Products Categories
Basic Information
| CAS No.: | 518060-42-9 |
|---|---|
| Name: | (R)-3-(1-AMINOETHYL)PHENOL |
| Article Data: | 7 |
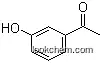
| Molecular Structure: | |
|
|
|
| Formula: | C8H11 N O |
| Molecular Weight: | 137.181 |
| Synonyms: | 3[(1R)-1-AMINOETHYL] PHENOL;(R)-3-(1-AMINOETHYL)PHENOL;1-(R)-(3-METHOXYPHENOL)ETHYLAMINE;Phenol, 3-[(1R)-1-aminoethyl]- (9CI);Phenol, 3-[(1R)-1-aminoethyl]- |
| Density: | 1.096±0.06 g/cm3(Predicted) |
| Boiling Point: | 266.3±15.0 °C(Predicted) |
| PSA: | 46.25000 |
| LogP: | 2.91420 |
Related products
- 81281-59-67-Benzylideneaminotheophylline
- 82993-81-5D-threo-Ritalinic acid hydrochloride
- 73441-42-6METHYL-5-CHLORO-2,2-DIMETHYLVALERATE
- 68439-39-4Poly(oxy-1,2-ethanediyl), alpha-(2-ethylhexyl)-omega-hydroxy-,
- 852475-26-4MC1568
- 958254-66-51H-Imidazo[4,5-b]pyridine-2-carboxaldehyde, 1-methyl-, hydrochloride
- 99170-93-1N-Methyl-2-oxazolamine
- 717878-06-31-(4-fluorophenyl)-4-nitro-1H-imidazole
- 914458-26-7[5-(2-fluorophenyl)-1-pentyl-1H-pyrrol-3-yl]-1-naphthalenyl-Methanone
- 548773-13-32-amino-4,6-dimethylpyrimidine-5-carboxylic acid
Synthetic route
- 1372628-56-2
3-((R)-1-((R)-1-phenylethylamino)ethyl)phenol

- 518060-42-9
(R)-1-(3-hydroxyphenyl)ethylamine
Conditions

| Conditions | Yield |
|---|---|
| With palladium 10% on activated carbon; hydrogen In acetic acid at 20℃; under 3102.97 Torr; for 48h; Inert atmosphere; optical yield given as %ee; enantioselective reaction; | 89.7% |

- 518060-42-9
(R)-1-(3-hydroxyphenyl)ethylamine
Conditions

| Conditions | Yield |
|---|---|
| Stage #1: (R)-1-(3-tert-butoxyphenyl)ethylamine With sulfuric acid; water at 85℃; for 3h; pH=1; Stage #2: With sodium hydroxide; water pH=9.1; | 73% |
- 871728-33-5
(R)-[1-(3-methoxyphenyl)ethyl]carbamic acid tert-butyl ester

- 518060-42-9
(R)-1-(3-hydroxyphenyl)ethylamine
Conditions

| Conditions | Yield |
|---|---|
| With boron tribromide In dichloromethane at -78 - 20℃; |
- 88196-70-7
(R)-1-(3-methoxyphenyl)ethylamine

- 518060-42-9
(R)-1-(3-hydroxyphenyl)ethylamine
Conditions

| Conditions | Yield |
|---|---|
| Multi-step reaction with 2 steps 1: Et3N / CH2Cl2 / 5 h / 20 °C 2: BBr3 / CH2Cl2 / -78 - 20 °C View Scheme | |
| Stage #1: (R)-1-(3-methoxyphenyl)ethylamine With boron tribromide In dichloromethane at -78℃; Stage #2: With methanol In dichloromethane at 20℃; | |
| With boron tribromide In dichloromethane for 48h; Inert atmosphere; Cooling with ice; |
- 121-71-1
3-Hydroxyacetophenone

A
- 63720-38-7, 123982-81-0
3-hydroxy-(S)-α-phenethylamine

B
- 518060-42-9
(R)-1-(3-hydroxyphenyl)ethylamine
Conditions

| Conditions | Yield |
|---|---|
| With glucose dehydrogenase; D-Glucose; L-alanin; Arthrobacter sp. CNB05-01 ω-transaminase; nicotiamide adenine dinucleotide; lactate dehydrogenase at 30℃; for 24h; pH=7; aq. phosphate buffer; Enzymatic reaction; |

- 518060-42-9
(R)-1-(3-hydroxyphenyl)ethylamine
Conditions

| Conditions | Yield |
|---|---|
| Multi-step reaction with 2 steps 1: sodium tetrahydroborate / ethanol; ethyl acetate / 3 h / 0 °C / Inert atmosphere 2: palladium 10% on activated carbon; hydrogen / acetic acid / 48 h / 20 °C / 3102.97 Torr / Inert atmosphere View Scheme |
- 121-71-1
3-Hydroxyacetophenone

- 518060-42-9
(R)-1-(3-hydroxyphenyl)ethylamine
Conditions

| Conditions | Yield |
|---|---|
| Multi-step reaction with 3 steps 1: titanium(IV) isopropylate / ethyl acetate / 3 h / Inert atmosphere 2: sodium tetrahydroborate / ethanol; ethyl acetate / 3 h / 0 °C / Inert atmosphere 3: palladium 10% on activated carbon; hydrogen / acetic acid / 48 h / 20 °C / 3102.97 Torr / Inert atmosphere View Scheme |

A
- 63720-38-7, 123982-81-0
3-hydroxy-(S)-α-phenethylamine

B
- 518060-42-9
(R)-1-(3-hydroxyphenyl)ethylamine
Conditions

| Conditions | Yield |
|---|---|
| With sodium hydroxide In water Overall yield = 1.41 g; |

A
- 63720-38-7, 123982-81-0
3-hydroxy-(S)-α-phenethylamine

B
- 518060-42-9
(R)-1-(3-hydroxyphenyl)ethylamine
Conditions

| Conditions | Yield |
|---|---|
| With sodium hydroxide In water |
- 24424-99-5
di-tert-butyl dicarbonate

- 518060-42-9
(R)-1-(3-hydroxyphenyl)ethylamine

- 266369-69-1
(R)-[1-(3-hydroxyphenyl)ethyl]carbamic acid tert-butyl ester
Conditions

| Conditions | Yield |
|---|---|
| With triethylamine In methanol at 0℃; for 6h; Inert atmosphere; | 81.7% |
| With triethylamine In dichloromethane at 20℃; for 0.5h; | 66% |
| With triethylamine In dichloromethane at 20℃; for 0.5h; | |
| With triethylamine In dichloromethane for 1h; |
Raw Materials