Ganapati D. Yadav, Sharad V. Lande
FULL PAPERS
Table 1 (cont.)
No. Reactant
Product
Enhancement factor[a] Time t [min] Yield [%] after time t
L-L PTC[b] L-L-L PTC[c]
11
12
6.0
4.6
60
60
28.6
33.5
96.7
95.4
The yield is the amount of product formed relative to the amount of substrate reacted. The yield is equal to selectivity here.
[a]
Enhancement factor is defined as the ratio of the initial rate of reaction under L-L-L PTC to that under L-L PTC (before
the third phase is created).
[b]
L-L PTC: nitroaromatics 0.0075 mol, sodium sulphide 0.1125 mol, TBAB 0.0025 mol, toluene 15 cm3, water 25 cm3, speed of
agitation 1500 rpm, temperature 808C.
L-L-L PTC: All same as for L-L PTC except TBAB 0.0075 (reusability of catalyst).
[c]
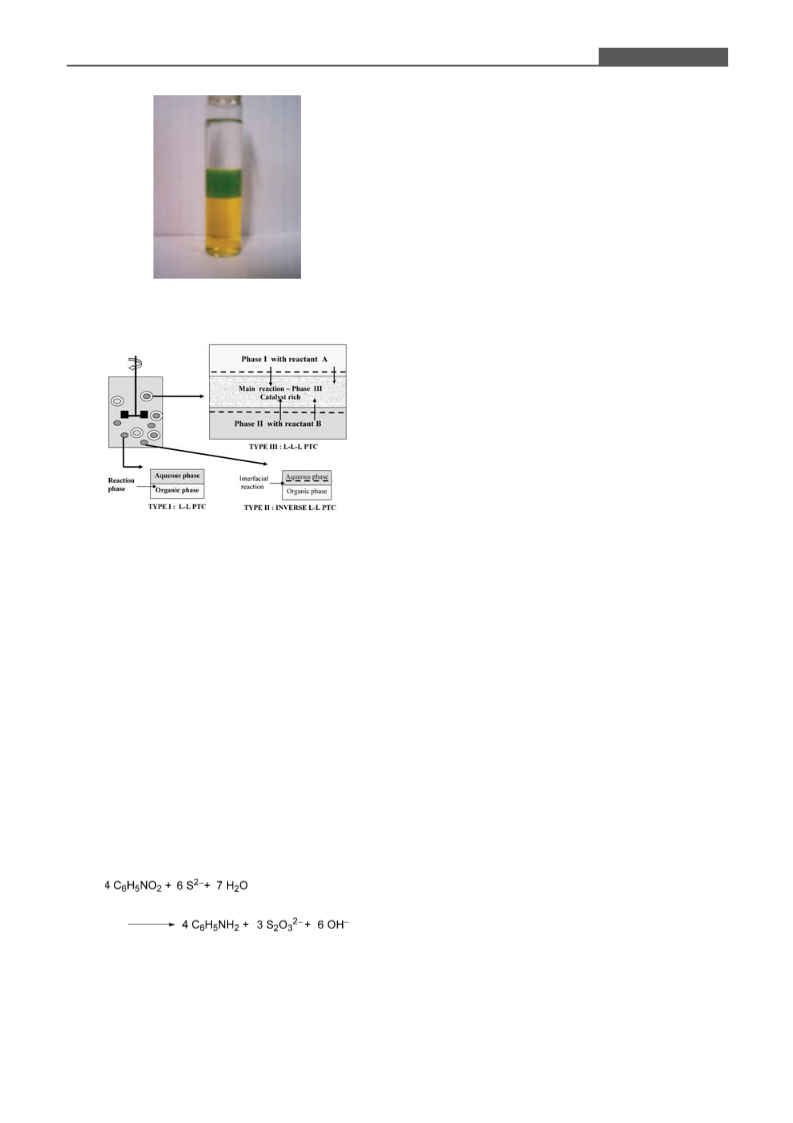
tinct and stable phases at the operating conditions (Figure 1).
The analysis of all reactions was done by GC (Chemito 8610)
using a stainless steel column (3.25 mmꢁ2 m) packed with a
liquid stationary phase of 10% SE-30, with a flame ionisation
detector.
[6] M. Beller, T. Gerdau, H. Strutz, Ger. Offen. DE 4316923,
1994.
[7] P. Baumeister, H.-U. Blaser, W. Scherer, Stud. Surf. Sci.
Catal. 1991, 59, 321.
[8] R. M. Despande, A. N. Mahajan, M. M. Diwakar, P. S.
Ozarde, R. V. Chaudhari, J .Org. Chem. 2004, 69, 4835.
[9] C. M. Starks, C. M. Liotta, M. Halpern, Phase Transfer
Catalysis: Fundamentals, Applications, and Perspectives,
Chapman and Hall, New York, 1994.
[10] Y. Sasson, R. Neumann, (Eds.), Handbook of Phase
Transfer Catalysis, Blackie Academic and Professional,
London, 1997.
[11] E. V. Dehmlow, S. S. Dehmlow, Phase Transfer Catalysis,
3rd Edn.; VCH, Weinheim, New York, 1993.
[12] a) G. D. Yadav, Chim. Ind. 2000, 6, 1; b) G. D. Yadav,
Top. Catal. 2004, 29, 145; c) G. D. Yadav. S. S. Naik,
Org. Proc. Res. Dev. 1999, 3, 83; d) G. D. Yadav, Y. B.
Jadhav, Langmuir 2002, 18, 5995; f) G. D. Yadav, B. V.
Haldavanekar, J. Phys. Chem. 1997, 101, 36; g) G. D. Ya-
dav, P. H. Mehta, Catal. Lett. 1993, 21, 391; h) G. D. Ya-
dav, P. M. Bisht, J. Mol. Catal. 2004, 221, 59.
[13] G. D. Yadav, P. H. Mehta, B. V. Haldavanekar, in: Heter-
ogeneous Catalysis and Fine Chemicals III, (Eds.: M.
Guisnet, J. Barbier, J. Barrault, D. Duprez, G. Perot, C.
Montassier), Elsevier, Amsterdam, 1993, p. 503.
[14] G. D. Yadav, C. K. Mistry, J. Mol. Catal. A: Chem. 1995,
102, 67.
[15] a) G. D. Yadav; S. S. Naik, Catal. Today 2001, 2357;
b) G. D. Yadav, C. A. Reddy, Ind. Eng. Chem. Res.
1999, 38, 2249; c) G. D. Yadav, Y. B. Jadhav Clean Tech.
Environ. Pol. 6, 2003, 32; d) G. D. Yadav, P. M. Bisht, J.
Mol. Catal. 2005, 223, 93–100; e) G. D. Yadav, S. V.
Lande, Appl. Catal. A. Gen. 2005, 287, 267–275.
[16] S. Hashimoto, H. Fiji, Doshisha Daigaku Rikogaku Ken-
kyu Hokoku 1973, 14, 168.
Isolation of the Product
The isolation of product is very simple. At the end of the reac-
tion, the organic phase, which contained the corresponding
amine and toluene, was washed with copious quantities of wa-
ter to remove any traces of catalyst and the solvent was distilled
under vacuum to get a pure product. All the products were also
confirmed by GC-MS. All the compounds reported are shown
in Table.1.
Acknowledgements
G. D. Yadav acknowledges Darbari Seth Endowment for sup-
porting the Chair. S. V.Lande acknowledges CSIR-NMITLI
for awarding JRF.
References
[1] P. F. Vogt, J. J. Gerulis, Ullmans Encyclopaedia of Indus-
trial Chemistry, 5th edn., Verlag Chemie, Weinheim 1985,
Vol. A2, pp 37–55.
[2] H.-U. Blaser, M. Studer, Appl. Catal. 1999, 189, 191.
[3] S. F. Birch, R. A. Dean, F. A. Fidler, R. A. Lowery, J.
Am. Chem. Soc. 1949, 71, 1362–1369.
[4] R. S. Downing, P. J. Kunkeler, H. Van Bekkum, Catal.
Today 1997, 47,121.
[5] P. Baumeister, N. Studer, F. Roessler, in: Handbook of
Heterogeneous Catalysis; (Eds.: G. Ertl, H. Knozinger,
J. Weitkamp), Wiley, New York, 1997, Vol. 5.
[17] A. Koller, P. Jaegar, C. Berther, Ger. Offen DE 2,204,726,
1972.
1240
ꢀ 2005 WILEY-VCH Verlag GmbH & Co. KGaA, Weinheim
asc.wiley-vch.de
Adv. Synth. Catal. 2005, 347, 1235–1241

 Yadav, Ganapati D.
Yadav, Ganapati D.
 Lande, Sharad V.
Lande, Sharad V.