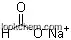Products Categories
| CAS No.: | 822-16-2 |
|---|---|
| Name: | Sodium stearate |
| Article Data: | 12 |
| Molecular Structure: | |
|
|
|
| Formula: | C18H35NaO2 |
| Molecular Weight: | 306.464 |
| Synonyms: | Octadecanoicacid, sodium salt (9CI);Stearic acid, sodium salt (8CI);AFCO-Chem B 65;AFCO-Chem NA;Bonderlube 234;Bonderlube 235;Edenor FHTI;FlexichemB;Nonsoul SN 1;Nonsoul SN 15;Prodhygine;Rhenogran NAST 50ACMF-GE1858;SNA 2000;SS 40N;Serfax MT 90;Sodium octadecanoate;Octadecanoic acid,sodium salt (1:1); |
| EINECS: | 212-490-5 |
| Density: | 0.2-0.3 g/cm3 |
| Melting Point: | 245 - 255 °C |
| Boiling Point: | 359.4 °C at 760 mmHg |
| Flash Point: | 162.4 °C |
| Solubility: | soluble in cold and hot water |
| Appearance: | white to off-white powder |
| Safety: | 24/25 |
| PSA: | 40.13000 |
| LogP: | 4.99780 |
- 82993-81-5D-threo-Ritalinic acid hydrochloride
- 852475-26-4MC1568
- 958254-66-51H-Imidazo[4,5-b]pyridine-2-carboxaldehyde, 1-methyl-, hydrochloride
- 99170-93-1N-Methyl-2-oxazolamine
- 914458-26-7[5-(2-fluorophenyl)-1-pentyl-1H-pyrrol-3-yl]-1-naphthalenyl-Methanone
- 894852-01-87-BROMO-2,2-DIMETHYL-2H-PYRIDO[3,2-B][1,4]OXAZIN-3(4H)-ONE
- 90221-55-92-bromo-5-methylbenzaldehyde
- 885590-99-82,3-DIFLUORO-4-IODOBENZALDEHYDE
- 97730-31-9(S)-4'-(2-Methylbutyl)Biphenyl-4-Carbonitrile
- 926293-55-26-Bromo-2-methylpyridine-3-carboxaldehyde
- 3420-72-2, 37951-13-6, 64680-84-8
2'-hydroxy-4,4',6'-trimethoxychalcone

- 638-08-4
stearic anhydride

B
- 822-16-2
sodium stearate

| Conditions | Yield |
|---|---|
| In pyridine; ethyl acetate | A 91% B n/a |


| Conditions | Yield |
|---|---|
| In ethanol |
- 75-75-2
methanesulfonic acid

- 1912-32-9
n-butyl methanesulfonate

- 123-95-5
n-butyl stearate

- 57-11-4
stearic acid

A
- 822-16-2
sodium stearate

B
- 2386-57-4
methanesulfonic acid sodium salt

| Conditions | Yield |
|---|---|
| With sodium hydroxide; water In butan-1-ol at 50 - 175℃; for 0.666667 - 1h; Conversion of starting material; |

- 82249-33-0BIS-DIGLYCERYL POLYACYLADIPATE-2
- 822-55-9Imidazole-4-methanol
- 82277-17-62-amino-6-(cyclopentyloxycarbonylamino)hexanoic Acid
- 822-84-43-Nitrothiophene
- 823202-99-9(2S)-beta-Alanyl-L-prolyl-2,4-diamino-N-(phenylmethyl)butanamide acetate
- 823204-45-1Bicyclo[3.1.1]hept-2-ene, 4-ethoxy-2,6,6-trimethyl-
- 823-22-3delta-Hexalactone
- 823-31-41,3,2-Dioxaphospholane, 2-ethoxy-, 2-oxide
- 104987-11-3Tacrolimus
- 141-53-7Sodium formate
- 8001-54-5Quaternary ammonium compounds, alkylbenzyldimethyl, chlorides
- 9003-39-8Povidone
- 10161-34-9Trenbolone acetate
- 402957-28-2Telaprevir
- 68-19-9Cyanocobalamin
Chemistry
IUPAC Name: Sodium octadecanoate
Following is the structure of Sodium stearate (CAS NO.822-16-2):

Molecular Formula: C18H35NaO2
Molecular Weight: 306.46
|EINECS: 212-490-5
Flash Point: 162.4 °C
Storage temp.: 2-8 °C
Enthalpy of Vaporization: 63.84 kJ/mol
Boiling Point: 359.4 °C at 760 mmHg
Vapour Pressure: 8.58E-06 mmHg at 25 °C
Stability: Stable. Incompatible with strong oxidizing agents.
Product Categories of Sodium stearate (CAS NO.822-16-2): Anionic Surfactants; Carboxylate (Surfactants); Functional Materials; Surfactants
Appearance: white to off-white solid
Water Solubility of Sodium stearate (CAS NO.822-16-2): Soluble in cold and hot water
Canonical SMILES: CCCCCCCCCCCCCCCCCC(=O)[O-].[Na+]
InChI: InChI=1S/C18H36O2.Na/c1-2-3-4-5-6-7-8-9-10-11-12-13-14-15-16-17-18(19)
20;/h2-17H2,1H3,(H,19,20);/q;+1/p-1
InChIKey: RYYKJJJTJZKILX-UHFFFAOYSA-M
Uses
Sodium stearate works by helping to break fat and oil droplets into small pieces.The hydrocarbon chains attached to the fat, leaving the sodium ends dangling in the water.Then the oils are now completely surrounded by water, and so they wash away in the rinse.
Production
Synthesis:
Sodium stearate can be made in the laboratory by combining stearic acid and glycerol in a heated environment with 2-step reaction :
6 C17H35COOH + 2 C3H5(OH)3 → 2 C3H5(C18H35O2)3 + 3 H2O
C3H5(C18H35O2)3 + 3 NaOH → C3H5(OH)3 + 3 C17H35COONa
Toxicity Data With Reference
Sodium stearate (CAS NO.822-16-2) hasn't been listed as a carcinogen by ACGIH, IARC, NTP, or CA Prop 65. And the toxicological properties have not been fully investigated. You can see actual entry in RTECS for complete information.
Safety Profile
Safety Statements: 24/25
S24/25:Avoid contact with skin and eyes.
WGK Germany: 1
RTECS: WI4725000
Specification
Sodium stearate , its cas register number 822-16-2. It also can be called Octadecanoic acid sodium salt ; Stearic acid sodium salt ; Sodium octadecanoate ; and Stearates . Its classification code is Pharmaceutic aid [emulsifying and stiffening agent].
Sodium stearate (CAS NO.822-16-2) could be stable under normal temperatures and pressures. It should avoid the condition like incompatible materials. It is not compatible with strong oxidizing agents, acids. And also prevent it to broken down into hazardous decomposition products: carbon monoxide, , carbon dioxide, sodium oxide. However, its hazardous polymerization will not occur.
Sodium stearate is the major component of soaps along with many types of solid deodorants, rubbers, latex paints, inks and in accelerators. It is also a component of some food additives and food flavorings.