Products Categories
| CAS No.: | 361441-97-6 |
|---|---|
| Name: | (S) - N- Boc- adamantylglycine |
| Article Data: | 6 |
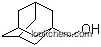
| Molecular Structure: | |
|
|
|
| Formula: | C17H27NO4 |
| Molecular Weight: | 309.406 |
| Synonyms: | (S) - N- Boc- adamantylglycine |
| Density: | 1.193 g/cm3 |
| Melting Point: | >300 °C (decomp)(Solv: ethyl acetate (141-78-6); ligroine (8032-32-4)) |
| Boiling Point: | 460.324 °C at 760 mmHg |
| Flash Point: | 232.196 °C |
| PSA: | 79.12000 |
| LogP: | 3.38510 |
- 483366-12-7(2S,4R)-1-Boc-2-cyano-4-hydroxypyrrolidine
- 361456-36-2METHYL (R)-(+)-ISOCYANATO-3-PHENYLPROPI&
- 5156-58-1N-(1-Benzyl-4-pipperidinyl)-N-phenylpropanamide HCl
- 81281-59-67-Benzylideneaminotheophylline
- 50288-62-5threo-Phenyl-2-piperidyl acetamide
- 82993-81-5D-threo-Ritalinic acid hydrochloride
- 47087-37-6Z-D-Glu-OMe
- 73441-42-6METHYL-5-CHLORO-2,2-DIMETHYLVALERATE
- 68439-39-4Poly(oxy-1,2-ethanediyl), alpha-(2-ethylhexyl)-omega-hydroxy-,
- 852475-26-4MC1568
- 24424-99-5
di-tert-butyl dicarbonate

- 102502-64-7
(S)-(+)-(adamant-1-yl)aminoacetic acid hydrochloride

- 361441-97-6
(alpha S)-alpha-[[(1,1-dimethylethoxy)carbonyl]amino]tricyclo[3.3.1.13,7]decane-1-acetic acid

| Conditions | Yield |
|---|---|
| With potassium carbonate In DMF (N,N-dimethyl-formamide) at 20℃; for 19h; | 92% |
| With triethylamine In methanol | 80% |
| Stage #1: (S)-(+)-(adamant-1-yl)aminoacetic acid hydrochloride With sodium hydroxide In water at 0 - 5℃; for 0.25h; Stage #2: di-tert-butyl dicarbonate In tetrahydrofuran; water at 0 - 5℃; | 80 g |
| With potassium carbonate In N,N-dimethyl-formamide at 20℃; for 19h; Inert atmosphere; | 4.07 g |

- 361441-97-6
(alpha S)-alpha-[[(1,1-dimethylethoxy)carbonyl]amino]tricyclo[3.3.1.13,7]decane-1-acetic acid

| Conditions | Yield |
|---|---|
| With sodium chlorite; potassium dihydrogenphosphate; 2-methyl-but-2-ene In tert-butyl alcohol at 0 - 10℃; for 19.5h; | 82% |
- 24424-99-5
di-tert-butyl dicarbonate

- 95853-35-3
(S)-(+)-β-amino-1-adamantaneacetic acid

- 361441-97-6
(alpha S)-alpha-[[(1,1-dimethylethoxy)carbonyl]amino]tricyclo[3.3.1.13,7]decane-1-acetic acid

| Conditions | Yield |
|---|---|
| With potassium carbonate In N,N-dimethyl-formamide for 19h; | 4.07 g |
- 361441-95-4
(S)-2-(adamantan-1-yl)-2-(((R)-2-hydroxy-1-phenylethyl)amino)acetonitrile

- 361441-97-6
(alpha S)-alpha-[[(1,1-dimethylethoxy)carbonyl]amino]tricyclo[3.3.1.13,7]decane-1-acetic acid

| Conditions | Yield |
|---|---|
| Multi-step reaction with 3 steps 1: 78 percent / aq. HCl; AcOH / 18 h / 80 °C 2: H2; AcOH / Pd(OH)2/C / methanol / 18 h / 2585.74 Torr 3: 4.07 g / K2CO3 / dimethylformamide / 19 h View Scheme | |
| Multi-step reaction with 3 steps 1: hydrogenchloride; water / acetic acid / 80 °C 2: hydrogen; palladium 10% on activated carbon / acetic acid 3: triethylamine / methanol View Scheme | |
| Multi-step reaction with 3 steps 1.1: hydrogenchloride; acetic acid; water / 20 - 95 °C 2.1: acetic acid; hydrogen; 10% palladium hydroxide on charcoal / methanol / 20 °C 3.1: sodium hydroxide / water / 0.25 h / 0 - 5 °C 3.2: 0 - 5 °C View Scheme | |
| Multi-step reaction with 3 steps 1: hydrogenchloride; water / acetic acid / 18 h / 80 °C 2: hydrogen; 20% palladium hydroxide-activated charcoal / methanol; acetic acid / 18 h / 2585.81 Torr 3: potassium carbonate / N,N-dimethyl-formamide / 19 h / 20 °C / Inert atmosphere View Scheme |

- 361441-97-6
(alpha S)-alpha-[[(1,1-dimethylethoxy)carbonyl]amino]tricyclo[3.3.1.13,7]decane-1-acetic acid

| Conditions | Yield |
|---|---|
| Multi-step reaction with 2 steps 1: H2; AcOH / Pd(OH)2/C / methanol / 18 h / 2585.74 Torr 2: 4.07 g / K2CO3 / dimethylformamide / 19 h View Scheme | |
| Multi-step reaction with 2 steps 1: hydrogen; palladium 10% on activated carbon / acetic acid 2: triethylamine / methanol View Scheme | |
| Multi-step reaction with 2 steps 1.1: acetic acid; hydrogen; 10% palladium hydroxide on charcoal / methanol / 20 °C 2.1: sodium hydroxide / water / 0.25 h / 0 - 5 °C 2.2: 0 - 5 °C View Scheme | |
| Multi-step reaction with 2 steps 1: hydrogen; 20% palladium hydroxide-activated charcoal / methanol; acetic acid / 18 h / 2585.81 Torr 2: potassium carbonate / N,N-dimethyl-formamide / 19 h / 20 °C / Inert atmosphere View Scheme |
- 770-71-8
1-adamantanemethanol

- 361441-97-6
(alpha S)-alpha-[[(1,1-dimethylethoxy)carbonyl]amino]tricyclo[3.3.1.13,7]decane-1-acetic acid

| Conditions | Yield |
|---|---|
| Multi-step reaction with 5 steps 1: 98 percent / oxalyl chloride; DMSO; Et3N / CH2Cl2 / 1 h / -78 °C 2: 65 percent / NaHSO3 / H2O; methanol / 16 h / Heating 3: 78 percent / aq. HCl; AcOH / 18 h / 80 °C 4: H2; AcOH / Pd(OH)2/C / methanol / 18 h / 2585.74 Torr 5: 4.07 g / K2CO3 / dimethylformamide / 19 h View Scheme | |
| Multi-step reaction with 5 steps 1.1: potassium bromide; sodium hydrogencarbonate / dichloromethane / 0.25 h / 0 - 5 °C 1.2: 0 - 5 °C 2.1: sodium hydrogensulfite / methanol; water / 0 - 55 °C 3.1: hydrogenchloride; acetic acid; water / 20 - 95 °C 4.1: acetic acid; hydrogen; 10% palladium hydroxide on charcoal / methanol / 20 °C 5.1: sodium hydroxide / water / 0.25 h / 0 - 5 °C 5.2: 0 - 5 °C View Scheme | |
| Multi-step reaction with 5 steps 1.1: oxalyl dichloride; dimethyl sulfoxide / dichloromethane / 1 h / -78 °C / Inert atmosphere 1.2: 0.5 h / -78 °C / Inert atmosphere 2.1: sodium hydrogensulfite / methanol; water / 18 h / 0 - 20 °C / Reflux 3.1: hydrogenchloride; water / acetic acid / 18 h / 80 °C 4.1: hydrogen; 20% palladium hydroxide-activated charcoal / methanol; acetic acid / 18 h / 2585.81 Torr 5.1: potassium carbonate / N,N-dimethyl-formamide / 19 h / 20 °C / Inert atmosphere View Scheme |
- 828-51-3
1-Adamantanecarboxylic acid

- 361441-97-6
(alpha S)-alpha-[[(1,1-dimethylethoxy)carbonyl]amino]tricyclo[3.3.1.13,7]decane-1-acetic acid

| Conditions | Yield |
|---|---|
| Multi-step reaction with 7 steps 1: 100 percent / diethyl ether; hexane; methanol / 3 h / 20 °C 2: 96 percent / LiAlH4 / tetrahydrofuran / 1.5 h / 0 - 20 °C 3: 98 percent / oxalyl chloride; DMSO; Et3N / CH2Cl2 / 1 h / -78 °C 4: 65 percent / NaHSO3 / H2O; methanol / 16 h / Heating 5: 78 percent / aq. HCl; AcOH / 18 h / 80 °C 6: H2; AcOH / Pd(OH)2/C / methanol / 18 h / 2585.74 Torr 7: 4.07 g / K2CO3 / dimethylformamide / 19 h View Scheme | |
| Multi-step reaction with 7 steps 1.1: thionyl chloride / 3 h / 20 °C 2.1: lithium aluminium tetrahydride / tetrahydrofuran / 2 h / 0 - 20 °C / Inert atmosphere 3.1: potassium bromide; sodium hydrogencarbonate / dichloromethane / 0.25 h / 0 - 5 °C 3.2: 0 - 5 °C 4.1: sodium hydrogensulfite / methanol; water / 0 - 55 °C 5.1: hydrogenchloride; acetic acid; water / 20 - 95 °C 6.1: acetic acid; hydrogen; 10% palladium hydroxide on charcoal / methanol / 20 °C 7.1: sodium hydroxide / water / 0.25 h / 0 - 5 °C 7.2: 0 - 5 °C View Scheme | |
| Multi-step reaction with 7 steps 1.1: methanol; diethyl ether; hexane / 3 h / 20 °C 2.1: lithium aluminium tetrahydride / tetrahydrofuran / 1.5 h / 20 °C / Inert atmosphere 3.1: oxalyl dichloride; dimethyl sulfoxide / dichloromethane / 1 h / -78 °C / Inert atmosphere 3.2: 0.5 h / -78 °C / Inert atmosphere 4.1: sodium hydrogensulfite / methanol; water / 18 h / 0 - 20 °C / Reflux 5.1: hydrogenchloride; water / acetic acid / 18 h / 80 °C 6.1: hydrogen; 20% palladium hydroxide-activated charcoal / methanol; acetic acid / 18 h / 2585.81 Torr 7.1: potassium carbonate / N,N-dimethyl-formamide / 19 h / 20 °C / Inert atmosphere View Scheme |
- 2094-74-8
1-Adamantanecarbaldehyde

- 361441-97-6
(alpha S)-alpha-[[(1,1-dimethylethoxy)carbonyl]amino]tricyclo[3.3.1.13,7]decane-1-acetic acid

| Conditions | Yield |
|---|---|
| Multi-step reaction with 4 steps 1: 65 percent / NaHSO3 / H2O; methanol / 16 h / Heating 2: 78 percent / aq. HCl; AcOH / 18 h / 80 °C 3: H2; AcOH / Pd(OH)2/C / methanol / 18 h / 2585.74 Torr 4: 4.07 g / K2CO3 / dimethylformamide / 19 h View Scheme | |
| Multi-step reaction with 4 steps 1: sodium hydrogensulfite / 0 - 60 °C 2: hydrogenchloride; water / acetic acid / 80 °C 3: hydrogen; palladium 10% on activated carbon / acetic acid 4: triethylamine / methanol View Scheme | |
| Multi-step reaction with 4 steps 1.1: sodium hydrogensulfite / methanol; water / 0 - 55 °C 2.1: hydrogenchloride; acetic acid; water / 20 - 95 °C 3.1: acetic acid; hydrogen; 10% palladium hydroxide on charcoal / methanol / 20 °C 4.1: sodium hydroxide / water / 0.25 h / 0 - 5 °C 4.2: 0 - 5 °C View Scheme | |
| Multi-step reaction with 4 steps 1: sodium hydrogensulfite / methanol; water / 18 h / 0 - 20 °C / Reflux 2: hydrogenchloride; water / acetic acid / 18 h / 80 °C 3: hydrogen; 20% palladium hydroxide-activated charcoal / methanol; acetic acid / 18 h / 2585.81 Torr 4: potassium carbonate / N,N-dimethyl-formamide / 19 h / 20 °C / Inert atmosphere View Scheme |
- 711-01-3
methyl adamantane-1-carboxylate

- 361441-97-6
(alpha S)-alpha-[[(1,1-dimethylethoxy)carbonyl]amino]tricyclo[3.3.1.13,7]decane-1-acetic acid

| Conditions | Yield |
|---|---|
| Multi-step reaction with 6 steps 1: 96 percent / LiAlH4 / tetrahydrofuran / 1.5 h / 0 - 20 °C 2: 98 percent / oxalyl chloride; DMSO; Et3N / CH2Cl2 / 1 h / -78 °C 3: 65 percent / NaHSO3 / H2O; methanol / 16 h / Heating 4: 78 percent / aq. HCl; AcOH / 18 h / 80 °C 5: H2; AcOH / Pd(OH)2/C / methanol / 18 h / 2585.74 Torr 6: 4.07 g / K2CO3 / dimethylformamide / 19 h View Scheme | |
| Multi-step reaction with 6 steps 1.1: lithium aluminium tetrahydride / tetrahydrofuran / 2 h / 0 - 20 °C / Inert atmosphere 2.1: potassium bromide; sodium hydrogencarbonate / dichloromethane / 0.25 h / 0 - 5 °C 2.2: 0 - 5 °C 3.1: sodium hydrogensulfite / methanol; water / 0 - 55 °C 4.1: hydrogenchloride; acetic acid; water / 20 - 95 °C 5.1: acetic acid; hydrogen; 10% palladium hydroxide on charcoal / methanol / 20 °C 6.1: sodium hydroxide / water / 0.25 h / 0 - 5 °C 6.2: 0 - 5 °C View Scheme | |
| Multi-step reaction with 6 steps 1.1: lithium aluminium tetrahydride / tetrahydrofuran / 1.5 h / 20 °C / Inert atmosphere 2.1: oxalyl dichloride; dimethyl sulfoxide / dichloromethane / 1 h / -78 °C / Inert atmosphere 2.2: 0.5 h / -78 °C / Inert atmosphere 3.1: sodium hydrogensulfite / methanol; water / 18 h / 0 - 20 °C / Reflux 4.1: hydrogenchloride; water / acetic acid / 18 h / 80 °C 5.1: hydrogen; 20% palladium hydroxide-activated charcoal / methanol; acetic acid / 18 h / 2585.81 Torr 6.1: potassium carbonate / N,N-dimethyl-formamide / 19 h / 20 °C / Inert atmosphere View Scheme |

- 361441-97-6
(alpha S)-alpha-[[(1,1-dimethylethoxy)carbonyl]amino]tricyclo[3.3.1.13,7]decane-1-acetic acid

| Conditions | Yield |
|---|---|
| With N-ethyl-N,N-diisopropylamine; N-[(dimethylamino)-3-oxo-1H-1,2,3-triazolo[4,5-b]pyridin-1-yl-methylene]-N-methylmethanaminium hexafluorophosphate In N,N-dimethyl-formamide at 0 - 20℃; | 93% |

What can I do for you?
Get Best Price
Specification
The (S)-Adamantylglycine is an organic compound with the formula C17H27NO4. The systematic name of this chemical is (2S)-[(tert-butoxycarbonyl)amino](tricyclo[3.3.1.1~3,7~]dec-1-yl)ethanoic acid. With the CAS registry number 361441-97-6, it is also named as N-Boc-L-Adamantylglycine.
Physical properties about (S)-Adamantylglycine are: (1)ACD/LogP: 3.80; (2)#H bond acceptors: 5; (3)#H bond donors: 2; (4)#Freely Rotating Bonds: 5; (5)Polar Surface Area: 75.63 Å2; (6)Index of Refraction: 1.54; (7)Molar Refractivity: 81.419 cm3; (8)Molar Volume: 259.315 cm3; (9)Polarizability: 32.277×10-24cm3; (10)Surface Tension: 48.088 dyne/cm; (11)Density: 1.193 g/cm3; (12)Flash Point: 232.196 °C; (13)Enthalpy of Vaporization: 79.007 kJ/mol; (14)Boiling Point: 460.324 °C at 760 mmHg.
You can still convert the following datas into molecular structure:
(1)SMILES: O=C(OC(C)(C)C)N[C@H](C(=O)O)C13CC2CC(CC(C1)C2)C3
(2)InChI: InChI=1/C17H27NO4/c1-16(2,3)22-15(21)18-13(14(19)20)17-7-10-4-11(8-17)6-12(5-10)9-17/h10-13H,4-9H2,1-3H3,(H,18,21)(H,19,20)/t10?,11?,12?,13-,17?/m1/s1
(3)InChIKey: LJUATQZYDYSZPV-CQCMJFKXBN
(4)Std. InChI: InChI=1S/C17H27NO4/c1-16(2,3)22-15(21)18-13(14(19)20)17-7-10-4-11(8-17)6-12(5-10)9-17/h10-13H,4-9H2,1-3H3,(H,18,21)(H,19,20)/t10?,11?,12?,13-,17?/m1/s1
(5)Std. InChIKey: LJUATQZYDYSZPV-CQCMJFKXSA-N