G Model
CATTOD-9178; No. of Pages8
ARTICLE IN PRESS
2
S. Bednarz et al. / Catalysis Today xxx (2014) xxx–xxx
concentration each chemical compounds was made. The numer-
ical integration of the system and estimation of rate constants
were performed by using computational program Dynafit [17]. The
experimental data describing persulfate concentration vs. decom-
position time for different initial concentrations of choline salts
were fitted to the model by non-linear least-square regression
method based on the Levenberg–Marquardt algorithm.
Fig. 1. Free-radical polymerization of itaconic acid.
More recently, we have reported an alternative strategy to
prepare itaconic-based polymers [15]. In our approach, we have
used a DES, formed by melting of itaconic acid and choline chlo-
ride, as the polymerization system. The results prove usefulness
liminary comparative studies have shown that persulfate initiated
copolymerization of itaconic acid is faster in the DES than in water
which indicates acceleration effect inducted probably by presence
of choline cation. The mechanism of this phenomenon has not been
explored, yet [15].
In this work, we continue our investigations of the application of
choline salts as activators in free-radical polymerization of itaconic
acid. We focused on aqueous solutions of the salts and propose
possible explanation of the acceleration effect on the rate of the
polymerization. Additionally, we demonstrated that choline salts
due to its complexing properties, can be a component of solvents
for itaconic acid, useful for the polymerization of itaconic acid.
2.5. Degradation experiment
The reaction of decomposition of persulfate was conducted in
unbuffered D2O solution at 75 0.1 ◦C in borosilicate glass vial
heated in a thermostated aluminum block. Mixture of initially
0.1 mol/L ChCl and 0.01 mol/L APS was heated for 2 h. After cooling
to room temperature, 1H NMR spectra of the solution was taken
(Varian Mercury VX-300).
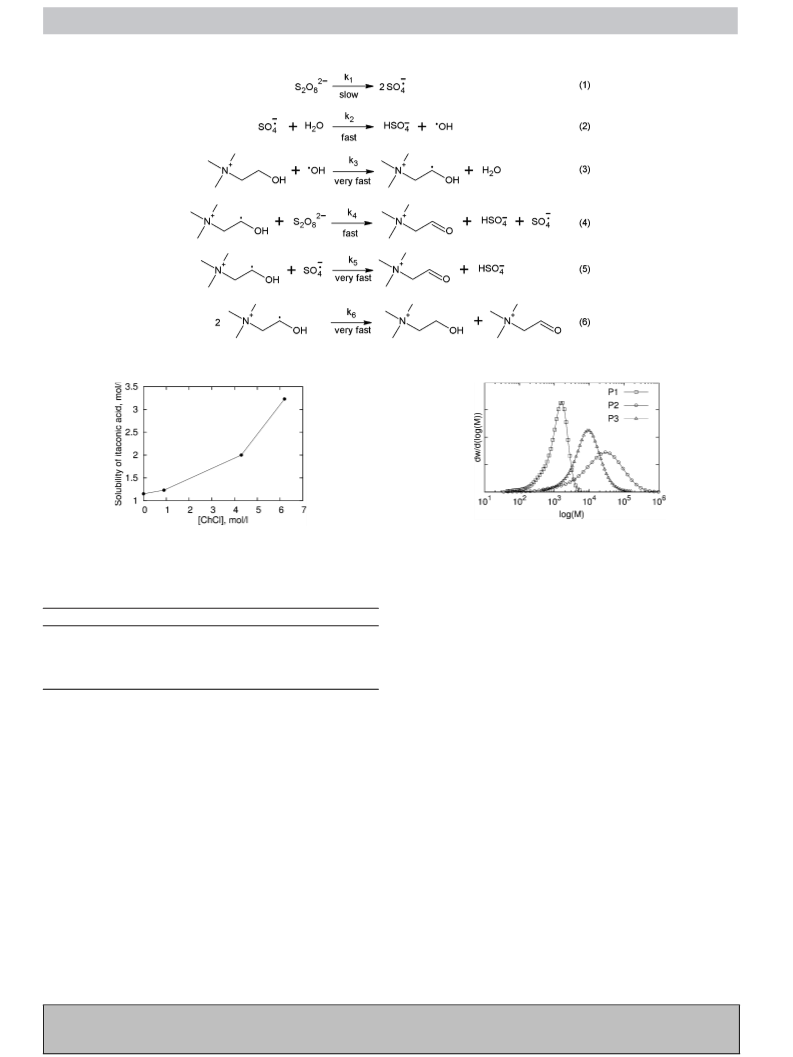
2.6. Determination of itaconic acid solubility
The solubility of itaconic acid in water or choline chloride solu-
tion was estimated using visual dynamic method. Experiments
were carried out in a glass vial (7 mL) placed in a thermostatic block
equipped with a magnetic stirrer. Itaconic acid in portions of 50 mg
was slowly added to a vigorously mixed solution until saturated
solutions were obtained. The measurement was repeated two times
and average value was taken.
2. Materials and methods
2.7. Synthesis of poly(itaconic acid)
2.1. Materials
Mixture of 25 mL of freshly distilled water, 5.20 g itaconic acid
(40 mmol), appropriate amount of choline chloride (0.91 g–4 mmol
or 9.1 g–40 mmol) and 0.456 g ammonium persulfate (2 mmol) was
heated for 96 h at 55 ◦C in a stoppered Erlenmeyer flask in cir-
culating oven. Next, the solution was dialyzed through cellulose
membrane (MWCO 1000 Da) against distilled water to remove
low-molecular weight products, and finally lyophilized. Yield of
polymerization was calculated as a ratio of weight of the starting
monomer and weight of the obtained polymer. Molecular weight
distribution of poly(itaconic acid) was determined using gel per-
meation chromatography (Phenomenex Poly-Sep-P Linear column,
eluent 0.1 M NaNO3 1 mL/min, RI detector). Polyethylene glycols
standards were used for calibration.
Analytical grade choline chloride (ChCl), itaconic acid (IA) and
ammonium persulfate (APS) were purchased from Sigma–Aldrich
(USA) and used as received. Potassium hydroxide, absolute ethanol,
glacial acetic acid and phosphoric acid (85%) were obtained from
POCH (Poland). In all the experiments distilled water was used.
2.2. Preparation of choline salts
Equimolar amount of choline chloride and KOH were dissolved
separately in absolute ethanol, and then the solutions were mixed
together with stirring and cooled to −25 ◦C for 24 h. Precipitated
KCl was separated by filtration through a sintered funnel. The
obtained choline hydroxide solutions were neutralized by appro-
priate amount of acids (H3PO4, CH3COOH, itaconic acid) yielded
salts: choline monohydrophosphate (Ch2HPO4), choline acetate
(ChAc), choline hydrogenitaconate (ChHIA) and choline itaconate
(Ch2IA). Ethanol and water were removed from the salt solutions
under vacuum using a rotary evaporator.
3. Results and discussion
3.1. Kinetics of choline-activated persulfate decomposition
The thermal decomposition of persulfate in water is commonly
written as:
2.3. Kinetics experiments
2−
•−
S2O8 → 2SO4
The reactions of decomposition of ammonium persulfate were
studied in unbuffered aqueous solution at 75 0.1 ◦C in borosili-
cate glass vials heated in a thermostated aluminum block. All the
of 0.01 mol/L with varied concentrations of choline salts ranged
from 0.0001 to 6.2 mol/L for ChCl and 0.001–1 mol/L for ChAc and
Ch2HPO4. The decomposition process was monitored by the deter-
mination of APS concentration using a spectrophotometric method
[16]. In additional experiments, it has been confirmed that choline
salt do not interfere with persulfate analysis.
However, it is well known that the process is quite complex and
the mechanism depends on changes in pH, presence of metal ions or
organic reducing agents, which can form a redox couple with per-
sulfate and thus catalyze the decomposition [18,19]. Additionally,
monomers, polymeric latexes or surfactants [20–24].
Recent studies suggest that presence of choline chloride could
accelerate polymerization of itaconic acid initiated by persulfates
[15]. A possible reason may be increasing rate of the initiator
decomposition by the salt. It is well known [25], that under the
steady state assumption, rate of free-radical polymerization is
directly proportional to square root of an initiator decomposition
rate constant. In other words, speeding up the initiation pro-
cess/decomposition rate enhances polymerizations.
2.4. Kinetic modeling
On the basis of the postulated reaction mechanism, a system
first-order ordinary differential equations describing changes of
Please cite this article in press as: S. Bednarz, et al., Free-radical polymerization of itaconic acid in the presence of choline salts:

 Bednarz, Szczepan
Bednarz, Szczepan
 B?aszczyk, Alicja
B?aszczyk, Alicja
 B?azejewska, Diana
B?azejewska, Diana
 Bogda?, Dariusz
Bogda?, Dariusz