148
F. Xue et al. / Catalysis Communications 72 (2015) 147–149
Table 1
[14]. It is well recognized that residues F12, P175, N176, Y177, L178,
Y185, F186, Y187 and W249 suggested to play critical roles in cata-
lytic activity and enantioselectivity [20], thus will commonly act as
the target sites for rational design. The mutant libraries were
screened for the enantioselectivity toward 1,3-DCP by using auto-
mated chiral GC to obtain the respective conversion and ee values.
The cell free extracts of wild-type (WT) HheC and mutants were
purified by one-step nickel affinity chromatography on Ni-NTA
resin as described in our previous work [21]. It is found that there
is no difference in the molecular mass between the WT and mutant
HheC (Fig. S1).
For most of the mutants, the resulting ECH from 1,3-DCP was racemic,
suggesting that no positive mutant with improved enantioselectivity was
isolated at position F12, N176, Y177, L178, Y185 and F186. Among site-
saturation mutagenesis libraries at position P175 and W249, two mutants
P175S and W249P, were identified, displaying higher enantioselectivity
toward 1,3-DCP (Table 1). W249P and P175S increased the ee value of
(S)-ECH from 5.2% to 10.4% and 89.3%. Combination of mutations can
have additive effects in the case of enantioselectivity. The double-
mutant mutant (P175S/W249P) exhibited higher enantioselectivity (up
to 95.3% ee) than either of the two single-mutated mutants (Table 1).
The enhanced enantioselectivity is caused by the decreasing steric hin-
drance of one of the halogen-bearing carbon atoms of 1,3-DCP, resulting
in asymmetric dehalogenation.
The stereoselective dehalogenation of 1,3-DCP with recombinant halohydrin
dehalogenase.
Halohydrin
Enzyme
Specific
ee epoxide
(%)
Analytical
yield (%)b
Abs.
activitya
config.c
1,3-DCP
WT
W249P
P175S
34.7
24.3
43.9
40.6
5.2
10.4
89.3
95.3
43.8
36.2
90.9
93.7
(S)
(S)
(S)
(S)
P175S/W249P
a
In Na2HPO4–NaH2PO4, pH 8.0, substrate concentration is 20 mM, activity is expressed
as μmol/min/mg.
b
The maximum analytical yield is 100%.
Absolute configuration of the epoxide.
c
2. Materials and methods
2.1. Strains and plasmids
Escherichia coli BL21(DE3), pET-28b(+), pCDFDuet-1 were used for
expression experiments. The plasmid pET28b-HheC hosting the gene
encoding HHDH gene from A. radiobacter AD1 was used as the template
for construction of the mutation genes [18]. The E. coli strain expressing
AmEH mutant VDF (W182F/S207V/N240D) from A. mediolanus
ZJB120203 was developed as an efficient catalyst for the preparation
of enantiopure ECH by kinetic resolution [19].
3.2. Synthesis of (S)-ECH by mutant HheC
2.2. Analytical methods
The HheC mutant P175S/W249P was then evaluated for its asym-
metric conversions of 1,3-DCP (Table 2). The ee of (S)-ECH produced
was affected by the initial pH over the range of 8.0–10.0, and the yield
of (S)-ECH decreased from 89.3% to 50.7% as the pH of the reaction mix-
ture was changed from 10.0 to 8.0. The effect of 1,3-DCP concentration
on the optical purity of the (S)-ECH was also investigated. The ee and
yield of (S)-ECH respectively decreased from 92.7% and 91.4% to 90.4%
and 58.0% as the 1,3-DCP concentration was increased from 20 to
100 mM. A slight decrease in the optical purity of (S)-ECH from 93.5%
to 92.2% has been observed, due to spontaneous dehalogenation rate in-
creases with the elevated temperature.
The optical purity and conversion were determined by GC-14C gas
chromatography (Shimadzu, Tokyo, Japan) equipped with FID detector
and BGB-175 chiral column using He as carrier gas. The initial column
temperature was set at 90 °C and the inlet and detector temperatures
were both 220 °C. The retention times of (S)-ECH, (R)-ECH were 5.4
and 5.7 min. The ee was derived from the remaining epoxide of the
two enantiomers [ee (%) = (S − R) / (S + R) × 100]. Optical rotation
was measured on an Autopol IV automatic polarimeter (Rudolph
Research Analytical, USA).
See Supplementary data for other experimental details.
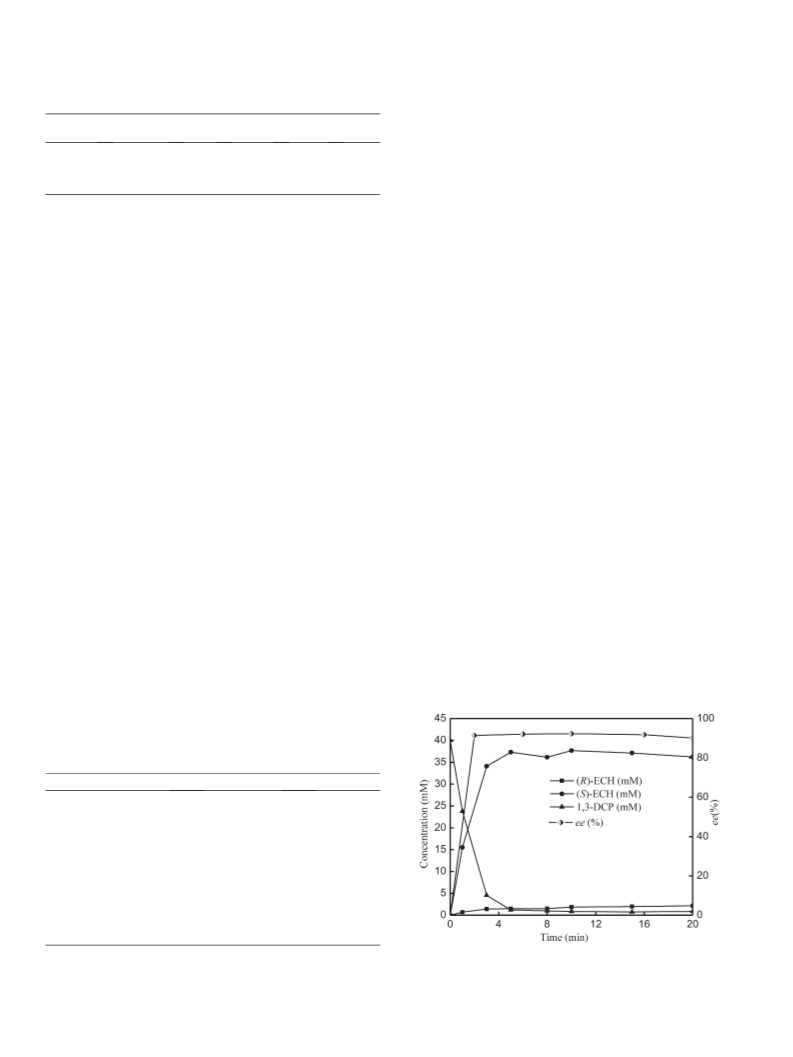
The production profile of (S)-ECH from 40 mM 1,3-DCP with time by
HheC mutant (P175S/W249P) with initial pH at 10.0 is shown in Fig. 1.
(S)-ECH was obtained with 93.2% yield and 92.3% ee determined by GC
in a 5-min reaction. Prochiral 1,3-DCP was transformed into optically
active ECH by HHDHs, which has been reported in the literature. (S)-
ECH with ee N 60% was obtained and a conversion was 95.2% in a
2-min reaction at pH 10.0 by HHDHTm from Tistrella mobilis ZJB1405
3. Results and discussion
3.1. Construction and screening of mutant libraries of HheC
In order to choose appropriate randomization sites, the published
X-ray crystal structure and catalytic mechanism of HheC from
A. radiobacter AD1 was used as template for comparison and analysis
Table 2
Optimization of reaction conditions for production of (S)-ECH from 1,3-DCP using HheC
mutant (P175S/W249P).
Reaction conditions
Analytical yield (%)a
(S)-ECH (ee %)
pHb
8.0 (Na2HPO4–NaH2PO4)
9.0 (Gly-NaOH)
50.7
71.4
89.3
94.2
94.5
92.3
10.0 (Gly-NaOH)
Temperature (°C)c
20
30
40
90.2
90.7
91.1
93.5
92.4
92.2
1,3-DCP concentration (mM)d
20
40
80
100
91.4
90.2
64.9
58.0
92.7
92.1
91.3
90.4
a
The maximum analytical yield is 100%.
40 mM 1,3-DCP, 35 °C, 3 min.
40 mM 1,3-DCP, 3 min pH 10.0, 200 mM Gly-NaOH.
200 mM Gly-NaOH (pH 10.0), 35 °C.
b
Fig. 1. Time course of the transformation of 1,3-DCP into (S)-ECH catalyzed by HheC
mutant (P175S/W249P). The reaction was performed at 35 °C in 200 mM glycine-NaOH
buffer (pH 10.0).
c
d

 Xue, Feng
Xue, Feng
 Liu, Zhi-Qiang
Liu, Zhi-Qiang
 Wang, Ya-Jun
Wang, Ya-Jun
 Zhu, Hang-Qin
Zhu, Hang-Qin
 Wan, Nan-Wei
Wan, Nan-Wei
 Zheng, Yu-Guo
Zheng, Yu-Guo