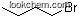Products Categories
| CAS No.: | 149-64-4 |
|---|---|
| Name: | Scopolamine butylbromide |
| Article Data: | 3 |
| Molecular Structure: | |
|
|
|
| Formula: | C21H30NO4.Br |
| Molecular Weight: | 440.377 |
| Synonyms: | 1aH,5aH-Tropanium, 8-butyl-6b,7b-epoxy-3a-hydroxy-,bromide, (-)-tropate (8CI);3-Oxa-9-azoniatricyclo[3.3.1.02,4]nonane,9-butyl-7-[(2S)-3-hydroxy-1-oxo-2-phenylpropoxy]-9-methyl-, bromide, (1a,2b,4b,5a,7b)- (9CI);N-Butylscopolammonium bromide (6CI,7CI);(-)-Scopolaminebutylbromide;N-Butylhyoscinium bromide;N-Butylscopolaminium bromide; |
| EINECS: | 205-744-1 |
| Melting Point: | 142-144 °C |
| Solubility: | Water: 50 mg/mL |
| Appearance: | White crystalline solid |
| Hazard Symbols: |
 Xn Xn
|
| Risk Codes: | 22 |
| PSA: | 59.06000 |
| LogP: | -0.80420 |
- 483366-12-7(2S,4R)-1-Boc-2-cyano-4-hydroxypyrrolidine
- 173606-50-3BOC-10-AMINODECANOIC ACID
- 361456-36-2METHYL (R)-(+)-ISOCYANATO-3-PHENYLPROPI&
- 5156-58-1N-(1-Benzyl-4-pipperidinyl)-N-phenylpropanamide HCl
- 81281-59-67-Benzylideneaminotheophylline
- 50288-62-5threo-Phenyl-2-piperidyl acetamide
- 82993-81-5D-threo-Ritalinic acid hydrochloride
- 47087-37-6Z-D-Glu-OMe
- 73441-42-6METHYL-5-CHLORO-2,2-DIMETHYLVALERATE
- 211914-50-0N-[[2-[[[4-(Aminoiminomethyl)phenyl]amino]methyl]-1-methyl-1H-benzimidazol-5-yl]carbonyl]-N-(2-pyridinyl)-beta-alanine ethyl ester hydrochloride

| Conditions | Yield |
|---|---|
| In acetonitrile Ambient temperature; |

| Conditions | Yield |
|---|---|
| With acetonitrile |

- 149-64-4
hyoscine-N-butyl bromide

| Conditions | Yield |
|---|---|
| With sodium methylate In methanol at 40℃; for 3h; | 64% |

| Conditions | Yield |
|---|---|
| With CH3COONa buffer; acetic acid In water pH 4.20; |

| Conditions | Yield |
|---|---|
| In ethanol; water for 0.25h; |
- 149-64-4
hyoscine-N-butyl bromide

| Conditions | Yield |
|---|---|
| Multi-step reaction with 2 steps 1: sodium methylate / methanol / 3 h / 40 °C 2: chloroform / 5 h / 40 °C View Scheme |
- 149-64-4
hyoscine-N-butyl bromide

| Conditions | Yield |
|---|---|
| Multi-step reaction with 3 steps 1: sodium methylate / methanol / 3 h / 40 °C 2: chloroform / 5 h / 40 °C 3: hydrogenchloride / water View Scheme |

| Conditions | Yield |
|---|---|
| With ruthenium trichloride; potassium metaperiodate; diperiodatocuprate(III) dihydrate; water; potassium nitrate; potassium hydroxide at 28℃; for 6h; Kinetics; Catalytic behavior; Concentration; Temperature; Inert atmosphere; |

What can I do for you?
Get Best Price
Specification
The Scopolamine-n-butyl bromide with CAS registry number of 149-64-4 is also known as 3-Oxa-9-azoniatricyclo[3.3.1.02,4]nonane,9-butyl-7-[(2S)-3-hydroxy-1-oxo-2-phenylpropoxy]-9-methyl-, bromide, (1a,2b,4b,5a,7b)- (9Cl). The systematic name is 1-alpha-H,5-alpha-H-Tropanium, 8-butyl-6-beta,7-beta-epoxy-3-alpha-hydroxy-, bromide, (-)-tropate. It belongs to product categories of Miscellaneous; Chiral Reagents; Intermediates & Fine Chemicals; Pharmaceuticals; Acetylcholine Receptor. Its EINECS registry number is 205-744-1. In addition, the formula is C21H30NO4.Br and the molecular weight is 440.37. This chemical is a white crystalline solid and should be stored in sealed containers in cool, dry place. Besides, this chemical is a extracted from dried flowers of datura metm L and used as pharmaceutical raw material. It will be harmful if swallowed.
Physical properties about Scopolamine-n-butyl bromide are: (1)#H bond acceptors: 5; (2)#H bond donors: 1; (3)#Freely Rotating Bonds: 9; (4)Polar Surface Area: 59.06Å2; (5)Rotatable Bond Count: 8; (6)Exact Mass: 439.135821; (7)MonoIsotopic Mass: 439.135821; (8)Topological Polar Surface Area: 59.1; (9)Heavy Atom Count: 27; (10)Complexity: 500; (11)Defined Atom StereoCenter Count: 3; (12)Undefined Atom StereoCenter Count: 2; (13)Covalently-Bonded Unit Count: 2.
You can still convert the following datas into molecular structure:
1. SMILES: [Br-].OC[C@H](c1ccccc1)C(=O)O[C@@H]2C[C@@H]3[C@H]4O[C@H]4[C@H](C2)[N+]3(C)CCCC
2. InChI: InChI=1/C21H30NO4.BrH/c1-3-4-10-22(2)17-11-15(12-18(22)20-19(17)26-20)25-21(24)16(13-23)14-8-6-5-7-9-14;/h5-9,15-20,23H,3-4,10-13H2,1-2H3;1H/q+1;/p-1/t15-,16-,17-,18+,19-,20+,22?;/m1./s1
3. InChIKey: HOZOZZFCZRXYEK-INRBTMLCBV
4. Std. InChI: InChI=1S/C21H30NO4.BrH/c1-3-4-10-22(2)17-11-15(12-18(22)20-19(17)26-20)25-21(24)16(13-23)14-8-6-5-7-9-14;/h5-9,15-20,23H,3-4,10-13H2,1-2H3;1H/q+1;/p-1/t15-,16-,17-,18+,19-,20+,22?;/m1./s1
5. Std. InChIKey: HOZOZZFCZRXYEK-GSWUYBTGSA-M
The toxicity data is as follows:
| Organism | Test Type | Route | Reported Dose (Normalized Dose) | Effect | Source |
|---|---|---|---|---|---|
| guinea pig | LD50 | subcutaneous | 155mg/kg (155mg/kg) | Archives Internationales de Pharmacodynamie et de Therapie. Vol. 137, Pg. 375, 1962. Link to PubMed | |
| mammal (species unspecified) | LD50 | intraperitoneal | 65mg/kg (65mg/kg) | Farmaco, Edizione Scientifica. Vol. 39, Pg. 3, 1984. Link to PubMed | |
| mammal (species unspecified) | LD50 | oral | 750mg/kg (750mg/kg) | Farmaco, Edizione Scientifica. Vol. 39, Pg. 3, 1984. Link to PubMed | |
| mammal (species unspecified) | LD50 | subcutaneous | 155mg/kg (155mg/kg) | Farmaco, Edizione Scientifica. Vol. 39, Pg. 3, 1984. Link to PubMed | |
| mouse | LD50 | intraperitoneal | 65mg/kg (65mg/kg) | United States Patent Document. Vol. #4558054, | |
| mouse | LD50 | intravenous | 10300ug/kg (10.3mg/kg) | BEHAVIORAL: CONVULSIONS OR EFFECT ON SEIZURE THRESHOLD BEHAVIORAL: ATAXIA LUNGS, THORAX, OR RESPIRATION: RESPIRATORY DEPRESSION | Oyo Yakuri. Pharmacometrics. Vol. 5, Pg. 581, 1971. |
| mouse | LD50 | oral | 1170mg/kg (1170mg/kg) | BEHAVIORAL: CONVULSIONS OR EFFECT ON SEIZURE THRESHOLD | Therapie. Vol. 14, Pg. 1096, 1959. Link to PubMed |
| mouse | LD50 | subcutaneous | 243mg/kg (243mg/kg) | Yaoxue Tongbao. Bulletin of Pharmacology. Vol. 18, Pg. 217, 1983. | |
| rat | LD50 | intraduodenal | 180mg/kg (180mg/kg) | Archives Internationales de Pharmacodynamie et de Therapie. Vol. 180, Pg. 155, 1969. | |
| rat | LD50 | intravenous | 24mg/kg (24mg/kg) | SENSE ORGANS AND SPECIAL SENSES: MYDRIASIS (PUPILLARY DILATION): EYE AUTONOMIC NERVOUS SYSTEM: "SMOOTH MUSCLE RELAXANT (MECHANISM UNDEFINED, SPASMOLYTIC)" | Oyo Yakuri. Pharmacometrics. Vol. 23, Pg. 461, 1982. |
| rat | LD50 | oral | 1040mg/kg (1040mg/kg) | Archives Internationales de Pharmacodynamie et de Therapie. Vol. 180, Pg. 155, 1969. | |
| rat | LD50 | subcutaneous | 450mg/kg (450mg/kg) | Arzneimittel-Forschung. Drug Research. Vol. 4, Pg. 341, 1954. |