Organic Process Research & Development 2007, 11, 237−240
Development of a Concise Scaleable Synthesis of 2-Chloro-5-(pyridin-2-yl)
Pyrimidine via a Negishi Cross-Coupling
Carlos Pe´rez-Balado,† Albert Willemsens,* Dominic Ormerod, Wim Aelterman, and Narda Mertens
Chemical DeVelopment, Johnson & Johnson Pharmaceutical Research & DeVelopment, Turnhoutseweg 30,
2340 Beerse, Belgium
Abstract:
A practical and scaleable synthesis of 2-chloro-5-(pyridin-2-yl)
pyrimidine, an intermediate in the synthesis of a selective
PDE-V inhibitor, was developed. A Negishi cross-coupling
between the in situ prepared 2-pyridylzinc chloride and 5-iodo-
2-chloropyrimidine catalyzed by Pd(PPh3)4 afforded the product
in one step. Development of a convenient purification did away
with the necessity of chromatography, allowing the preparation
Figure 1. Cross-coupling approach.
of the product on kilogram scale.
Kumada,5 and Negishi6 couplings. Due to the toxicity of tin,
the Stille coupling was not considered as a valuable alterna-
Introduction
tive. By far, the preferred cross-coupling in process develop-
ment is the Suzuki-Miyaura coupling, since the boronic
acids are thermally stable compounds and the reaction does
not require anhydrous conditions. However, there are only
few examples of the scaling-up of the Kumada7 or Negishi8
cross-couplings.
The pyridine fragment was selected for the preparation
of the organometallic partner. 5-Bromo-2-chloropyrimidine,
6, and 5-iodo-2-chloropyrimidine, 8, were prepared from the
2-hydroxypyrimidine hydrochloride in two steps (Scheme
2). In the case of the bromo derivative, the bromination was
carried out with Br2 in concentrated aqueous HCl to afford
5-bromo-2-hydroxypyrimidine, 5, subsequent chlorination
with POCl3 in the presence of N,N-dimethylaniline gave 6
in a 65% overall yield. The iodo derivative was prepared by
iodination of 4 with ICl in H2O-AcOH, followed by
chlorination with POCl3. With acetonitrile as cosolvent only
1 equiv of POCl3 was needed for the chlorination, making
the workup easier. Following this procedure 8 was prepared
in multikilogram scale.9
During our program for the development and scaling up
of a selective PDE-V inhibitor, a short and efficient synthesis
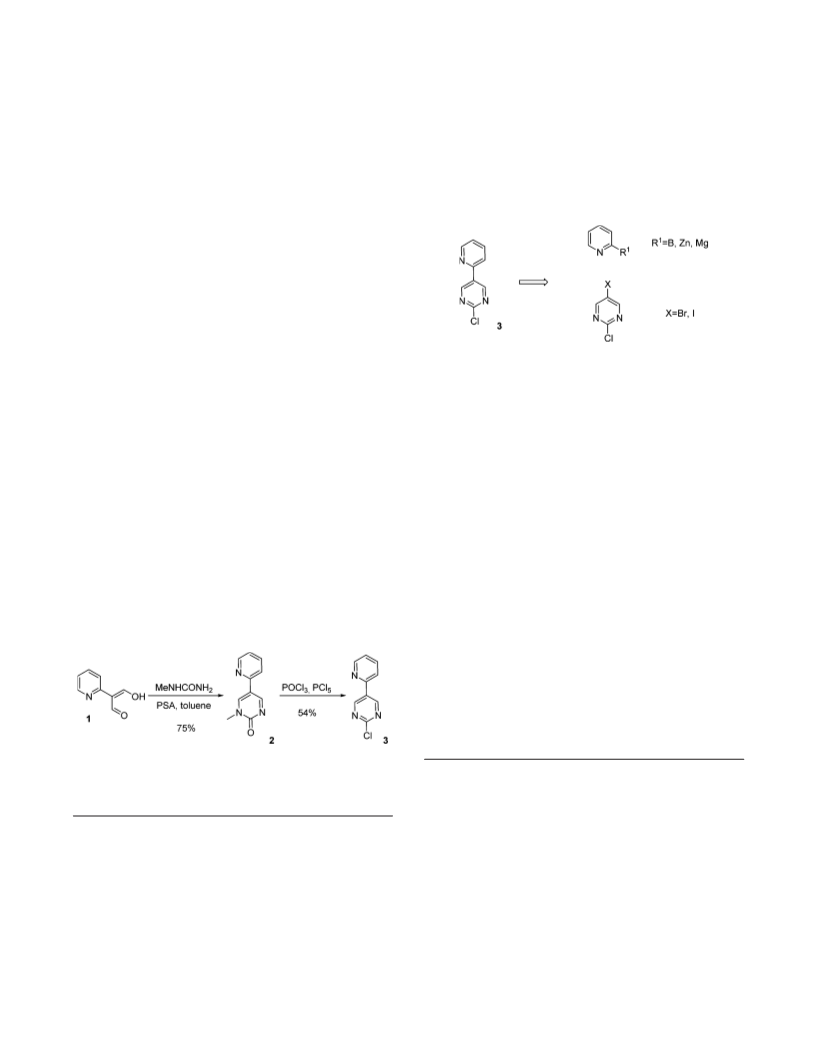
of the 2-chloro-5-(pyridin-2-yl) pyrimidine, 3, was required.
This was a key intermediate in the last step of our convergent
synthetic strategy.1 The medicinal chemistry route involved
the condensation of the 2-pyridyl malonaldehyde 1 with
methylurea followed by demethylation/chlorination with a
mixture of POCl3 and PCl5 (Scheme 1) to give 3 in a overall
40% yield.2 This synthetic route proved unsuitable for
scaling-up due to the difficulties to prepare 1 in bulk
quantities. Therefore, it was decided to explore a synthetic
approach using a cross-coupling between the two heterocyclic
rings (Figure 1).
Scheme 1. Medicinal chemistry route
In order to perform the Suzuki-Miyaura coupling, we
tried the synthesis of the 2-pyridylboronic acid. Unfortu-
Results and Discussion
Classical and well-established cross-coupling methods
mediated by Pd or Ni include the Stille,3 Suzuki-Miyaura,4
(5) Tamao, K.; Sumitani, K.; Kumada, M. J. Am. Chem. Soc. 1972, 94, 4374.
(6) Negishi, E. I.; Hu, Q.; Huang, Z.; Qian, M.; Wang, G. Aldrichimica Acta.
2005, 38, 71
(7) (a) Ikunaka, M.; Maruoka, K.; Okuda, Y.; Ooi, T. Org. Process Res. DeV.
2003, 7, 644. (b) Banno, T.; Hayakawa, Y.; Umeno, M. J. Organomet. Chem.
2002, 653, 288.
(8) (a) Denni-Dischert, D.; Marterer, W.; Baenziger, M.; Yusuff, N.; Batt, D.;
Ramsey, T.; Geng, P.; Michael, W.; Wang, R.-M. B.; Taplin, F., Jr.; Versace,
R.; Cesarz, D.; Pe´rez, L. B. Org. Process Res. DeV. 2006, 10, 70. (b) Cai,
S.; Dimitroff, S. M.; McKennon, T.; Reider, M.; Robarge, L.; Ryckman,
D.; Shang, X.; Therrien, J. Org. Process Res. DeV. 2004, 8, 353. (c) Ragan,
J. A.; Raggon, J. W.; Hill, P. D.; Jones, B. P.; McDermott, R. E.; Munchhof,
M. J.; Marx, M. A.; Casavant, J. M.; Cooper, B. A.; Doty, J. L.; Lu, Y.
Org. Process Res. DeV. 2003, 7, 676. (d) Manley, P. W.; Acemoglu, M.;
Marterer, W.; Pachinger, W. Org. Process Res. DeV. 2003, 7, 436.
(9) Pyrimidine 8 has been previously described: (a) Brown, D. J.; Arantz, B.
W. J. Chem. Soc. (C) 1971, 10, 1889. (b) von Arenger, S. In Science of
Synthesis; Yamamoto, Y., Ed.; Georg Thieme Verlag: Stuttgart, 2004; Vol.
16, p 379.
† Current address: Department of Organic Chemistry, Universidad de Vigo,
(1) Willemsens, B.; Vervest, I.; Ormerod, D.; Aelterman, W.; Fannes, C.;
Mertens, N.; Marko´, I. E.; Lemaire, S. Org. Process Res. DeV. 2006, 10,
1275.
(2) (a) Jiang, W.; Guan, J.; Macielag, M. J.; Zhang, S.; Qiu, Y.; Kraft, P.;
Bhattacharjee, S.; John, T. M.; Haynes-Johnson, D.; Lundeen, S.; Sui, Z. J.
Med. Chem. 2005, 48, 2126. (b) Sui, Z.; Macielag, M. J.; Guan, J; Jiang,
W; Lanter, J. C. PCT Int. Appl. WO 2001087882, 2001.
(3) de Meijere, A., Diederich, F., Eds. Metal-Catalysed Cross-Coupling
Reactions, 2nd ed.; Wiley-VCH: Weinheim, 2004.
(4) (a) Horn, K. A. Chem. ReV. 1995, 95, 1317. (b) Marshall, J. A. Chem. ReV.
2000, 100, 3163.
10.1021/op060241c CCC: $37.00 © 2007 American Chemical Society
Published on Web 02/17/2007
Vol. 11, No. 2, 2007 / Organic Process Research & Development
•
237

 Perez-Balado, Carlos
Perez-Balado, Carlos
 Willemsens, Albert
Willemsens, Albert
 Ormerod, Dominic
Ormerod, Dominic
 Aelterman, Wim
Aelterman, Wim
 Mertens, Narda
Mertens, Narda