxidation of Dicyanoaurate(I)
of the irradiation the spectral changes are obscured the cyanide 7r* orbital is pushed below the empty
by a secondary photolysis. The primary photoprod- 6s orbital of gold and becomes the LUMO [13]
uct [Au(CN)2Cl2l~ undergoes a photosubstitution. of the complex. Accordingly, MLCT excitation of
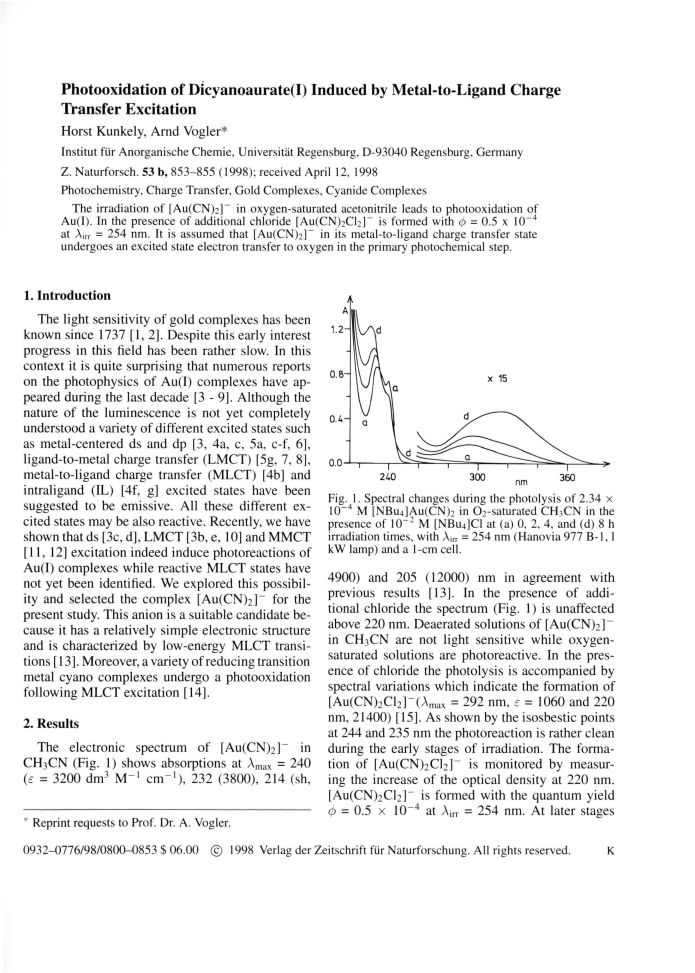
After prolonged irradiation (Fig. 1) the absorption
[Au(CN)2]~ induces the photooxidation of Au(I)
spectrum approaches that of [AuCl4]_ (Amax = 323 to Au(III). Although the overall photoreaction is
nm, e = 5800; 227 nm, 44800) [15].
a two-electron oxidation of Au(I) it may initially
proceed by a one-electron oxidation generating an
Au(II) intermediate which either disproportionates
to Au(I) and Au(III) or is oxidized to Au(III) in a
second electron transfer step. In this context it is of
interest that the existence of Au2+ ions in different
systems even under ambient conditions has been
confirmed quite recently [18, 19].
The photoproduct [Au(CN)2Cl2]- undergoes a
further photolysis. The irradiation leads to a photo-
substitution which finally yields [AuCU]- . The na-
ture of the reactive excited state of [Au(CN)2Cl2]~
is not clear since low-energy LF as well as LMCT
states exist in close proximity [15]. In analogy to
other d8 metals such as Pt(II) [20], LF states are
certainly reasonable candidates for inducing substi-
tutions.
3. Discussion
The electronic spectrum of [Au(CN)2]- is char-
acterized by a set of MLCT absorptions [13]. At
room temperature solutions of [Au(CN)2]- do not
show any emission, and are not photoreactive in
the absence of a suitable electron acceptor. How-
ever, in the presence of oxygen a photooxidation
takes place which in the presence of chloride yields
[
Au(CN)2Cl2]~ as a well defined product [16].
In analogy to [AuC^]- and [AuBr2]~[3d] it is
suggested that the lowest-energy excited state of
[
Au(CN)2]~ undergoes electron transfer to oxy-
gen in the primary photochemical step. The low
quantum yield of photooxidation is certainly re-
lated to the small concentration of oxygen in so-
lution. The lowest-energy and reactive excited state
of [AuCl2]~ and [AuB^]- is of the metal-centered
ds type [3d, 17]. In contrast to these halide com-
plexes the lowest excited state of [Au(CN)2]~ is of
the MLCT type because by overlap with the gold 6p
Acknowledgment
Support of this research by the Deutsche Forschungs-
gemeinschaft and the Fonds der Chemischen Industrie is
gratefully acknowledged.
[
1] H. Hellot, Histoire de 1‘academie royale des sciences,
p. 101 (1737).
c) V. W.-W. Yam, J. Chem. Soc. Dalton Trans. 1990,
3747;
[
2] V. Balzani, V. Carassiti, Photochemistry of Coordi-
nation Compounds, p. 273, Academic Press, London
1970).
3] a) A. Vogler, C. Quett, H. Kunkely, Ber. Bunsenges.
d) D. Li, C.-M. Che, S.-M. Peng, S.-T. Liu, Z.-Y.
Zhou, T. C. W. Mak, J. Chem. Soc. Dalton. Trans.
1993, 189;
e) S.-J. Shieh, D. Li, S.-M. Peng, C.-M. Che, J.
Chem. Soc. Dalton Trans. 1993, 195;
(
[
Phys. Chem. 92, 1486(1988);
b) H. Kunkely, A. Vogler, J. Photochem. Photobiol.,
A: Chem. 105,7(1997);
f) B.-C. D. Li, S.-M. Peng, C.-M. Che, J. Chem.
Soc. Dalton Trans. 1993, 2365;
c) H. Kunkely, A. Vogler, J. Organomet. Chem. 541,
g) D. Li, X. Hong, C.-M. Che, W.-C. Lo, S.-M.
Peng, J. Chem. Soc. Dalton Trans. 1993, 2929;
h) C.-M. Che, H.-K. Yip, W.-C. Lo, S.-M. Peng,
Polyhedron 13, 887 (1994);
i) X. Hong, K.-K. Cheung, C.-X. Guo, C.-M. Che,
J. Chem. Soc. Dalton Trans. 1994. 1867;
j) S.-J. Shieh, X. Hong, S.-M. Peng, C.-M. Che, J.
Chem. Soc. Dalton Trans. 1994. 3067;
k) C.-K. Chan, C.-X. Guo, K.-K. Cheung, D. Li, C -
M. Che, J. Chem. Soc. Dalton Trans. 1994. 3677.
1
77 (1997);
d) H. Kunkely, A. Vogler, Inorg. Chem. 31, 4539
1992);
e) H. Kunkely, A. Vogler, Z. Naturforsch. 51b. 1067
1996).
(
(
[
4] a) C.-M. Che, H.-L. Kwong, V. W.-W. Yam, K.-C.
Cho, J. Chem. Soc. Chem. Commun. 1984. 885;
b) C.-M. Che, W.-T. Wong, T.-F. Lai. H.-L. Kwong,
J. Chem. Soc. Commun. 1989. 243;
Brought to you by | New York University Bobst Library Technical Services
Authenticated
Download Date | 7/21/15 12:48 PM

 Kunkely, Horst
Kunkely, Horst
 Vogler, Arnd
Vogler, Arnd