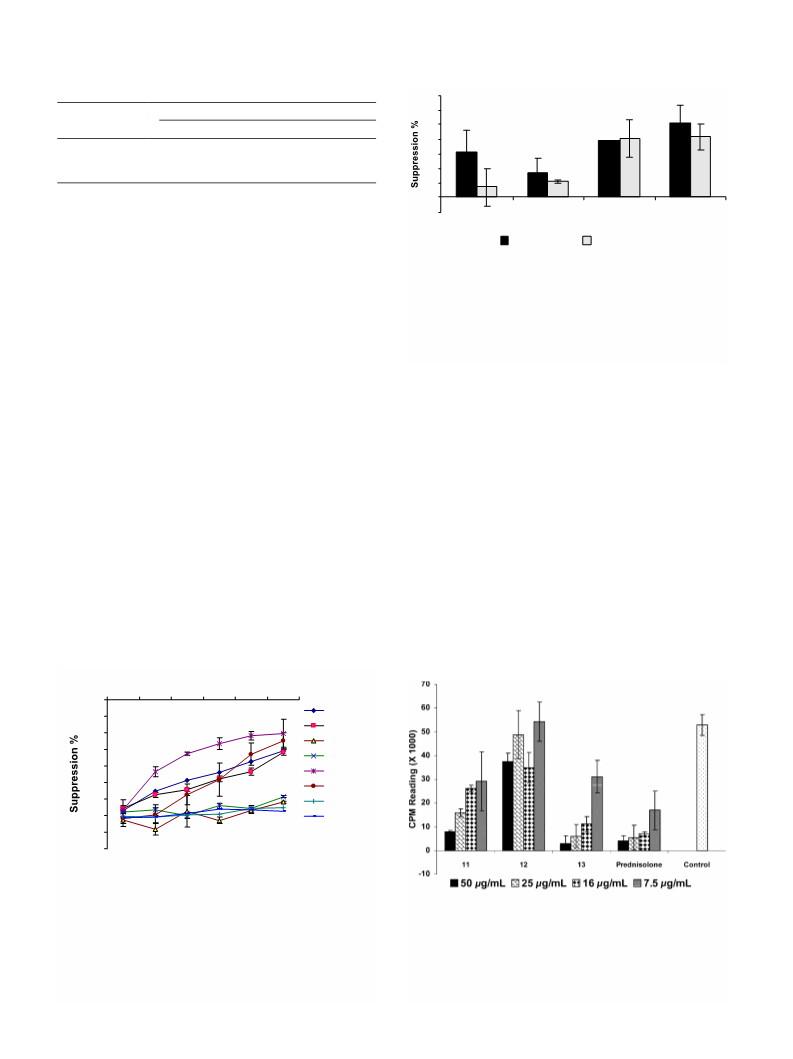
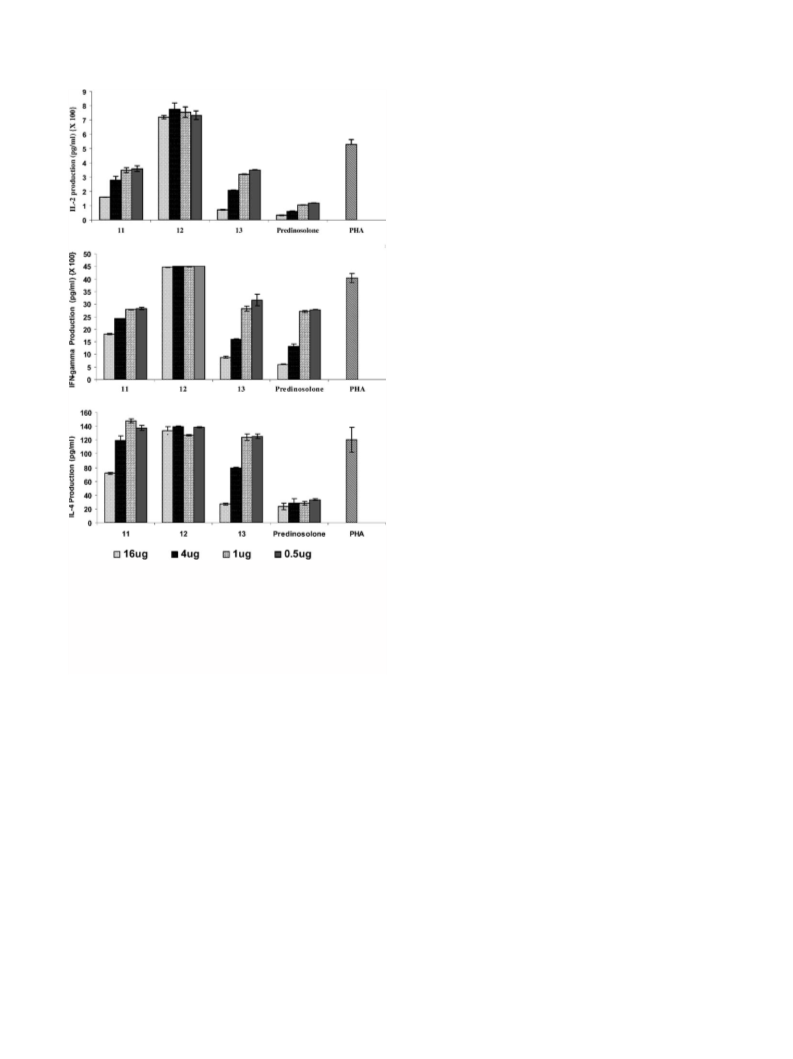
2054
M. A. Mesaik et al. / Bioorg. Med. Chem. 12 (2004) 2049–2057
3.92 (s, 3H, OCH3), 3.87 (s, 3H, OCH3), 7.19 (br s, 1H,
H-6), 6.91 (s, 1H, H-60), 7.40 (t, 1H, J ¼ 7:2 Hz, H-400),
7.50 (d, 1H, J ¼ 7:5 Hz, H-60), 7.57 (d, 1H, J ¼ 7:2 Hz,
H-50), 8.10 (d, 2H, J ¼ 6:9 Hz, H-200,600), 8.15 (dd, 2H,
J ¼ 1:8, J ¼ 6:9 Hz, H-300,500); IR (KBr): 1783, 1771,
1666, 1497, 1462, 1329, 1286, 1160, 1112, 1013, 994, 917,
889, 867 cmꢀ1; UV kmax (CHCl3): 396, 260, 201 nm; EI-
MS m=z (rel abund %): 309 (Mþ, 43), 176 (6) 131 (2), 105
(100), 51 (5). Anal. Calcd for C18H15NO4: C, 69.89; H,
4.89; N, 4.52. Found C, 69.87; H, 4.88; N, 4.50.
1641, 1563, 1486, 1388, 1268, 1184, 1066, 982, 743 cmꢀ1
;
UV kmax (CHCl3): 378, 372, 359, 259, 255, 251, 243, 230,
203, 198 nm; EI-MS m=z (rel abund %): 283 (Mþ, 63),
141 (9), 104 (100), 51 (39). Anal. Calcd for C16H10NO2: C,
67.74; H, 3.55; N, 4.94. Found: C, 67.73; H, 3.54; N, 4.96.
6.1.7.
4-[(E)-3-Methylbutylidene]-2-phenyl-1,3-oxazol-
5(4H)-one (7). Isovaleraldehyde (1.63 mL, 15.3 mmol),
N-benzoylglycine (2.5 g, 13.96 mmol), acetic anhydride
(3.93 mL, 36.3 mmol) and fused sodium acetate (1.14 g,
13.9 mmol) were treated in the same way as described
for compound 3. The orange yellow product obtained
was 2.0 g (63%). Mp: 136 ꢁC; Rf : 0.56 (n-butanol–acetic
6.1.4. 4-[(E)-(3-Nitrophenyl)methylidene]-2-phenyl-1,3-
oxazol-5(4H)-one (4). The compound 4 was prepared
in the same way as described for compound 3 from
3-nitro benzaldehyde (1.20 g, 7.94 mmol). The product
was obtained as a light orange solid 1.55 g (66%). Mp:
1
acid–H2O 3:3:1); H NMR (300 MHz, CDCl3) d: 1.50
(d, 6H, J ¼ 7:5 Hz, (CH3)2), 2.95 (m, 1H, CH), 7.24 (s,
1H, H-6), 7.35 (d, 2H, J ¼ 7:9 Hz, H-30,50), 7.46 (t, 1H,
J ¼ 7:6 Hz, H-40), 7.54 (d, 2H, J ¼ 7:9 Hz, H-20,60); UV
kmax (CHCl3): 382, 378, 274, 237, 202, 194 nm; EI-MS
m=z (rel abund %): 229 (Mþ, 45), 105 (100), 77 (45).
Anal. Calcd for C14H15NO2: C, 73.34; H, 6.60; N, 6.10.
Found: C, 73.33; H, 6.59; N, 6.11.
1
162 ꢁC; Rf : 0.64 (n-butanol–acetic acid–H2O 3:3:1); H
NMR (300 MHz, CDCl3) d: 7.24 (br s, 1H, H-6), 7.54 (t,
1H, J ¼ 8:2 Hz, H-30), 7.59–7.65 (m, 1H, H-400, Ar-H),
7.67 (dd, 2H, J ¼ 3:8, J ¼ 8:0 Hz, H-300,500), 8.18 (dd,
2H, J ¼ 1:3, J ¼ 8:0 Hz, H-200,600), 8.28 (dd, 1H, J ¼ 1:3,
J ¼ 8:2 Hz, H-200), 8.40 (d, 1H, J ¼ 8:2 Hz, H-40), 9.30
(s, 1H, H-60, Ar-H); IR (KBr): 1788, 1657, 158, 1349,
1294, 1163, 994, 889 cmꢀ1; UV kmax (CHCl3): 3576, 268,
225 nm; EI-MS m=z (rel abund %): 294 (Mþ, 46), 105
(100), 77 (50). Anal. Calcd for C16H10N2O4: C, 65.30; H,
3.43; N, 9.52. Found: C, 65.28; H, 3.42; N, 9.58.
6.1.8. 4-[(E)-Pyrrol-3-ylmethylidene]-2-phenyl-1,3-oxa-
zol-5(4H)-one (8). Compound 8 was prepared in the
same way as described for compound 3, from pyrrole
carbaldehyde (1.5 g, 15.77 mmol). The product was
obtained as yellow solid 2.26 g (60%); Rf : 0.66 (n-butanol–
1
acetic acid–H2O 3:3:1); H NMR (300 MHz, CDCl3) d:
7.19 (br s, 1H, H-6), 7.3–7.8 (m, 3H, H-20,30,40), 8.5–8.8
(m, 5H, H-200,300,400,500,600, Ar-H); FD MS m=z: 238 (Mþ);
IR (KBr): 1795, 1775, 1665; 1555, 1515, 1480, 1460,
1410, 1380, 1360, 1315, 1245, 1125, 1095, 965, 875, 865,
765 cmꢀ1; UV kmax (CHCl3): 374, 273, 227, 262, 262 nm.
Anal. Calcd for C14H10N2O2: C, 70.58; H, 4.23; N,
11.76. Found: C, 70.55; H, 4.22; N, 11.77.
6.1.5.
4-[(E)-1H-Indol-3-ylmethylidene]-2-phenyl-1,3-
oxazol-5(4H)-one (5). Indole-3-carbaldehyde (1.50 g,
10.34 mmol), N-benzoylglycine (2.5 g, 13.97 mmol),
acetic anhydride (3.75 mL) and fused sodium acetate
were treated in the same way as described for compound
3. The product was obtained as yellow solid 1.81 g
(61%). Mp: 206 ꢁC; Rf : 0.65 (n-butanol–acetic acid–H2O
3:3:1); 1H NMR (300 MHz, CDCl3) d: 7.49 (s, 1H, H-6),
7.42 (dd, 2H, J ¼ 1:7, J ¼ 7:4 Hz, H-300,500), 7.54 (t, 2H,
J ¼ 7:4 Hz, H-200,600), 7.62 (t, 1H, J ¼ 7:4 Hz, H-400), 7.84
(dd, 1H, J ¼ 6:8, J ¼ 7:8 Hz, H-50), 8.13 (d, 2H,
J ¼ 7:2 Hz, H-2, H-60,70), 8.47 (d, 1H, J ¼ 7:4 Hz, H-80),
8.75 (s, 1H, CH); IR (KBr): 1781, 1646, 1581, 1339,
1291, 1153, 987, 875 cmꢀ1; UV kmax (CHCl3): 400, 377,
296, 203, 194 nm; EI-MS m=z (rel abund %): 288 (Mþ,
51), 105 (100), 77 (34). Anal. Calcd for C18H12N2O2: C,
74.99; H, 4.20; N, 9.72. Found: C, 74.89; H, 4.25; N,
9.82.
6.1.9. 4-[(E)-(3-Aminophenyl)methylidene]-2-phenyl-1,3-
oxazol-5(4H)-one (9). 3-Amino benzaldehyde (1.85 g,
15.3 mmol), N-benzoylglycine (2.5 g, 13.9 mmol), acetic
anhydride (3.93 mL, 36.3 mmol) and fused sodium ace-
tate (1.14 g, 13.89 mmol) were treated in the same way as
described for compound 3. The yellow solid product
obtained was 1.85 g (50%). Mp: 179 ꢁC; Rf : 0.65 (n-
butanol–acetic acid–H2O 3:3:1); 1H NMR (300 MHz,
CDCl3) d: 4.24 (s, 2H, NH2), 7.24 (br s, 1H, H-6), 7.4 (t,
1H, J ¼ 7:8 Hz, H-20), 7.51 (t, 1H, J ¼ 7:8 Hz, H-30),
7.60 (d, 1H, J ¼ 7:2 Hz, H-40), 7.69 (d, 2H, J ¼ 8:0 Hz,
H-200,600), 7.70 (d, 2H, J ¼ 8:0 Hz, H-300), 8.17 (t, 1H,
J ¼ 7:2 Hz, H-400), 8.30 (s, 1H, H-60); EI-MS m=z (rel
abund %): 264 (Mþ, 13), 248 (39), 171 (56), 127 (63), 105
(100), 77 (83); IR (KBr): 3458, 1769, 1751, 1655, 1576,
1486, 1367, 1278, 1163, 1061, 980, 739 cmꢀ1; UV kmax
(CHCl3): 313, 269, 227, 215, 200, 197, 193 nm. Anal.
Calcd for C16H12N2O2: C, 72.72; H, 4.58; N, 10.60.
Found: C, 72.72; H, 4.57; N, 10.60.
6.1.6. 4-[(E)-(3-Chlorophenyl)methylidene]-2-phenyl-1,3-
oxazol-5(4H)-one (6). 3-Chlorobenzaldehyde (1.98 g,
14.1 mmol), N-benzoylglycine (2.5 g, 13.97 mmol), acetic
anhydride (3.93 mL, 41.7 mmol) and fused sodium ace-
tate (1.14 g, 13.9 mmol) were treated in the same way as
described for compound 3. The product was obtained as
yellow solid 1.58 g (40%). Mp: 166 ꢁC; Rf : 0.67 (n-buta-
nol–acetic acid–H2O 3:3:1); 1H NMR (400 MHz,
CDCl3) d: 7.16 (s, 1H, H-6), 7.40–7.42 (m, 3H,
H-300,400,500, Ar-H), 7.52 (dd, 1H, J ¼ 0:93, J ¼ 8:2 Hz,
H-20, H-30), 7.56 (d, 2H, J ¼ 6:4 Hz, H-200,600), 7.61 (d,
1H, J ¼ 8:2 Hz, H-200), 8.18 (dd, 1H, J ¼ 2:2, J ¼ 7:1 Hz,
H-40, Ar-H), 8.28 (s, 1H, H-60, Ar-H); IR (KBr): 1786,
6.1.10. 4-[(E)-(2-Bromophenyl)methylidene]-2-phenyl-1,3-
oxazol-5(4H)-one (10). 2-Bromobenzaldehyde (1.79 mL,
15.29 mmol), N-benzoylglycine (2.5 g, 13.96 mmol),
acetic anhydride (3.93 mL, 36.3 mmol) and fused sodium

 Mesaik, Muhammad A.
Mesaik, Muhammad A.
 Rahat, Shagufta
Rahat, Shagufta
 Khan, Khalid M.
Khan, Khalid M.
 Zia-Ullah
Zia-Ullah
 Choudhary, Muhammad I.
Choudhary, Muhammad I.
 Murad, Shahnaz
Murad, Shahnaz
 Ismail, Zakiah
Ismail, Zakiah
 Atta-ur-Rahman
Atta-ur-Rahman
 Ahmad, Aqeel
Ahmad, Aqeel