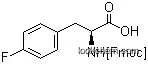
169243-86-1Relevant articles and documents
METHOD FOR PREPARING AROMATIC AMINO ACID DERIVATIVE
-
, (2022/05/13)
The present invention provides methods of efficiently producing various optically active aromatic amino acid derivatives by reacting, using an additive, a specific ester compound with an aromatic halide and zinc in the presence of a catalyst. The present invention also provides amino acid derivatives that can be produced by the methods.
LIGAND-CONTROLLED C(SP3)-H ARYLATION AND OLEFINATION IN SYNTHESIS OF UNNATURAL CHIRAL ALPHA AMINO ACIDS
-
, (2015/10/05)
The use of ligands to tune the reactivity and selectivity of transition metal-catalysts for C(-sp3)-H bond functionalization is a central challenge in synthetic organic chemistry. Herein, we report a rare example of catalyst-controlled C(sp3)-H arylation using pyridine and quinoline derivatives: the former promotes exclusive monoarylation, whereas the latter activates the catalyst further to achieve diarylation. Successive application of these ligands enables the sequential diarylation of a methyl group in an alanine derivative with two different aryl iodides, affording a wide range of β-Ar-p-Ar ' -cc-amino acids with excellent levels of diastereoselectivity (d.r. > 20:1). Both configurations of the β-chiral center can be accessed by choosing the order in which the aryl groups are installed. The use of a quinoline derivative as a ligand also enables C(sp3)-H olefination of a protected alanine.
Solid phase β-lactams synthesis using the Staudinger reaction, monitored by 19F NMR spectroscopy
Le Roy, Isabelle,Mouysset, Dominique,Mignani, Serge,Vuilhorgne, Marc,Stella, Lucien
, p. 3719 - 3727 (2007/10/03)
We report the use of 19F NMR as a simple means to monitor reactions on a solid phase. Multi-step sequences including protection, coupling, deprotection, condensation, cycloaddition and cleavage steps are described in the case of multicomponent reactions involving fluorinated α-aminoesters, aldehydes and acid chlorides.