◦
appeared, and on further cooling to −60 C these resolved into
NMR spectra were recorded from CD
2
Cl
2
solutions on a Bruker
1
19/117
two triplets and a singlet each with
Sn satellites [d = −138.5 (t,
DPX400, and referenced to residual H resonances of the solvent
or CFCl respectively. Microanalytical measurements were per-
2
119
19
117
19
J
FF = 55 Hz, Sn– F = 2703, Sn– F = 2583 Hz); d = −174.3
3
1
19
19
117
19
119
(
t, Sn– F = 2263, Sn– F = 2162 Hz); d = −145.8 (s, Sn–
formed by the microanalytical service at Strathclyde University.
1
9
117
19
F = 2547, Sn– F = 2437 Hz)] consistent with [SnF
4
(Me S) ],
2
2
with an approximate cis : trans ratio of 6 : 1. Evaporation of this
solution under reduced pressure resulted in precipitation of a
white, largely ligand-free, solid, and the complex has not been
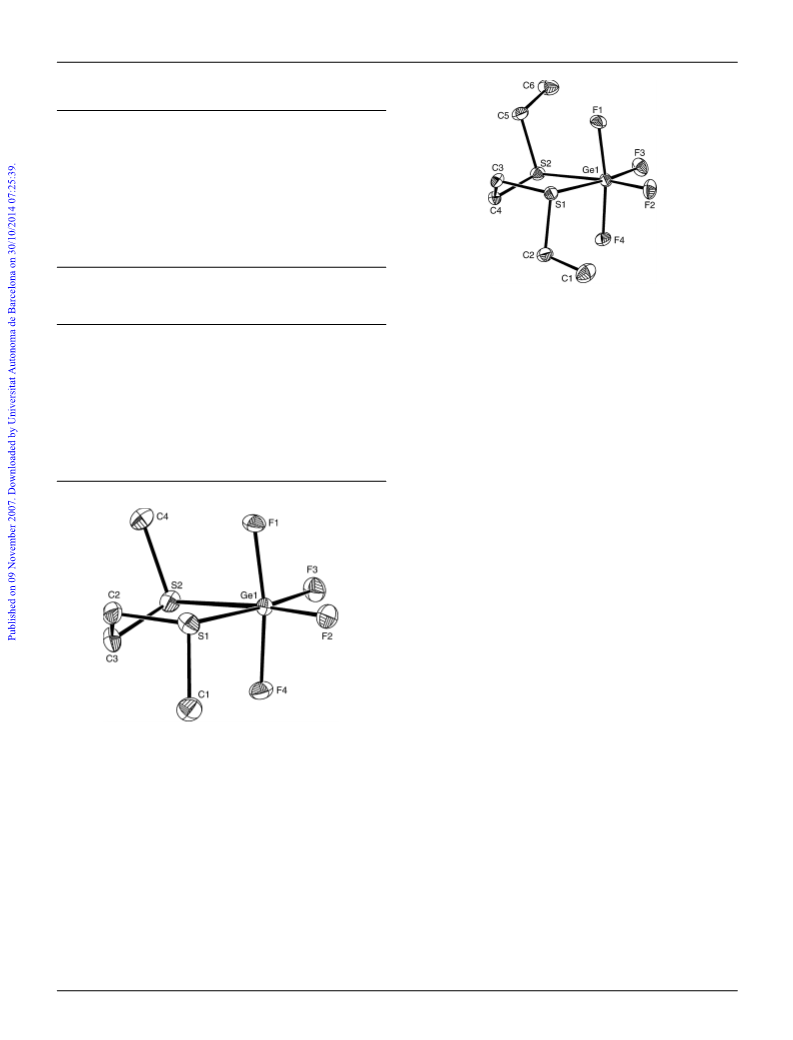
Preparations
[
GeF
4
{EtS(CH
2
)
2
SEt}]
SEt (0.018 g, 0.12 mmol) was added to
isolated. This contrasts with [SnX
4
(Me
2
S)
2
] (X = Cl or Br) which
The ligand EtS(CH
)
2 2
are readily isolated and are fully characterised both structurally
a solution of [GeF (CH
4
3
CN) ] (0.023 g, 0.10 mmol) in CH Cl
2
2
2
15,16
and spectroscopically.
(
15 mL) at room temperature. After stirring for 10 min, the
The germanium fluoride complexes described here are the only
thioether examples, so no further comparisons are possible, but a
solution was concentrated slowly by evaporation which gave
colourless crystalline solid which was isolated by filtration. Yield:
1
5,16
range of thioether adducts of SnCl
only isolated SnI example is the purple insoluble [SnI
although [SnI
{MeS(CH
4
and SnBr
4
are known
(the
}],
>
60%. C
C 22.4, H 4.5. H NMR (400 MHz, CD
CH ), 2.77 (q, 4H, CH
m, CH , coordinated L–L), 2.94 (q, CH
s, CH , uncoordinated L–L), 2.52 (q, CH
6
H
14
F
4
GeS
2
·CH
2
Cl
2
(383.84): calcd. C 21.9, H 4.2; found
{[9]aneS
3
1
◦
4
4
2
Cl
2
, 25 C): d = 2.98 (s, 4H,
)
n
SMe}] have been detected in solution
◦
4
2
2
2
), 1.35 (t, 6H, Me); (−70 C): d = 3.36 (br
1
19
16,17
at low temperatures by Sn NMR spectroscopy).
Comparison
2
2
, coordinated L–L), 2.64
of X-ray structural data on [SnX
4
{RS(CH
2
)
n
SR}] (X = F, Cl or
(
2
2
, uncoordinated L–L),
Br; n = 2 or 3) shows a small increase in d(Sn–S) as the halogen
1
.34 (t, Me, coordinated L–L), 1.16 (t, Me, uncoordinated L–L).
˚
˚
19
1
◦
2
changes F (2.584(1)–2.603(1) A) → Cl (2.619(2)–2.677(2) A) →
F{ H} NMR (CD
2
Cl
2
, −50 C): d = −117.9 (t), −85.6 (t, JFF
=
˚
−1
Br (2.700(7) A), the same trend seen in complexes with hard N-
7
6
4 Hz). IR (Nujol): 649, 635, 620, 605 sh m(GeF) cm . Raman:
3
,4
or O-donor ligands and softer phosphines, consistent with the
fluoride being the strongest Lewis acid towards the dithioethers.
Comparing the solution NMR data is less secure mainly due to
−1
52, 633, 620, 598 m(GeF) cm .
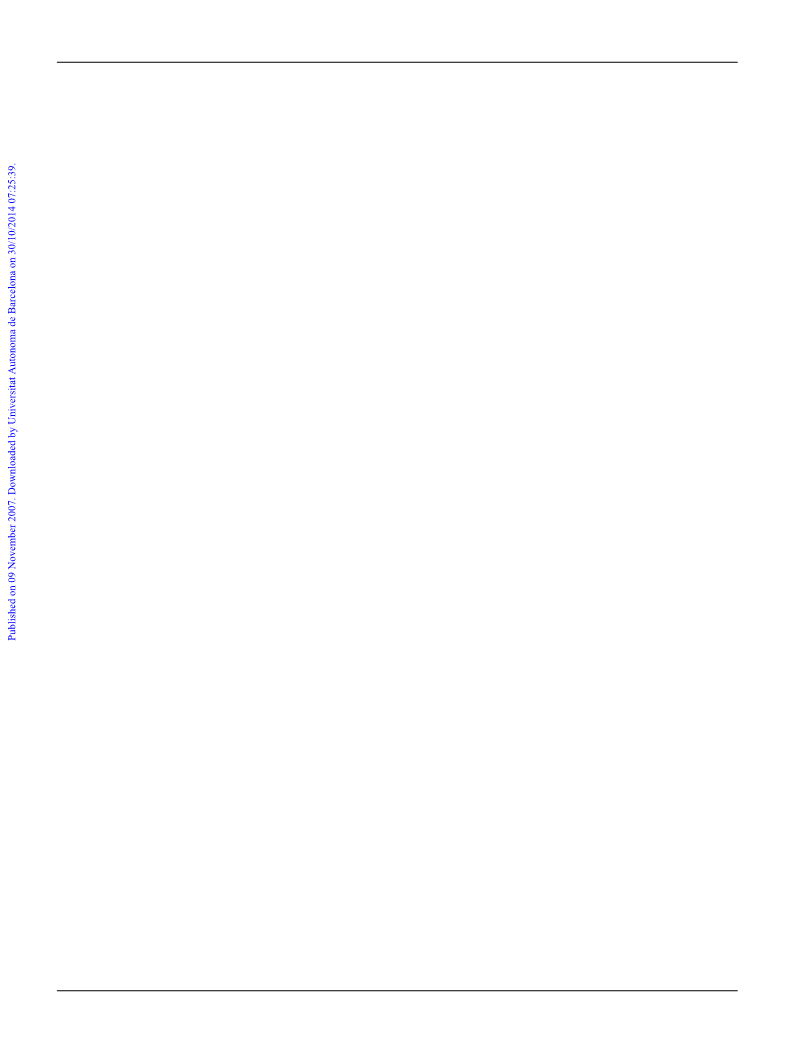
[
GeF
4
{MeS(CH
{MeS(CH
2
)
2
SMe}]
the tendency of the fluoride adducts to deposit SnF
is clear that the [SnX
4
polymer. It
[GeF
4
2
)
2
SMe}] was made analogously to the
4
{RS(CH SR}] (X = Cl or Br) are also
2
)
n
EtS(CH
2
)
2
SEt complex above, but using more CH
2
Cl
2
due
undergoing rapid exchange in solution at ambient temperatures
and on cooling the resonances sharpen and split as exchange
to the lower solubility of the target complex. 2,5-Dithiahexane
(0.015 g, 0.12 mmol) was added to a solution of [GeF (CH CN) ]
16
slows and invertomers are seen. However, in these cases dis-
sociation produces the tetrahedral molecular SnX which remains
in solution. The VT NMR results suggest that the binding of the
4
3
2
(0.023 g, 0.10 mmol) in CH
2
2
Cl (40 mL) at room temperature.
4
After stirring for 10 min, the solution was concentrated slowly
by evaporation to give a colourless crystalline solid which was
dithioethers in solution is SnF
4
∼ SnCl
4
> SnBr
4
>> SnI .
4
1
collected by filtration. Yield >60%. H NMR (400 MHz, CD
2
2
Cl ,
◦
19
1
2
(
(
6
5 C): d = 2.72 (s, 4H, CH
2
), 2.15 (s, 6H, Me). F{ H} NMR
Conclusions
◦
2
CD
2
Cl
2
, −50 C): d = −123.0 (t), −87.0 (t, JFF = 77 Hz). IR
−
1
Nujol): 645 sh, 629, 614, 590 sh m(GeF) cm . Raman: 651, 642,
The compounds described here represent the first authenticated
examples of main group metal/metalloid fluoride complexes
containing soft thioether ligands and also the first thioether
adducts of Ge. The results demonstrate that the molecular
] is a useful synthon to allow entry into these ex-
tremely unusual soft/hard donor/acceptor adducts. The absence
−
1
15, 589 m(GeF) cm .
{MeS(CH SMe}]
MeS(CH SMe (0.12 g, 1.00 mmol) was added dropwise to
[
SnF
4
)
2 2
[GeF
4
(MeCN)
2
)
2 2
a solution of [SnF
4
(MeCN) ] (0.278 g, 1.00 mmol) in CH Cl
2
2
2
of any evidence for adduct formation using GeCl
4
with similar
(
10 mL) and stirred for 0.5 h. Some white precipitate formed; the
9
thioether ligands shows that GeF
4
is a significantly better Lewis
solution was decanted off, and was left in the freezer for one week,
when it deposited a white powder, which was collected by filtration
acid even towards soft donor ligands, and will provide a starting
point for development of a substantial new coordination chemistry
for germanium(IV).
and dried in vacuo. Yield 0.22 g, 69%. C
C 15.2, H 3.2; found C 14.9, H 2.4. H NMR (300 MHz, CDCl
4
H
10
F
4
2
S Sn (316.94): calcd.
1
3
,
◦
19
1
2
5 C): d = 2.69 (s, 4H, CH
2
), 2.11 (s, 6H, Me). F{ H} NMR
◦
1
119
Experimental
(CH Cl /CD Cl , −70 C): d = −133.1 (t, J( SnF) = 2662,
2
2
2
2
1
117
1
119
1
117
J( SnF) = 2452 Hz), −159.5 (t, J( SnF) = 2221, J( SnF)
GeF
GeF
MeCN and CH
Aldrich) was dried over 4A molecular sieves. The dithioethers
4
was obtained from Aldrich and converted into
2
∼
2070 or not resolved, JFF = 53 Hz). IR (Nujol): 556 sh, 567 s,
6,11
12
[
4
(MeCN)
2
],
and [SnF
4
(MeCN)
2
] made as described.
. Me S
−
1
br, 588 sh m(SnF) cm .
2
Cl
2
were dried by distillation from CaH
2
2
(
[
SnF
4
{EtS(CH
2
)
2
SEt}]
18
were made as described and stored over molecular sieves. All
reactions were conducted using Schlenk, vacuum line and glove-
box techniques and under a dry dinitrogen atmosphere.
[SnF
4
(MeCN)
2
] (0.278 g, 1.00 mmol) was added to a solution of
EtS(CH SEt (0.34 g, 2.30 mmol) in CH
2
)
2
2
2
Cl (10 mL) and stirred
Infra-red spectra were recorded as Nujol mulls on a Perkin
Elmer PE 983G spectrometer. Raman spectra used a Perkin
Elmer FT-Raman 2000R with a Nd:YAG laser. H and F{ H}
for 1 h. A small amount of white precipitate occurred and the
reaction mixture was left in the freezer for 2 weeks where a few
colourless crystals grew on the walls of the Schlenk tube which
1
19
1
5
36 | Dalton Trans., 2008, 533–538
This journal is © The Royal Society of Chemistry 2008

 Davis, Martin F.
Davis, Martin F.
 Levason, William
Levason, William
 Reid, Gillian
Reid, Gillian
 Webster, Michael
Webster, Michael
 Zhang, Wenjian
Zhang, Wenjian