Month 2016
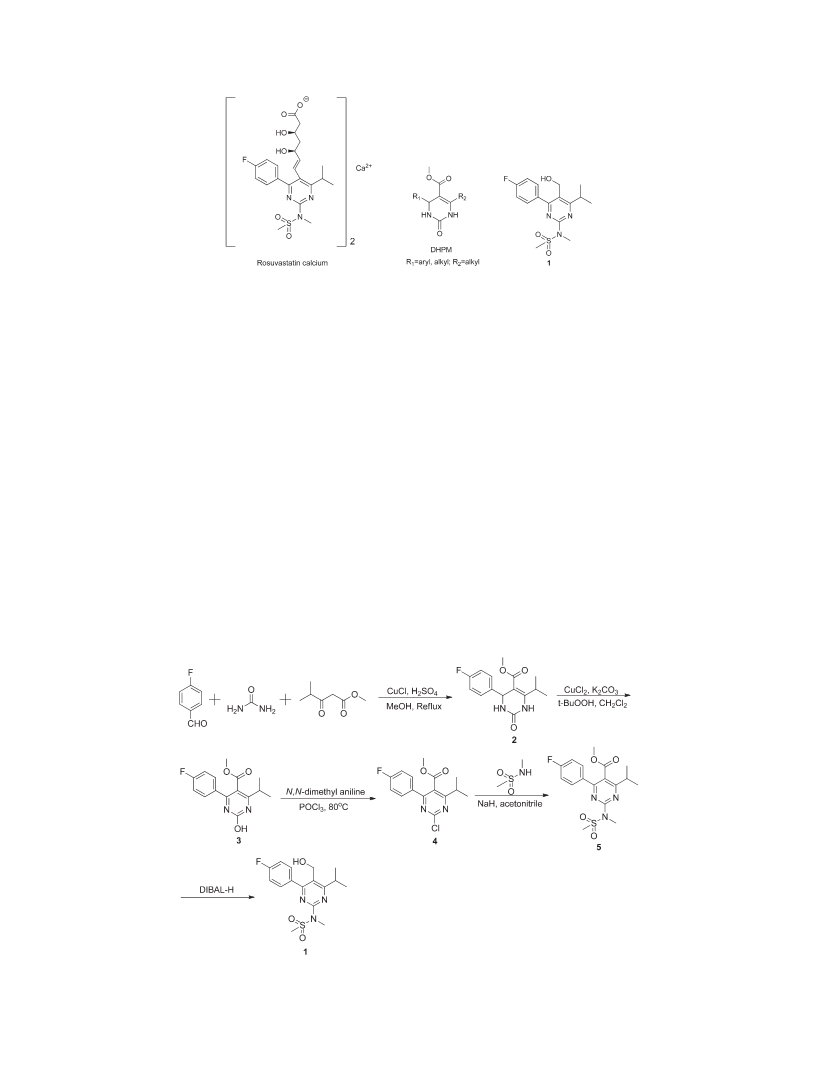
Efficient Construction of the Nucleus of Rosuvastatin Calcium
1
3
1
2
1
1
H). C NMR (100MHz, DMSO-d6) δ: 19.43, 19.65,
7.56, 51.42, 53.56, 98.41, 115.74 (d, JCF = 21.1Hz),
28.62 (d, JCF = 8.2Hz), 141.29 (d, JCF =2.9 Hz), 153.13,
Methyl 4-(4-Fluorophenyl)-6-Isopropyl-2-(Methanesulfon-
yl-Methyl-Amino)-Pyrimidine-5-Carboxylate (5).
Sodium
hydride (0.73 g, 0.018 mol, 60% suspension in mineral
oil) was suspended in acetonitrile (30 mL). N-methyl
Methanesulfonamide (3.15g, 0.030 mol) was added
dropwise. The resulting suspension was stirred for 5 min,
and compound 4 (4.68 g, 0.015 mol) was added, washed
in with acetonitrile (20 mL). The mixture was heated to
reflux and stirred for 7 h. After cooled to room
temperature, the mixture was extracted with ethyl acetate
(80 mL) and water (120mL). The organic phase was
separated and concentrated under reduced pressure to
give crude product. Then it was purified by
recrystallization in methanol. The solid was dried in
vacuum to yield 5 (5.33 g, 93.2% yield). mp 132.1–
57.35, 161.87 (d, J =241.8 Hz), 166.23. HRMS (ESI):
m/z [M+ Na] calcd for C H FN O 315.1121; found:
15.1121.
CF
+
1
5
17
2 3
3
Methyl 4-(4-Fluorophenyl)-2-Hydroxy-6-Isopropylpyrimi-
dine-5-Carboxylate (3). Dihydropyrimidinone 2 (5.00g,
7.00 mmol) was dissolved in methylene chloride
40 mL). Copper (II) chloride (0.03g, 0.17 mmol) and
potassium carbonate (0.23 g, 1.70mmol) were added, and
the mixture was cooled to 25°C. Then, aqueous TBHP
solution (70 wt%, 5.47g, 42.5mmol) was added
dropwise. The mixture was warmed to 40°C and stirred
for 7 h. At that time, the solution was cooled to room
temperature, and the potassium iodide-starch test paper
was used to detect residual peroxide. If the paper turned
to blue, 15% Na S O aqueous solution was added to
1
(
1
132.7°C. (lit.[5] 130.0–132.5°C). H NMR (400MHz,
CDCl ) δ: 1.35 (d, J = 6.8 Hz, 6H), 3.20–3.27 (m, 1H),
3
3.55 (s, 3H), 3.63 (s, 3H), 3.74 (s, 3H), 7.18 (t,
2
2 4
13
quench the peroxide. If not, the solution was diluted with
water (50 mL) and extracted with CH Cl (50 mL × 2).
J = 8.6 Hz, 2H), 7.70–7.73 (m, 2H). C NMR (100MHz,
CDCl ) δ: 21.79, 33.09, 33.37, 42.43, 52.69, 115.77 (d,
2
2
3
The organic phases were combined and concentrated
under reduced pressure to give a slightly yellow solid.
The solid was vacuum dried to give 4.75 g of 3 (95.66%
JCF = 21.8Hz), 118.82, 130.46 (d, JCF = 8.6 Hz), 133.80
(d, JCF = 3.3 Hz), 158.56, 163.00 (d, JCF = 32.0Hz),
165.34, 168.71, 174.79. HRMS (ESI): m/z [M+ Na]
+
1
yield). mp 186.0–190.0°C. (lit.[18] 193°C). H NMR
calcd for C H FN O S 404.1057; found: 404.1057.
17
20
3 4
(
(
400 MHz, CDCl ) δ: 1.47 (d, J = 6.4Hz, 6H), 3.24–3.27
m, 1H), 3.64 (s, 3H), 7.16 (t, J = 7.4Hz, 2H), 7.66 (s,
With extra NaH in this reaction, two side products were
detected.
3
2
H). (The hydroxy proton at 5.0ppm was not observed).
Methyl 4-(4-Fluorophenyl)-6-Isopropyl-2-(Methanesulfonyl-
13
C NMR (100 MHz, CDCl ) δ: 20.58, 31.85, 52.60,
Methyl-Amino)-Pyrimidine-5-Carboxylic Acid (6).
mp
3
1
1
1
15.67, 115.89, 130.07, 130.16, 163.08, 165.59, 167.13,
67.21, 167.34. HRMS (ESI): m/z [M+ Na] calcd for
208.8–209.6°C. H NMR (400MHz, CD OD) δ: 1.35
3
+
(d, J= 6.4 Hz, 6H), 3.33–3.34 (m, 1H), 3.54 (s, 3H),
3.58 (s, 3H), 7.23 (t, J = 8.6 Hz, 2H), 7.83–7.87 (m,
2H). (The hydroxy proton at 11.0 ppm was not
C H FN O 313.0965; found: 315.0966.
15
15
2 3
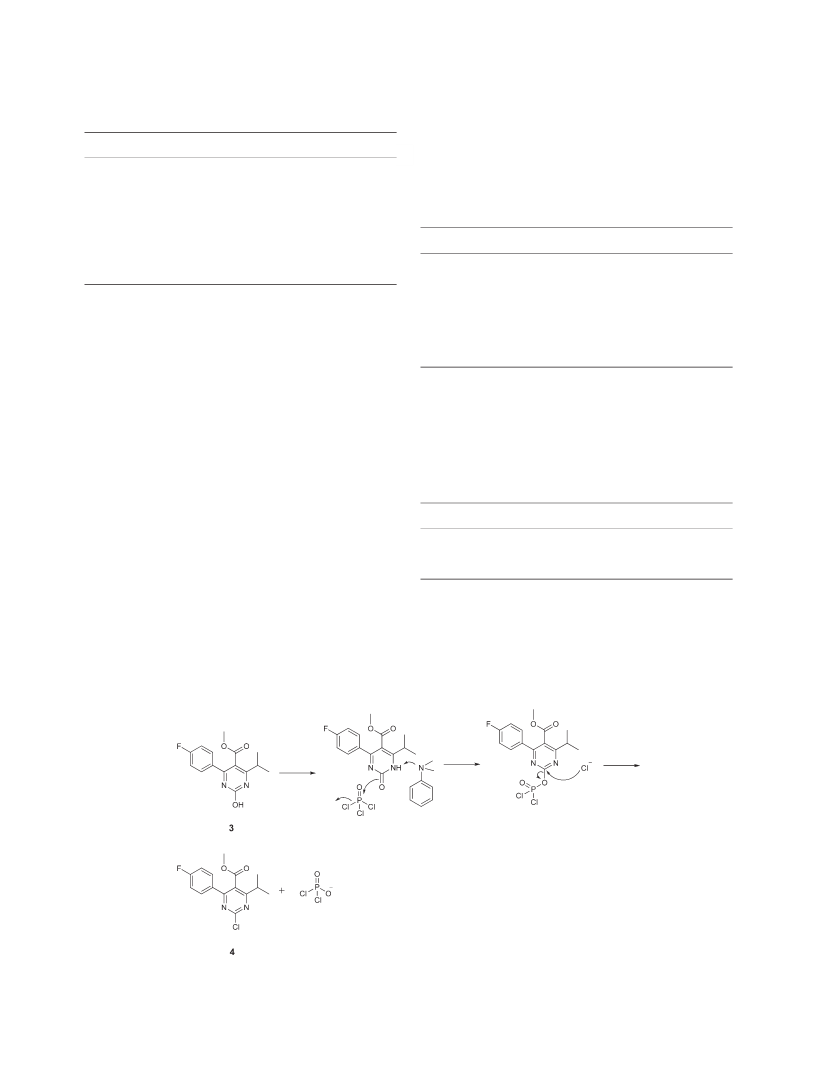
Methyl 2-Chloro-4-(4-Fluorophenyl)-6-Isopropylpyrimidi-
1
3
ne-5-Carboxylate (4).
Compound 3 (4.75 g, 0.016mol)
observed). C NMR (100 MHz, CD OD) δ: 20.76,
3
was suspended in POCl (20.08 g, 0.131 mol) at room
32.30, 33.21, 40.89, 115.11 (d, JCF = 22.0 Hz), 120.02,
130.58 (d, JCF = 8.7 Hz), 133.85 (d, JCF = 3.3 Hz),
158.50, 162.70 (d, JCF = 44.7 Hz), 165.40, 169.93,
3
temperature. N,N-dimethylaniline (0.97 g, 0.008 mol) was
added, and the mixture was stirred for 2.5h at 80°C
under nitrogen. At that time, the mixture was cooled to
+
173.91. HRMS (ESI): m/z [M + Na] calcd for
0
°C and slowly quenched with a dropwise addition of
C H FN O S 390.0900; found: 390.0906.
16
18
3 4
water (100 mL). When the addition was complete, the
mixture was warmed to room temperature and stirred for
Methyl 4-(4-Fluorophenyl)-6-Isopropyl-2-(Methylamino)-
Pyrimidine-5-Carboxylate (7). mp 100-100.6°C. H NMR
1
5
min. The aqueous solution was extracted with CH Cl2
(400MHz, CDCl ) δ: 1.28 (d, J=6.8Hz, 6H), 3.05 (d,
2
3
(
100 mL ×2). The organic phases were combined and
J=5.2Hz, 3H), 3.15–3.21 (m, 1H), 3.63 (s, 3H), 5.37 (s,
1H) 7.12 (t, J=8.6Hz, 2H), 7.60 (s, 2H). C NMR
13
concentrated under reduced pressure. The crude material
was purified by recrystallization in ethanol/water. The
solid was vacuum dried to give 4.68 g of 4 (93.60%
(100MHz, CDCl ) δ: 21.60, 28.01, 32.83, 52.04, 114.10,
3
115.34 (d, JCF =21.6Hz), 129.93 (d, JCF =8.3Hz), 135.32
(d, JCF =3.0Hz), 162.25, 163.27 (d, JCF =192.0Hz),
1
yield). mp 102.0°C (lit.[18] 99–101°C). H NMR
+
(
(
(
400 MHz, CDCl ) δ: 1.26 (d, J = 4.4Hz, 6H), 3.04–3.07
m, 1H), 3.68 (s, 3H), 7.08 (t, J = 5.8Hz, 2H), 7.59–7.62
m, 2H). C NMR (100MHz, CDCl ) δ: 21.58, 33.67,
164.78, 169.74, 174.94. HRMS (ESI): m/z [M+H] calcd
3
for C H FN O 304.1461; found: 304.1460.
16
18
3 2
13
4-(4-Fluorophenyl)-6-Isopropyl-2-(Methanesulfonyl-Methyl-
Amino)-Pyrimidin-5-yl Methanol (1). Compound 5 (5.00g,
0.013mol) was dissolved in toluene (30mL). The mixture
was cooled to À15°C, and DIBAL (1M in toluene,
33mL, 0.033mol) was added dropwise over 30min. The
internal temperature was kept below À15°C during the
3
5
2.98, 115.96 (d, JCF = 21.9Hz), 123.29, 130.58 (d,
JCF = 8.7Hz), 132.52 (d, JCF = 3.2Hz), 161.27, 163.08,
65.23 (d, JCF+= 72.1Hz), 167.81, 176.65. HRMS (ESI):
m/z [M+ Na] calcd for C H ClFN O 331.0626;
1
1
5
14
2 2
found: 331.0625.
Journal of Heterocyclic Chemistry
DOI 10.1002/jhet

 Zhou, Yingtao
Zhou, Yingtao
 Lin, Chenhui
Lin, Chenhui
 Xing, Yuzhi
Xing, Yuzhi
 Chen, Ligong
Chen, Ligong
 Yan, Xilong
Yan, Xilong