Organic Letters
Letter
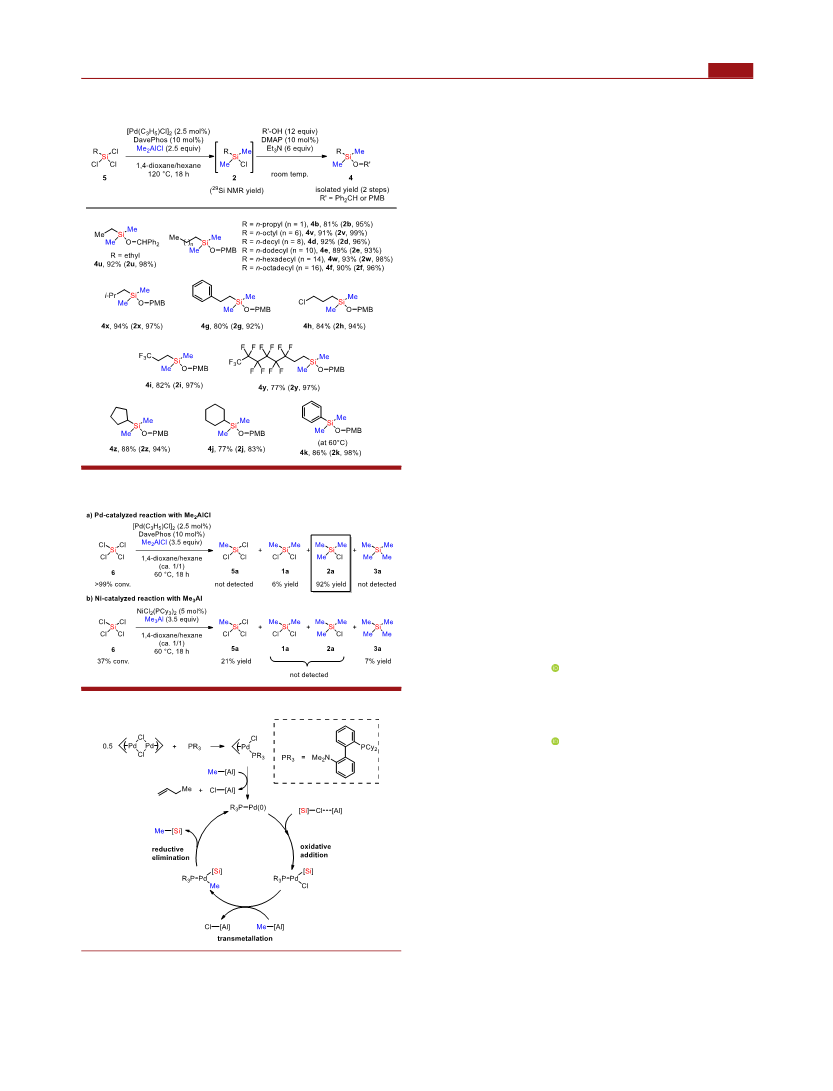
methyldichlorosilanes, the reactions of dichloromethylsilanes
of 2p was previously reported by a 3-step synthesis starting
from 1c, in 42% overall yield (Scheme 3b).
18
1
b−f bearing different n-alkyl chains were initially carried out
to examine the size influence of the alkyl chains. Regardless of
the carbon numbers, methylations of 1b−f resulted in the
exclusive formation of the corresponding monochlorosilanes
Scheme 3. (a) Gram-Scale Synthesis of 2p Utilizing
Selective Monomethylation of 1p with Me AlCl; (b)
2
2b−f with 95−99% yields. After alcoholysis workup, dimethyl-
Reported Synthesis of 2p in 3 Steps
monoalkoxysilanes 4b−f could be isolated in 87−91% yields in
two steps. Considering the functional group tolerance, the
reactions with dichloromethylsilanes 1g−i with 2-phenethyl, 3-
chloropropyl and 3,3,3-trifluoropropyl groups uniformly
proceeded to give the products 2g−i (95−96% yields) and
4
1
g−i (82−86% yields). The reaction of dichloromethylsilane
j having a cyclohexyl group quantitatively gave 2j (99% yield)
and 4j (91% yield). We then subjected dichloromethyl-
phenylsilane 1k to the present catalytic system, even though
aryl-substituted dichlorosilanes were already identified as
13
suitable substrates in the previous nickel catalysis. At the
standard reaction temperature (120 °C), 1k was converted to
2
k (89% yield) in conjunction with 3k (11% yield). However,
Not limited to the methylation reaction, we were able to
(SI)).
To further expand the utility of the Pd-catalyzed selective
methylation of chlorosilanes, modification of the reaction
conditions for dichlorosilanes was performed in the model
we could completely control the selectivity at a lower reaction
temperature (60 °C) to obtain 2k (99% yield) and 4k (85%
yield), respectively. Since alkenylsilanes are useful synthetic
intermediates and readily accessed via the silyl-Heck
9
,12
reaction,
the reaction of dichlorostyrylmethylsilane 1l was
performed to provide the corresponding product 2l (78%
yield). Since the corresponding p-methoxybenzyloxysilane was
slightly decomposed during the isolation process, we
determined the isolated yield as 63% after transformation to
the more stable diphenylmethoxysilane 4l. We next inves-
tigated the scope of dichlorosilanes with two identical alkyl
chains. The methylation of 1m−r with various n-alkyl chains
and a branched 2-ethylhexyl group gave 2m−r (78−99%
yields) and 4m−r (77−96% yields). In contrast, the reaction of
dichlorosilanes 1s and 1t having bulky secondary and tertiary
alkyl groups led to poorer reactivity (32% conversion for 1s
and almost no reaction for 1t) due to the steric hindrance. As
shown in Table 2, no substrates gave detectable dimethylated
products.
reaction using MeSiCl (5a) (Table 2). The reaction of 5a
3
with 1.5 equiv of Me AlCl under the same conditions for 1a
2
led to the formation of 1a and 2a in a ratio of 39:61 without
the detection of Me Si 3a (entry 1). However, switching the
4
product selectivity between mono- and dimethylation was
enabled by changing the amount of Me
reaction with 0.8 equiv of Me AlCl gave 1a as a major product
in 92% yield (entry 2), while the reaction with 2.5 equiv of
Me AlCl at an elevated temperature gave 2a as a major product
in 95% yield (entry 3). The byproduct 3a was not detected
AlCl. Namely, the
2
2
2
19
even at 120 °C (entry 4). In contrast, the reaction without
any catalysts and nickel-catalyzed reaction of 5a failed (entries
5 and 6).
With the optimized conditions for dimethylation of 5a in
hand, we investigated the scope of substrates (Scheme 4). A
variety of alkyltrichlorosilanes 5 with n-alkyl groups, primary
alkyl groups, halogenated alkyl groups, secondary alkyl groups,
and a phenyl group were all tolerated to give the corresponding
products 2 (83−99% yields) and 4 (77−94% yields),
respectively. The selective introduction of other alkyl chains
into 5a with trialkylaluminum reagents were also demonstrated
We also demonstrated a gram-scale synthesis starting from
dichlorodihexylsilane (1p) (40 mmol) to furnish the target
monochlorosilane 2p in 78% isolated yield (7.79 g) after
purification by simple distillation (Scheme 3a). A preparation
a
Methylation of 5a
(
In the category of chlorosilanes, considerable amounts of
tetrachlorosilane (6) are inexpensively produced as a by-
product of the polysilicon industry or from the chlorination of
20
b
b
ferrosilicon with chlorine. Therefore, 6 is obviously the most
basic and attractive starting material to synthesize various
organosilicon compounds. However, to the best of our
knowledge, there have been no satisfactory examples for
catalytic conversion of 6 into organosilicon compounds. The
present palladium catalyst could be applied to 6 to achieve
selective triple methylation, furnishing 2a in 92% NMR yield
entry
x (equiv)
1.5
0.8
2.5
2.5
2.5
T (°C) % conv (5a)
% yield (1a/2a/3a)
1
2
3
4
60
60
90
120
90
90
>99
>99
>99
>99
<5
39/61/n.d.
92/8/n.d.
n.d./95/n.d.
n.d./86/n.d.
n.d./n.d./n.d.
n.d./6/32
c
5
d
6
2.5 (Me Al)
41
3
(
Scheme 5a). In contrast, the nickel-catalyzed reaction of 6 led
to the formation of 5a and 3a with poor selectivity (Scheme
b).
A possible reaction mechanism for the palladium-catalyzed
a
Reaction conditions: [Pd(C H )Cl] (2.5 mol %), DavePhos (10
mol %), 5a (0.5 mmol), Me AlCl in dioxane, 18 h. Determined by
Si{ H} NMR spectroscopic analysis with trimethyl(phenyl)silane
0.5 mmol) as an internal standard. Without catalyst. NiCl (PCy )
3
5
2
b
5
2
2
9
1
c
d
(
cross-coupling of chlorosilanes and methylaluminum species is
shown in Scheme 6. The initial step is the generation of the
2
3 2
was used as catalyst.
6
03
Org. Lett. 2021, 23, 601−606

 Naganawa, Yuki
Naganawa, Yuki
 Nakajima, Yumiko
Nakajima, Yumiko
 Sakamoto, Kei
Sakamoto, Kei