min at 0 °C, quenched with H2O (30 mL), and extracted with
EtOAc (3x 20 mL). The combined organic extracts were
washed with brine (60 mL), dried over Na2SO4, and
concentrated. The residue was purified by silica gel column
chromatography (EtOAc/hexane 1:7) to give 579 mg of
oxazoline 34 (83%) and 39 mg of epi-34 (6%). Oxazoline 34: a
N-((4S,4aS,7S,7aR)-4-(((Benzyloxy)methoxy)methyl)-2,2,
7-trimethyl-6-oxotetrahydro-4H-furo[3,2-d][1,3]dioxin-7-yl)
-2,2,2-trichloroacetamide (41): A solution of CCl3CO2H (14.4
mg, 88.4 mol), H2O (1.6 L) and CH2Cl2 (4.4 mL) was added
to a solution of hydroxylactone 39 (38.8 mg, 88.4 mol) and
CH2Cl2 (4.4 mL) at room temperature. The solution was
maintained at room temperature for 19 h, and quenched with
Et3N (25 L, 180 mol) at room temperature.
2,2-Dimethoxypropane (430 L, 3.5 mmol) and CSA (41.1 mg,
177 mol) were then added to the solution of the diol at room
temperature. The resulting solution was maintained at room
temperature for 3 d, quenched with saturated aqueous NaHCO3
(5 mL) and extracted with CHCl3 (3x 5 mL). The combined
organic extracts were washed with brine (10 mL), dried over
Na2SO4, and concentrated. The residue was purified by silica
gel column chromatography (EtOAc/hexane 1:9 to 1:7) to give
39.0 mg of cyclic acetal 41 (89%): a colorless oil; []27D +93.5
(c 1.02, CHCl3); IR (film) 3409, 2942, 2888, 1789, 1722, 1512,
1163, 1119, 1045 cm-1; 1H NMR (500 MHz, CDCl3)
7.37–7.28 (m, 5H), 7.08 (s, 1H), 4.81 (s, 2H), 4.78 (d, J = 2.3
Hz, 1H), 4.64 (d, J = 11.8 Hz, 1H), 4.61 (d, J = 11.8 Hz, 1H),
4.40 (dd, J = 2.3, 2.3 Hz, 1H), 4.23 (ddd, J = 6.9, 6.9, 2.3 Hz,
1H), 3.84 (dd, J = 10.0, 6.9 Hz, 1H), 3.78 (dd, J = 10.0, 6.9 Hz,
1H), 1.64 (s, 3H), 1.44 (s, 3H), 1.33 (s, 3H); 13C NMR (125
MHz, CDCl3) 175.6 (C), 161.3 (C), 137.8 (C), 128.6 (CH),
128.00 (CH), 127.98 (CH), 98.8 (C), 95.4 (CH2), 92.0 (C), 71.6
(CH), 71.1 (CH), 70.0 (CH2), 67.4 (CH), 66.8 (CH2), 62.6 (C),
28.9 (CH3), 19.2 (CH3), 17.2 (CH3); HMRS (ESI) calcd for
C20H24Cl3NO7Na+ (M+Na)+ 518.0516, found 518.0511.
colorless oil; []25 +33.4 (c 1.11, CHCl3); IR (film) 2951,
D
1
2886, 1743, 1661, 1258, 1117, 1040, 947, 794 cm-1; H NMR
(500 MHz, CDCl3) 7.40–7.27 (m, 5H), 6.00 (dtd, J = 15.8,
5.2, 0.9 Hz, 1H), 5.74 (ddt, J = 15.8, 7.4, 1.5 Hz, 1H), 5.01 (dd,
J = 7.4, 0.9 Hz, 1H), 4.76 (s, 2H), 4.60 (s, 2H), 4.14 (dd, J =
5.2, 1.5 Hz, 2H), 3.71 (s, 3H), 1.66 (s, 3H); 13C NMR (125
MHz, CDCl3)170.2 (C), 163.4 (C), 137.7 (C), 133.2 (CH),
128.6 (CH), 128.0 (CH), 127.9 (CH), 124.1 (CH), 94.0 (CH2),
92.1 (CH), 86.4 (C), 78.4 (C), 69.6 (CH2), 66.5 (CH2), 52.7
(CH3), 23.9 (CH3); HMRS (ESI) calcd for C18H20Cl3NO5Na+
(M+Na)+ 458.0305, found 458.0321. Oxazoline epi-34: a
colorless oil; []22 +49.4 (c 1.27, CHCl3); IR (film) 2952,
D
1
2888, 1736, 1653, 1243, 1115, 1039, 935, 795 cm-1; H NMR
(500 MHz, CDCl3) 7.39–7.27 (m, 5H), 6.07 (dt, J = 15.5, 5.2
Hz, 1H), 5.82 (ddt, J = 15.5, 7.2, 1.5 Hz, 1H), 5.58 (d, J = 7.2
Hz, 1H), 4.80 (s, 2H), 4.62 (s, 2H), 4.20 (dd, J = 5.2, 1.5 Hz,
2H), 3.83 (s, 3H), 1.43 (s, 3H); 13C NMR (125 MHz, CDCl3)
172.5 (C), 162.6 (C), 137.8 (C), 132.9 (CH), 128.6 (CH),
128.03 (CH), 127.95 (CH), 123.7 (CH), 94.2 (CH2), 88.3 (CH),
86.4 (C), 77.4 (C), 69.7 (CH2), 66.8 (CH2), 53.4 (CH3), 20.3
(CH3); HMRS (ESI) calcd for C18H20Cl3NO5Na+ (M+Na)+
458.0305, found 458.0305.
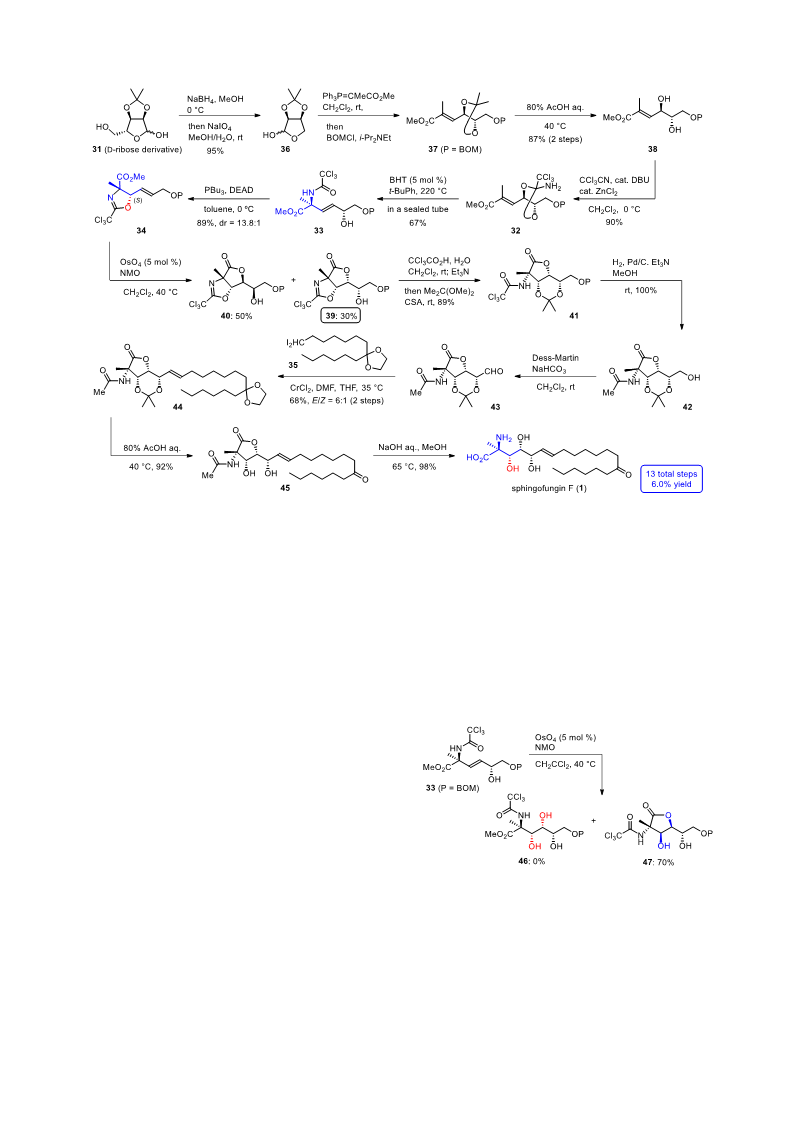
(3aS,6R,6aR)-6-((S)-2-((Benzyloxy)methoxy)-1-hydroxyet
hyl)-3a-methyl-2-(trichloromethyl)-6,6a-dihydrofuro[3,4-d]
oxazol-4(3aH)-one (39): N-Methylmorpholine N-oxide (114
mg, 976 mol) was added to a solution of oxazoline 34 (213
mg, 488 mol), osmium tetroxide (0.1 M in CH2Cl2, 240 L,
24.4 mol) and CH2Cl2 saturated with H2O (16 mL) at 40 °C.
After stirring at 40 °C for 15 h, the reaction was quenched with
saturated aqueous NaHSO3 (15 mL) and extracted with CHCl3
(3x 10 mL). The combined organic extracts were washed with
brine (15 mL), dried over Na2SO4, and concentrated. The
residue was purified by silica gel column chromatography
(EtOAc/hexane 1:4) to give 65.2 mg of hydroxylactone 39
(30%) and 107 mg of hydroxylactone 40 (50%).
N-((4S,4aS,7S,7aR)-4-(Hydroxymethyl)-2,2,7-trimethyl-6-
oxotetrahydro-4H-furo[3,2-d][1,3]dioxin-7-yl)acetamide
(42): Palladium on carbon (10 wt%, 10.2 mg) was added to a
solution of cyclic acetal 41 (5.1 mg, 10.3 mol), Et3N (10 L,
72 mol) and MeOH (1.5 mL). The flask was purged with
hydrogen. The mixture was stirred under hydrogen atmosphere
(1 atm) at room temperature for 2.5 h, filtered through Celite,
washed with MeOH and concetrated. The residue was purified
by silica gel column chromatography (MeOH/CHCl3 1:19) to
give 2.8 mg of primary alcohol 42 (100%): white crystals, mp
224–225 °C; []28 +157.7 (c 1.04, CHCl3); IR (film) 3311,
D
2967, 2896, 1785, 1658, 1539, 1170, 1117, 1057 cm-1; 1H
NMR (500 MHz, CDCl3) 5.91 (s, 1H), 4.82 (d, J = 2.0 Hz,
1H), 4.37 (dd, J = 2.0, 1.7 Hz, 1H), 4.21 (ddd, J = 7.2, 5.5, 1.7
Hz, 1H), 3.92–3.79 (m, 2H), 2.05 (brs, 1H), 2.00 (s, 3H), 1.60
(s, 3H), 1.46 (s, 3H), 1.35 (s, 3H); 13C NMR (125 MHz,
CDCl3) 176.9 (C), 170.1 (C), 98.8 (C), 71.6 (CH), 71.5 (CH),
68.9 (CH), 62.3 (CH2), 61.6 (C), 29.1 (CH3), 23.5 (CH3), 19.2
(CH3), 18.0 (CH3); HMRS (ESI) calcd for C12H19NO6Na+
(M+Na)+ 296.1110, found 296.1104.
Hydroxylactone 39: white crystals, mp 146-147 °C; []23
D
+60.9 (c 0.80, CHCl3); IR (film) 3428, 2927, 2871, 1757, 1644,
1101, 1016, 989, 813 cm-1; 1H NMR (500 MHz, CDCl3)
7.43–7.27 (m, 5H), 5.08 (d, J = 5.7 Hz, 1H), 4.83 (s, 2H),
4.72 (dd, J = 5.7, 5.7 Hz, 1H), 4.67 (d, J = 12.0 Hz, 1H), 4.62
(d, J = 12.0 Hz, 1H), 4.14 (dddd, J = 5.7, 5.2, 4.9, 4.0 Hz, 1H),
3.89 (dd, J = 10.9, 4.9 Hz, 1H), 3.85 (dd, J = 10.9, 4.0 Hz, 1H),
2.67 (d, J = 5.2 Hz, 1H), 1.68 (s, 3H); 13C NMR (125 MHz,
CDCl3) 172.6 (C), 162.9 (C), 137.6 (C), 128.7 (CH), 128.1
(CH), 128.0 (CH), 95.8 (CH2), 87.2 (CH), 85.5 (C), 80.6 (CH),
70.3 (CH2), 69.2 (CH), 69.0 (C), 69.0 (CH2), 20.7 (CH3);
HMRS (ESI) calcd for C17H18Cl3NO6Na+ (M+Na)+ 460.0097,
N-((4S,4aR,7S,7aR)-4-((E)-8-(2-Hexyl-1,3-dioxolan-2-yl)o
ct-1-en-1-yl)-2,2,7-trimethyl-6-oxotetrahydro-4H-furo[3,2-d
][1,3]dioxin-7-yl)acetamide (44): A solution of primary
alcohol 42 (4.2 mg, 15.4 mol) and CH2Cl2 (1.5 mL) was
added to a mixture of Dess-Martin periodinane (32.7 mg, 77.0
mol) and NaHCO3 (32.3 mg, 385 mol) and CH2Cl2 (1.1 mL)
at 0 °C. The mixture was allowed to warm to room temperature
and stirred at room temperature for 2.5 h. The resulting mixture
was filtrated to remove white solid, which was washed with
EtOAc (15 mL). The combined filtrate was then concentrated
to give unstable aldehyde 43, which was used in the next step
without further purification.
found 460.0091. Hydroxylactone 40: a colorless oil; []26
D
+43.1 (c 1.13, CHCl3); IR (film) 3436, 2938, 2886, 1788, 1650,
1282, 1102, 1046, 800 cm-1; 1H NMR (500 MHz, CDCl3)
7.38–7.30 (m, 5H), 5.26 (d, J = 1.2 Hz, 1H), 4.82 (d, J = 6.9
Hz, 1H), 4.77 (d, J = 6.9 Hz, 1H), 4.65 (d, J = 12.0 Hz, 1H),
4.64 (dd, J = 1.4, 1.2 Hz, 1H), 4.62 (d, J = 12.0 Hz, 1H), 4.06
(dddd, J = 9.2, 8.1, 4.0, 1.4 Hz, 1H), 3.87 (dd, J = 10.9, 4.0 Hz,
1H), 3.69 (dd, J = 10.9, 8.1 Hz, 1H), 3.25 (d, J = 9.2 Hz, 1H),
1.71 (s, 3H); 13C NMR (125 MHz, CDCl3) 173.2 (C), 162.9
(C), 137.2 (C), 128.7 (CH), 128.2 (CH), 128.1 (CH), 95.8
(CH2), 89.3 (CH), 85.5 (C), 82.9 (CH), 70.9 (CH), 70.4 (CH2),
70.1 (C), 70.1 (CH2), 19.8 (CH3); HMRS (ESI) calcd for
C17H18Cl3NO6Na+ (M+Na)+ 460.0097, found 460.0096.
In a glove box, CrCl2 (28.4 mg, 231 mol) was dissolved
in THF (1.0 mL) and DMF (18 L, 230 mol). Meanwhile, a
mixture of above aldehyde 43 and diiodide 35 (24.6 mg, 48.4
mol), which was dried by azeotroping with toluene (3x 500
L) beforehand, was dissolved in THF (2.1 mL, dehydrated,

 Sugai, Tomoya
Sugai, Tomoya
 Usui, Shunme
Usui, Shunme
 Tsuzaki, Shun
Tsuzaki, Shun
 Oishi, Hiroki
Oishi, Hiroki
 Yasushima, Daichi
Yasushima, Daichi
 Hisada, Shoko
Hisada, Shoko
 Fukuyasu, Takahiro
Fukuyasu, Takahiro
 Oishi, Takeshi
Oishi, Takeshi
 Sato, Takaaki
Sato, Takaaki
 Chida, Noritaka
Chida, Noritaka