´
RAMIREZ ET AL.
306
6. Chuchani, G.; Dom´ınguez, R. M.; Rotinov, A.; Mart´ın,
I. J Phys Org Chem 1999, 12, 612.
CONCLUSIONS
7. Chuchani, G.; Dom´ınguez, R. M.; Herize, A.; Romero,
R. J Phys Org Chem 2000, 13, 757.
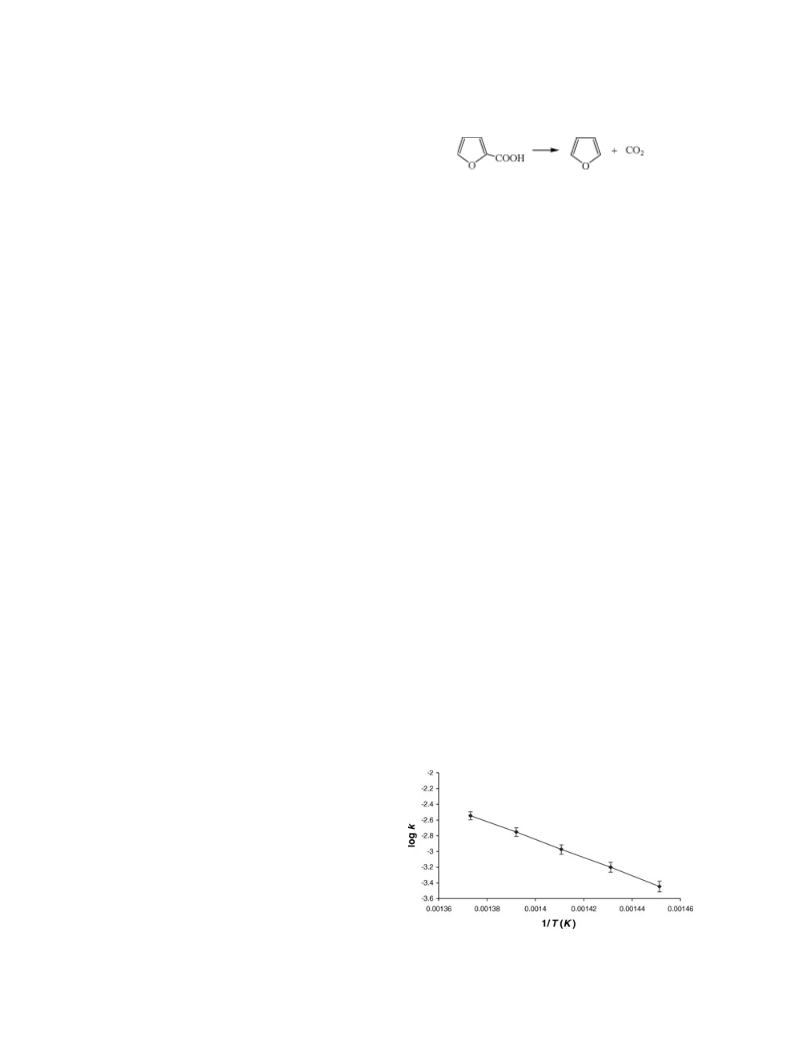
The experimental data show that the elimination pro-
cess of 2-furoic acid in the gas phase is homogeneous,
unimolecular, and follows a first-order rate law. Theo-
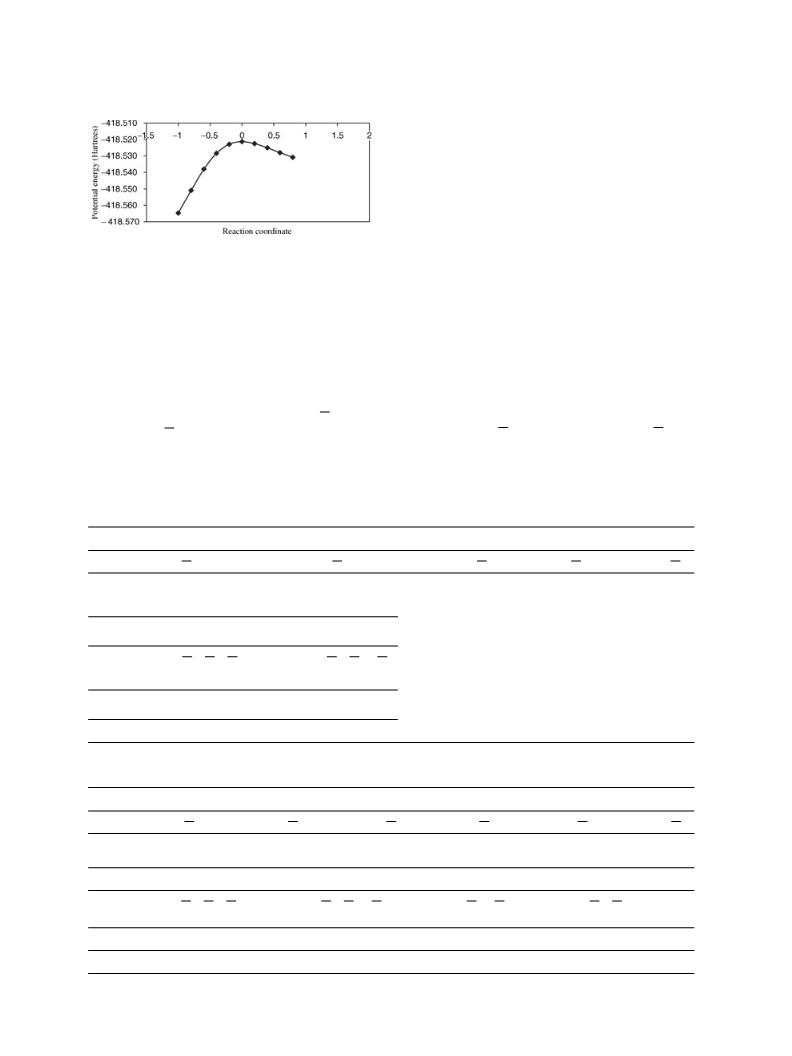
retical calculations suggest that the reaction proceeds
in a concerted asynchronous mechanism. Two con-
certed mechanisms were found for the thermal decom-
position of 2-furoic acid. Calculated activation param-
eters are in better accord with mechanism 1 at the
B3LYP/6-31++G∗∗ level of theory. The TS structure
for mechanism 1 is a four-membered ring, where the
four atoms lie in a plane forming 120◦ with the furan
ring plane. The process is concerted polar asynchronic,
dominated by the acid hydrogen transfer to the car-
bon bearing the carboxylic moiety in the furan ring.
Mechanism 2 is described as a cyclic TS structure in
which the five atoms involved are in a plane that is at
about 30◦ to the furan ring. This process is also dom-
inated by the breaking of the O H bond of the acid
moiety and concerted polar in nature. An experimen-
tal log A of 13.28 suggests that the reaction is likely
to occur through a four-membered-ring type of mech-
anism. Additionally, the formation of the zwitterion
species in the gas phase is unlikely because it cannot be
stabilized by solvent interactions. The small negative
value of the entropy of activation suggests loose po-
lar TS. These arguments favor mechanism 1; however,
mechanism 2 is not ruled out. NBO analysis suggests
that the polarization of the O H bond is the determin-
ing factor in the decomposition process and implies
a polar asynchronic process. A reasonable agreement
is achieved between the theoretical and experimental
results.
8. Ensuncho, A.; Lafont, J.; Rotinov, A.; Dom´ınguez,
R. M.; Herize, A.; Quijano, J.; Chuchani, G. Int J Chem
Kinetic 2000, 33, 465, and references cited therein.
9. Dominguez, R. M.; Tosta, M.; Chuchani, G. J Phys Org
Chem 2003, 16, 869.
10. Lafont, J.; Ensuncho, A.; Dominguez, R. M.; Rotinov,
A.; Herize, A.; Quijano, J.; Chuchani, G. J Phys Org
Chem 2003, 16, 84.
11. Maccoll, A. J Chem Soc 1955, 965.
12. Swinbourne, E. S. Aust J Chem 1958, 11, 314.
13. Dominguez, R. M.; Herize, A.; Rotinov, A.; Alvarez-
Aular, A.; Visbal, G.; Chuchani, G. J Phys Org Chem
2004, 17, 399.
14. Frisch, M. J.; Trucks, G. W.; Schlegel, H. B.; Scuseria,
G. E.; Robb, M. A.; Cheeseman, J. R.; Zakrzewski,
V. G.; Montgomery, J. A., Jr.; Stratmann, R. E.; Burant,
J. C.; Dapprich, S.; Millam, J. M.; Daniels, A. D.; Kudin,
K. N.; Strain, M. C.; Farkas, O.; Tomasi, J.; Barone,
V.; Cossi, M.; Cammi, R.; Mennucci, B.; Pomelli,
C.; Adamo, C.; Clifford, S.; Ochterski, J.; Petersson,
G. A.; Ayala, P. Y.; Cui, Q.; Morokuma, K.; Malick,
D. K.; Rabuck, A. D.; Raghavachari, K.; Foresman,
J. B.; Cioslowski, J.; Ortiz, J. V.; Stefanov, B. B.; Liu,
G.; Liashenko, A.; Piskorz, P.; Komaromi, I.; Gomperts,
R.; Martin, R. L.; Fox, D. J.; Keith, T.; Al-Laham,
M. A.; Peng, C. Y.; Nanayakkara, A.; Gonzalez, C.;
Challacombe, M.; Gill, P. M. W.; Johnson, B.; Chen,
W.; Wong, M. W.; Andres, J. L.; Gonzalez, C.;
Head-Gordon, M.; Replogle, E. S.; Pople, J. A. Gaussian
98, Revision A.3; Gaussian, Inc.: Pittsburgh, PA, 1998.
15. McQuarrie, D. Statistical Mechanics; Harper & Row:
New York, 1986.
16. Foresman, J. B.; Frish, Æ. Exploring Chemistry with
Electronic Methods, 2nd ed.; Gaussian, Inc: Pittsburgh,
PA, 1996.
17. Rotinov, A.; Dominguez, R. M.; Co´rdova, T.; Chuchani,
G. J Phys Org Chem 2005, 18, 616.
18. Benson, S. W. The Foundations of Chemical Kinetics;
Mc-Graw-Hill: New York, 1960.
BIBLIOGRAPHY
19. O’Neal, H. E.; Benson, S. W. J Phys Chem 1967, 71,
2903; Benson, S. B. Thermochemical Kinetics; John
Wiley & Sons: New York, 1968.
20. Lendvay, G. J Phys Chem 1989, 93, 4422.
21. Reed, A. E.; Weinstock, R. B.; Weinhold, F. J Chem
Phys 1985, 83(2), 735.
1. Safont, V. S.; Moliner, V.; Andres, J.; Domingo, L. R.
J Phys Chem A 1997, 101, 1859.
2. Domingo, L. R.; Andres, J.; Moliner, V.; Safont, V. S.
J Am Chem Soc 1997, 119, 6415.
3. Domingo, L. R.; Pitcher, M. T.; Andres, J.; Moliner, V.;
Safont, V. S.; Chuchani, G. Chem Phys Lett 1997, 274,
422.
22. Reed, A. E.; Curtiss, L. A.; Weinhold, F. Chem Rev
1988, 88, 899.
4. Domingo, L. R.; Pitcher, M. T.; Safont, V.; Andres,
23. Wiberg, K. B. Tetrahedron 1968, 24, 1083.
24. Gaussian NBO, version 3.1.
J.; Chuchani, G.
3935.
J Phys Chem A 1999, 103,
25. Moyano, A.; Periclas, M. A.; Valenti, E. J Org Chem
1989, 54, 573.
5. Rotinov, A.; Chuchani, G.; Andres, J.; Domingo, L. R.;
Safont, V. S. Chem Phys 1999, 246, 1.
International Journal of Chemical Kinetics DOI 10.1002/kin

 Ramirez, Beatriz C.
Ramirez, Beatriz C.
 Dominguez, Rosa M.
Dominguez, Rosa M.
 Herize, Armando
Herize, Armando
 Tosta, Maria
Tosta, Maria
 Cordova, Tania
Cordova, Tania
 Chuchani, Gabriel
Chuchani, Gabriel