Z.-q. Yu et al. / Tetrahedron Letters 54 (2013) 1261–1263
1263
Table 2
Diazotization/fluorodediazoniation in a continuous flow reactor
ArNH a
s
(s)
Y
1
b
(%)
Cosolvent
T
2
(oC)
Y
2
c
(%)
Yd (%)
Entry
2
1
1
2
H
2-Me
15
10
96
95
Fluorobenzene
1,2-Dichlorobenzene
125
150
150
100
180
180
190
200
200
200
130
110
160
125
130
180
96
95
95
92
97
97
95
79
97
35
68
92
90
96
91
96
92
90
90
79
93
93
93
72
95
34
58
88
87
89
79
93
1-Fluoro-2-Methylbenzene
3
4
2-Et
2-Cl
10
20
86
96
Petroleum ether
1,2-Dichlorobenzene
1-Chloro-2-Fluorobenzene
5
6
7
8
9
0
1
2
3
4
2,4-Cl
2,4-F
2,5-Cl
20
20
20
20
10
10
20
10
10
20
98
91
98
98
85
96
97
93
87
97
1,2-Dichlorobenzene
1,2-Dichlorobenzene
1,2-Dichlorobenzene
1,2-Dichlorobenzene
Chlorobenzene
3-NO
2
3,4-OMe
3,5-Me
3,5-Cl
4-Me
4-OMe
4-Cl
1
1
1
1
1
Chlorobenzene
1,2-Dichlorobenzene
1-Fluoro-4-methylbenzene
1-Fluoro-4-methoxybenzene
1-Chloro-4-fluorobenzene
a
b
c
All reactions were performed on a 100 mmol scale.
Y
Y
1
is the yield of ArN
is the yield of ArF and calculated based on ArN
2
BF
4
and calculated based on ArNH
BF
2
.
2
2
4
.
d
2
Y is the yield of ArF and calculated based on ArNH .
References and notes
1.
(a) Riva, E.; Gagliardi, S.; Mazzoni, C.; Passarella, D.; Rencurosi, A.; Vigo, D.;
Martinelli, M. J. Org. Chem. 2009, 74, 3540–3543; (b) Wegner, J.; Ceylan, S.;
Kirschning, A. Adv. Synth. Catal. 2012, 354, 17–57; (c) Petersen, T. P.; Ritzén, A.;
Ulven, T. Org. Lett. 2009, 11, 5134–5137; (d) Riva, E.; Gagliardi, S.; Mazzoni, C.;
Passarella, D.; Rencurosi, A.; Vigo, D.; Rencurosi, A. Tetrahedron 2010, 66, 3242–
3247; (e) Kulkarni, A. A.; Kalyani, V. S.; Joshi, R. A.; Joshi, R. R. Org. Process Res.
Dev. 2009, 13, 999–1002; (f) Wahab, B.; Ellames, G.; Passey, S.; Watts, P.
Tetrahedron 2010, 66, 3861–3865; (g) Wiles, C.; Watts, P. Green Chem. 2012, 14,
38–54.
2
3
.
.
Balz, G.; Schiemann, G. Chem. Ber. 1927, 60, 1186–1190.
(a) Taylor, S. D.; Kotoris, C. C.; Hum, G. Tetrahedron 1999, 55, 12431–12477; (b)
Lal, G. S.; Pez, G. P.; Syvret, R. G. Chem. Rev. 1996, 96, 1737–1755; (c) Laali, K. K.;
Gettwert, V. J. J. Fluorine Chem. 2001, 107, 31–34; (d) Heredia-Moya, J.; Kirk, K.
L. J. Fluorine Chem. 2007, 128, 674–678.
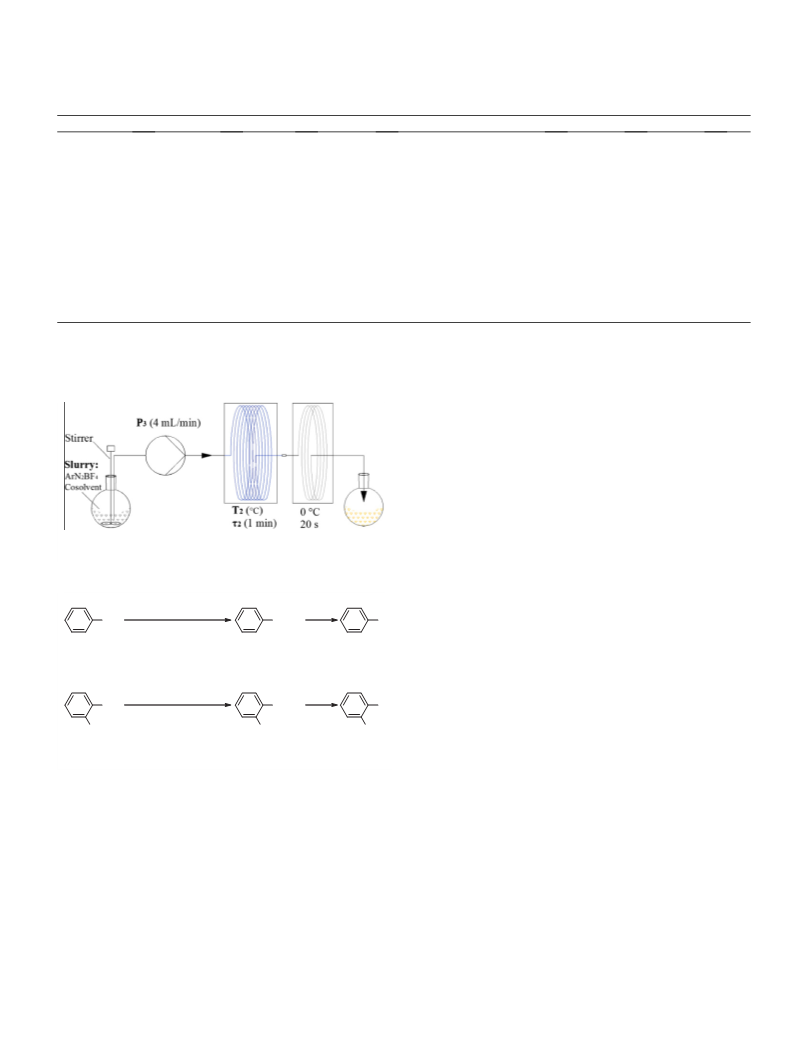
Figure 4. Continuous fluorodediazoniation set-up (T
thermostat, is the residence time in reacting tube).
2
is the temperature of
s
2
4
5
.
.
Fortt, R.; Wootton, R. C. R.; de Mello, A. J. Org. Process Res. Dev. 2003, 7, 762–768.
Malet-Sanz, L.; Madrzak, J.; Holvey, R. S.; Underwood, T. Tetrahedron Lett. 2009,
5
0, 7263–7267.
6. Wootton, R. C. R.; Fortt, R.; de Mello, A. J. Lab Chip 2002, 2, 5–7.
Malet-Sanz, B.; Madrzak, J.; Ley, S. V.; Baxendale, I. R. Org. Biomol. Chem. 2010,
, 5324–5332.
8. Flood, D. T. Org. Synth. 1933, 13, 295–297.
Momota, K.; Yonezawa, T. Jpn. Kokai Tokkyo Koho JP 199103232828 A.
10. Dolensky, B.; Takeuchi, Y.; Cohen, L. A.; Kirk, K. L. J. Fluorine Chem. 2001, 107,
47–148.
1
1
.8 eq HCl, 1.2 eq HBF4
.05 eq NaNO2
7
.
Heat
8
NH2
NH2
N2BF4
F
F
0
°C
9
.
Y : 83% in batch
Y : 92% in batch
1
2
9
6% in flow
96% in flow
1
1
1. (a) Sawaguchi, M.; Fukuhara, T.; Yoneda, N. J. Fluorine Chem. 1999, 97, 127–133;
(b) Ferm, R. L.; VanderWerf, C. A. J. Am. Chem. Soc. 1950, 72, 4809–4810; (b)
Stepaniuk, N. J.; Lamb, B. J. U.S. Patent 4,822,927, 1989.
1
1
.8 eq HCl, 1.2 eq HBF4
.05 eq NaNO2
Heat
12. (a) Müeller, A.; Roth, U.; Siegert, S.; Miethchen, R. Z. Chem. 1986, 26, 169–170;
(b) Hara, S.; Fukuhara, T. PCT Int. Appl. WO 2004050676.
N2BF4
0
°C
13. Lv, Y. W.; Yu, Z. Q.; Su, W. K. Org. Process Res. Dev. 2011, 15, 471–475.
Et
Et
Et
Y : 72% in batch
Y : 89% in batch
1
2
14. Typical continuous diazotization procedure: Material A (50 mL of aqueous
8
6% in flow
92% in flow
solution containing amine (100 mmol), fluoroboric acid (120 mmol),
hydrochloric acid (180 mmol)), and material B (50 mL of aqueous solution
containing sodium nitrite (105 mmol)) were pumped into the T-joint at 4 mL/
min, respectively, after a residence time of about 15 s at 25 °C in a reacting
tube, the mixture flowed through the outlet and accumulated in the cooling
vessel. Vigorous stirring was maintained. The solid was filtered with suction
after the slurry was cooled to À5 °C. The solid was washed with methanol and
then dried in vacuo to yield the corresponding diazonium tetrafluoroborate.
5. Typical continuous fluorodediazoniation procedure: Slurry of the diazonium
tetrafluoroborate prepared as above in 300 mL of cosolvent was introduced
into a reacting tube continuously at a flow rate of 4 mL/min. The mixture was
maintained for 1 min at setting temperature and then cooled in the tandem
tube. The collected liquid was washed with aqueous NaOH and water, nearly
colorless liquid was obtained.
Scheme 2. Typical batch comparison.
batch procedures. This work is a new horizon to the Balz–Schie-
mann reaction and furthers the development of diazonium species
in continuous flow processes.
1
1
Acknowledgments
We are grateful for the National Natural Science Foundation of
China (No. 21176222) and the Zhejiang Technology and Service
Platform of New Drug Innovation (PNDI) (No. 2011E61003) for
financial support.
6. Typical batch experiment. Batch reaction for fluorobenzene (compared to Table 2,
entry 1): A mixture of aniline (9.31 g, 100 mmol), 40% aqueous fluoroboric acid
(120 mmol), and 31% aqueous hydrochloric acid (180 mmol) were poured into
a
glass bottle and cooled to 0 °C. 2.1 mol/L of aqueous sodium nitrite
(
1
105 mmol) was added. The mixture was stirred vigorously for another
5 min when the addition was complete. After cooling to À5 °C, the solid
Supplementary data
was filtered with suction. The white cake was washed with methanol and dried
in vacuo to yield solid diazonium (83 mmol, 83%). The solid powder was placed
in a distilling flask and heated gently until no more white fumes of boron
fluoride evolved. The distillate was washed with aqueous NaOH and water, and
2 4
a yellowish liquid was obtained (76.36 mmol, 92% based on PhN BF ).

 Yu, Zhi-Qun
Yu, Zhi-Qun
 Lv, Yan-Wen
Lv, Yan-Wen
 Yu, Chuan-Ming
Yu, Chuan-Ming
 Su, Wei-Ke
Su, Wei-Ke