163619-04-3 Usage
Description
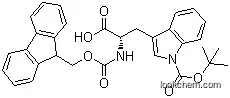
Fmoc-D-Trp(Boc)-OH, also known as N-(9-fluorenylmethoxycarbonyl)-D-tryptophan(Boc), is an amino acid derivative specifically designed for use in peptide chemistry. It is a white powder with unique chemical properties that make it a valuable component in the synthesis of peptides and proteins.
Uses
Used in Pharmaceutical Industry:
Fmoc-D-Trp(Boc)-OH is used as a building block for the synthesis of peptides and proteins due to its unique structure and reactivity. It plays a crucial role in the development of new drugs and therapeutic agents, as well as in the study of protein structure and function.
Used in Research and Development:
In the field of research and development, Fmoc-D-Trp(Boc)-OH is used as a key component in the synthesis of various peptide-based compounds for scientific studies. Its incorporation into peptide sequences allows researchers to investigate the properties and interactions of these molecules, leading to a better understanding of biological processes and the development of novel therapeutic strategies.
Used in Chemical Synthesis:
Fmoc-D-Trp(Boc)-OH is also utilized in chemical synthesis as a protected amino acid derivative. Its protected structure facilitates the selective incorporation of D-tryptophan into peptide chains during solid-phase peptide synthesis, ensuring the desired peptide sequence is obtained with minimal side reactions or racemization.
Overall, Fmoc-D-Trp(Boc)-OH is a versatile and essential compound in the fields of pharmaceuticals, research, and chemical synthesis, contributing to the development of new drugs, therapeutic agents, and a deeper understanding of peptide and protein chemistry.
Check Digit Verification of cas no
The CAS Registry Mumber 163619-04-3 includes 9 digits separated into 3 groups by hyphens. The first part of the number,starting from the left, has 6 digits, 1,6,3,6,1 and 9 respectively; the second part has 2 digits, 0 and 4 respectively.
Calculate Digit Verification of CAS Registry Number 163619-04:
(8*1)+(7*6)+(6*3)+(5*6)+(4*1)+(3*9)+(2*0)+(1*4)=133
133 % 10 = 3
So 163619-04-3 is a valid CAS Registry Number.
InChI:InChI=1/C31H30N2O6/c1-31(2,3)39-30(37)33-17-19(20-10-8-9-15-27(20)33)16-26(28(34)35)32-29(36)38-18-25-23-13-6-4-11-21(23)22-12-5-7-14-24(22)25/h4-15,17,25-26H,16,18H2,1-3H3,(H,32,36)(H,34,35)/t26-/m1/s1
163619-04-3Relevant articles and documents
COMPOUND FOR PREPARATION OF ANTIBODY-PAYLOAD CONJUGATE AND USE THEREOF
-
, (2022/03/15)
The present application relates to a novel linker for use in bioconjugation, comprising two or more electrophilic carbon atoms of a carbonyl group, and a click chemistry functional group and, more specifically, to a linker through which a compound, a peptide, and/or a protein can be directly and/or indirectly linked by a substitution reaction to a desired target molecule, that is, a target molecule.
DIPHENYLMETHANE COMPOUND
-
, (2010/10/19)
Compounds having a diphenylmethane skeleton are superior in broad utility and stability, and are useful as a protecting reagent (anchor) of amino acid and/or peptide in the liquid phase synthesis and the like of a peptide having a C-terminal etc., which are of a carboxamide(-CONHR)-type, and in organic synthetic reaction methods (particularly peptide liquid phase synthetic methods), and may be contained in a kit for peptide liquid phase synthesis.
Controlled drug delivery system using the conjugation of drug to biodegradable polyester
-
, (2008/06/13)
The present invention relates to the molecular sustained controlled release system constructed by the conjugation of molecules to be released with biodegradable polyester polymer via covalent bond and method for preparation thereof. In accordance with the present invention, the system may be formulated into microspheres, nanoparticles, or films. The molecular release rate from the above system can be regulated to be proportional to the chemical degradation rate of the biodegradable polyester polymers, resulting in near zero order kinetics profile of release without showing a burst effect. Moreover, the high loading efficiency of hydrophilic drugs can be achieved.