DOI: 10.1002/cctc.201900367
Full Papers
1
2
3
4
5
6
7
8
9
0
1
2
3
4
5
6
7
8
9
0
1
2
3
4
5
6
7
8
9
0
1
2
3
4
5
6
7
8
9
0
1
2
3
4
5
6
7
8
9
0
1
2
3
4
5
6
7
A Reusable Cobalt Catalyst for Reversible Acceptorless
[b]
[a]
[b]
[a]
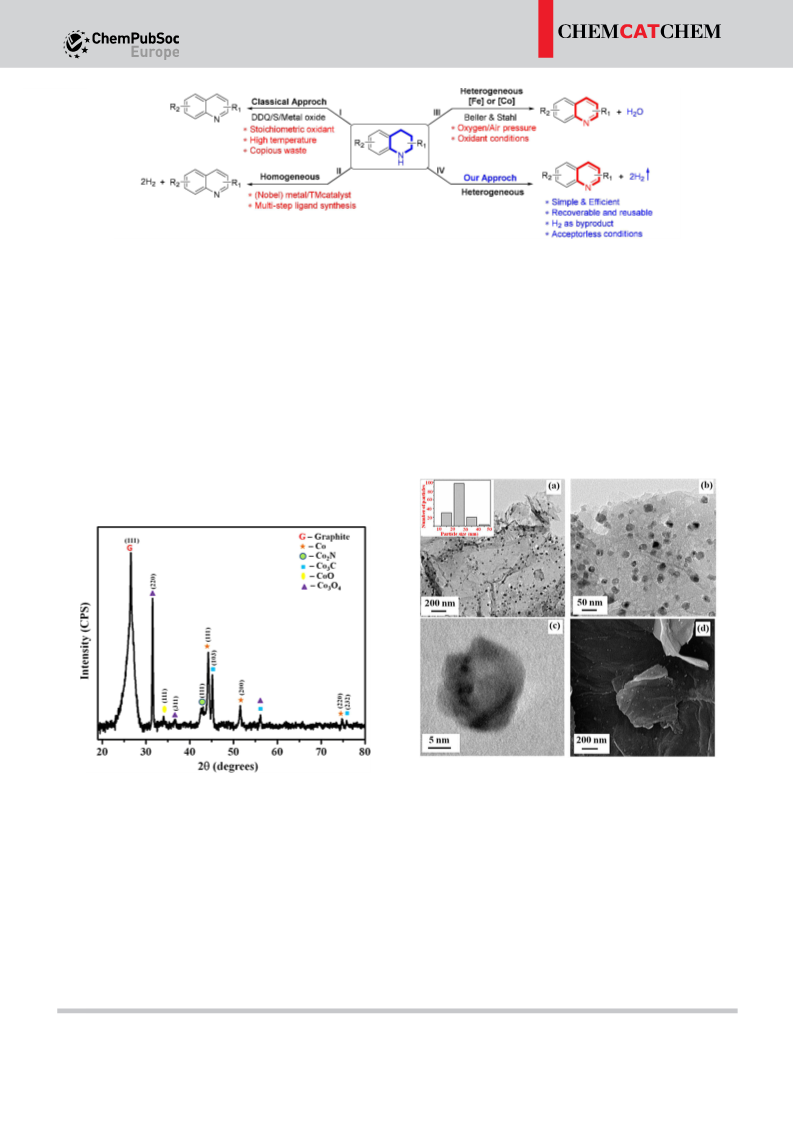
The development of robust catalytic systems based on base-
metals for reversible acceptorless dehydrogenation (ADH) and
hydrogenation of feedstock chemicals is very important in the
context of ‘hydrogen storage’. Herein, we report a highly
efficient reusable cobalt-based heterogeneous catalyst for
reversible dehydrogenation and hydrogenation of N-hetero-
cycles. Both the ADH and the hydrogenation processes operate
under mild, benign conditions.
1
1
1
1
1
1
1
1
1
1
2
2
2
2
2
2
2
2
2
2
3
3
3
3
3
3
3
3
3
3
4
4
4
4
4
4
4
4
4
4
5
5
5
5
5
5
5
5
Introduction
Indeed, it is highly challenging and demanding to develop an
efficient and robust, reusable catalytic system to explore its
catalytic performance for the ADH of N-heterocycles.
Significant contributions have been made in the transition-
metal catalyzed ADH reactions of N-heterocyclic compounds by
[17]
N-heteroaromatics are extensively used in the synthesis of
natural products, bioactive molecules, and pharmaceuticals.
[
1]
Besides, these molecules show profound applications in materi-
al science and as Liquid Organic Hydrogen Carriers (LOHCs) in
various research groups using well-defined metal complexes
[2]
[18–19]
fuel cells. Owing to this, a sustainable catalytic approach for
their synthesis is highly demanding (Scheme 1). Hence, catalytic
dehydrogenation reaction of partially saturated N-heterocycles
to N-heteroaromatics is highly desirable and paid much
under homogeneous conditions.
Despite all, the use of
precious metal complexes with multistep synthesized ligands,
and difficulties in reuse of the soluble catalysts are the major
concerns. Alternatively, heterogeneous catalysis could be a
better option over homogeneous catalysis. The significant
advantage of heterogeneous catalyst is its capability for easy
separation from the reaction mixture and durability of recycling
for several runs. Notably, several heterogeneous catalytic
systems have been well-documented for the oxidative dehydro-
genation of N-heterocycles using air or oxygen as a sole
[3–5]
attention in contemporary science.
Conventionally, dehydro-
genation reactions were performed using the stoichiometric
amount of strong oxidants such as DDQ, peroxides, iodates,
chromium (IV) reagents and metal oxides, which often produce
excess hazardous waste. An alternative to these toxic oxidants,
pressurized air or oxygen was used as the sole oxidant.
[6]
[
7–12]
[20]
Catalytic dehydrogenation can also be performed in presence
of hydrogen acceptors such as sterically hindered alkenes, and
oxidant.
Indeed, a robust, reusable catalyst for the ADH
reaction of N-heterocycles remains an essential goal in chemical
research. In a continuous effort to discover first-row transition
[13]
ketones. The overall process is redox neutral and does not
involve net hydrogen evolution; however, the byproduct is the
stoichiometric amount of sacrificial organic waste. In this
context, catalytic acceptorless dehydrogenation reaction with
the liberation of hydrogen gas is the preferred and promising
route for many synthetic transformations and indeed hydrogen
is expected to play a key role as an energy carrier in the
[21]
metal catalysts for sustainable catalysis, we have focused our
[22]
attention on heterogeneous cobalt catalyst. Notably, a semi-
nal work reported by the research group of Beller and Stahl for
the oxidative dehydrogenation of N-heterocycles to N-aromatics
[12a–b]
using air/oxygen as the oxidant.
It is noteworthy that the
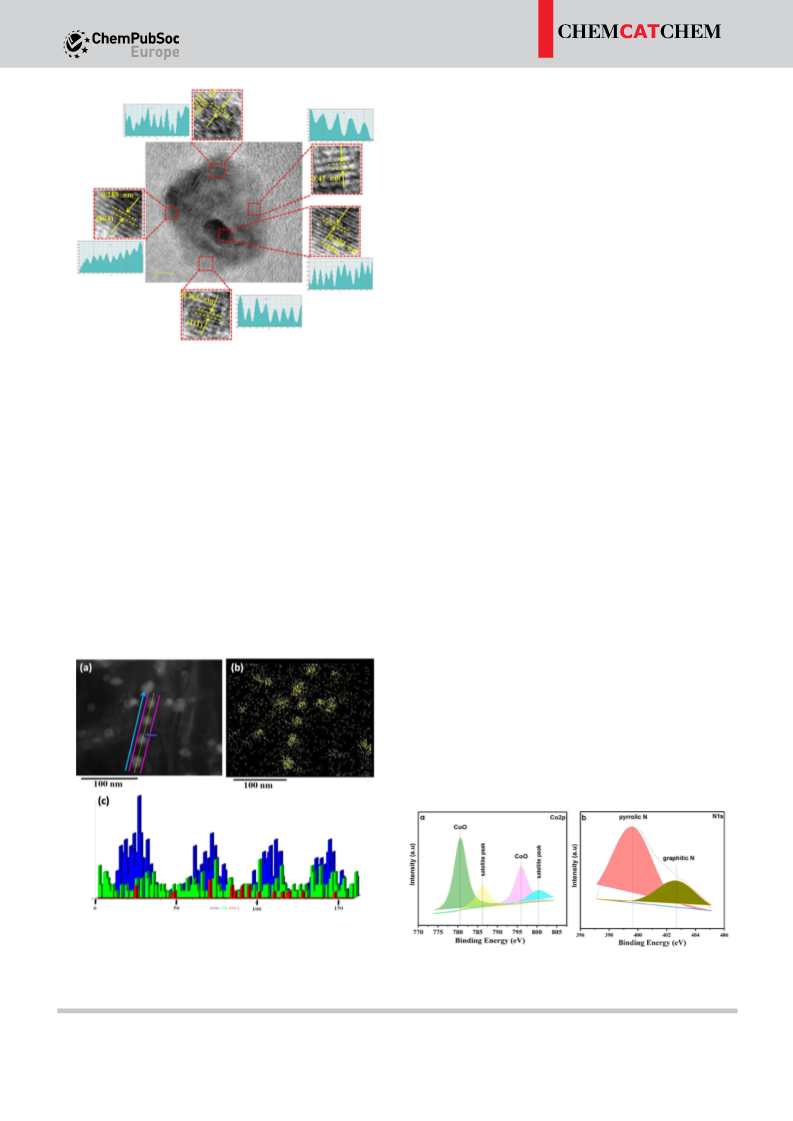
activity of non-noble metal nanoparticles can be controlled by
M:L composition, nature of carbon support, pyrolysis temper-
[14–15]
future.
[
23]
Notably, removal of hydrogen atoms from adjacent atomic
centers of a saturated organic molecule is highly challenging
and thermodynamically uphill process. Recent DFT calculations
and experimental results showed that the presence of a
nitrogen atom in the cyclic system makes the dehydrogenation
ature and finally the microstructure of the nanomaterial.
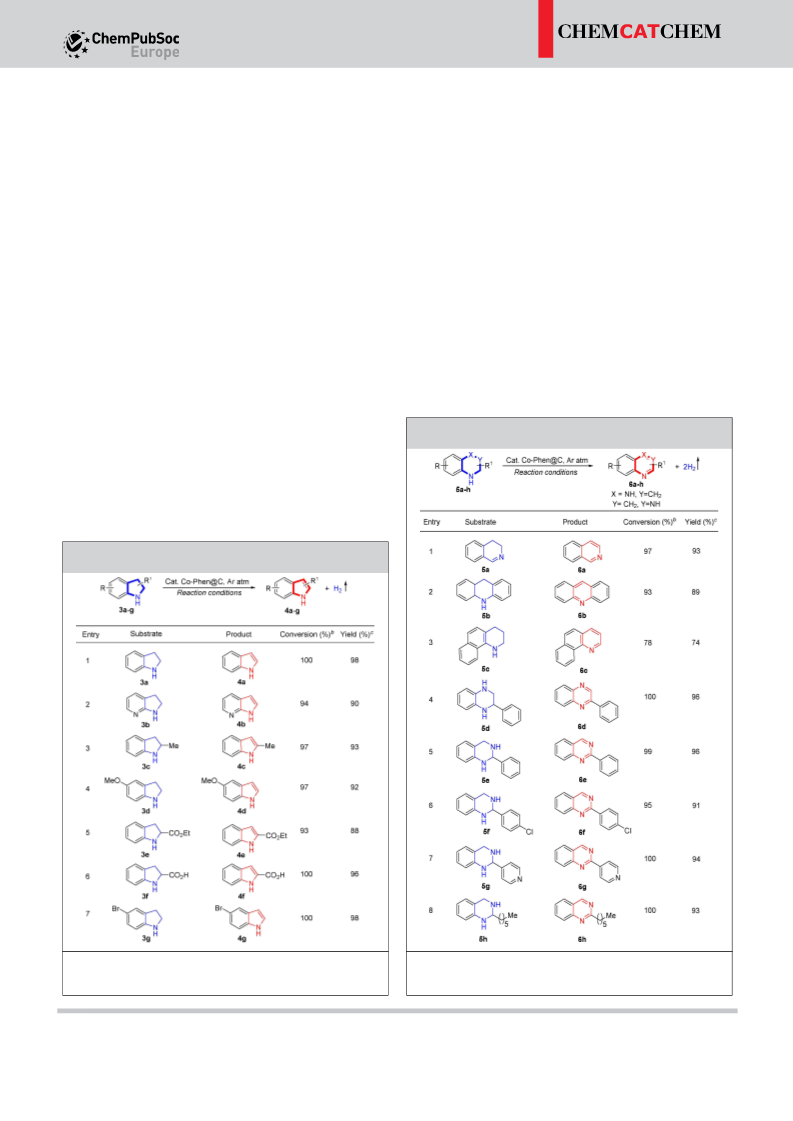
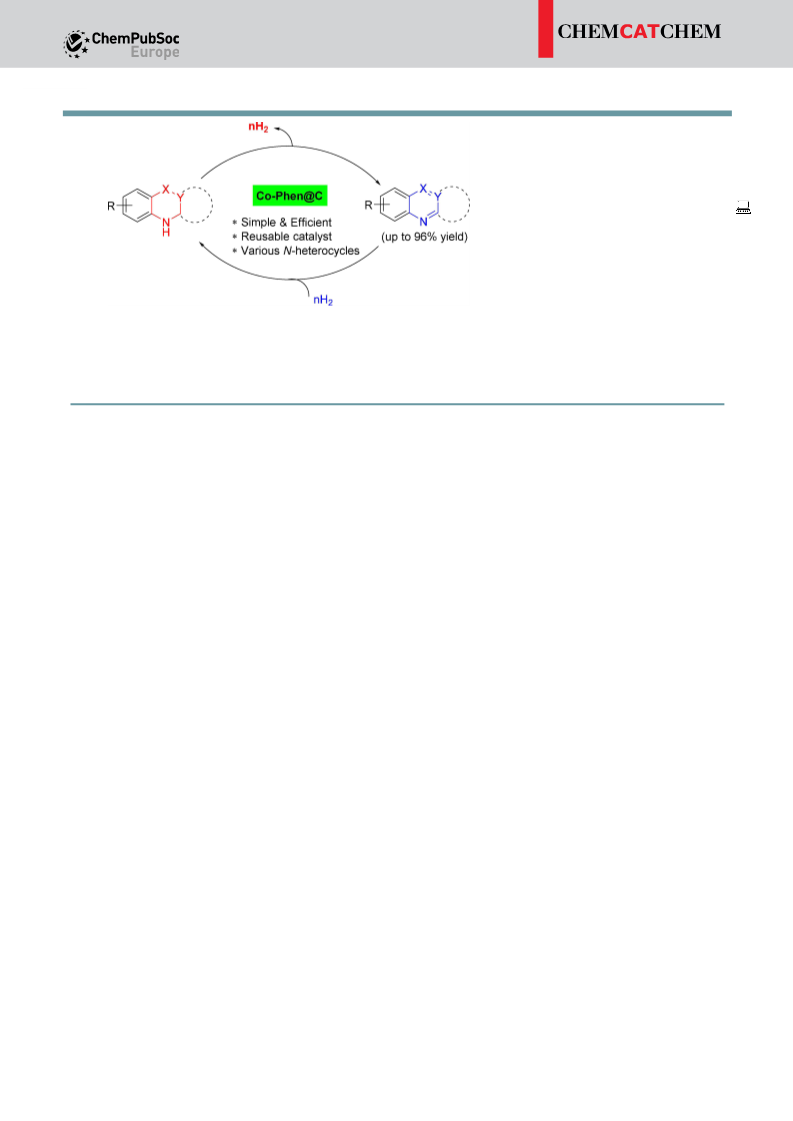
Gratifyingly, the newly synthesized cobalt nanocatalyst showed
superior activity in the ADH reaction of partially saturated N-
heterocycles to N-heteroaromatics with the liberation of H gas.
2
The present nano-catalyst performs excellently with a five-run
recycling test. In the context of ‘hydrogen storage’, the micro-
scopic reversible hydrogenation reaction is also equally impor-
tant. Despite notable advancement in the development of
catalytic ADH and hydrogenation of N-heterocycles, systems
that utilize a single transition-metal catalyst for both processes
[16]
process more accessible by lowering the activation energy.
[
a] M. Subaramanian, Prof. E. Balaraman
Department of Chemistry
Indian Institute of Science Education and Research Tirupati (IISER-Tirupati)
Tirupati – 517507 (India)
E-mail: eb.raman@iisertirupati.ac.in
[17b,18a,19a,d–e,24–25]
are rarely studied.
We have also successfully
[
b] G. Jaiswal, M. K. Sahoo
Organic Chemistry Division
CSIR-National Chemical Laboratory (CSIR-NCL)
Dr. Homi Bhabha Road, Pune – 411008 (India)
demonstrated the hydrogenation of N-heteroarenes to the
corresponding N-heterocycles catalysed by the same cobalt
nano-catalyst.
ChemCatChem 2019, 11, 1–10
1
© 2019 Wiley-VCH Verlag GmbH & Co. KGaA, Weinheim
��
These are not the final page numbers!

 Jaiswal, Garima
Jaiswal, Garima
 Subaramanian, Murugan
Subaramanian, Murugan
 Sahoo, Manoj K.
Sahoo, Manoj K.
 Balaraman, Ekambaram
Balaraman, Ekambaram