
Bioscience, Biotechnology and Biochemistry p. 1280 - 1286 (2015)
Update date:2022-08-24
Topics:
 Nakamura, Takashi
Nakamura, Takashi
 Asai, Shinji
Asai, Shinji
 Nakata, Kaori
Nakata, Kaori
 Kunimoto, Kohei
Kunimoto, Kohei
 Oguri, Masateru
Oguri, Masateru
 Ishikawa, Kazuhiko
Ishikawa, Kazuhiko
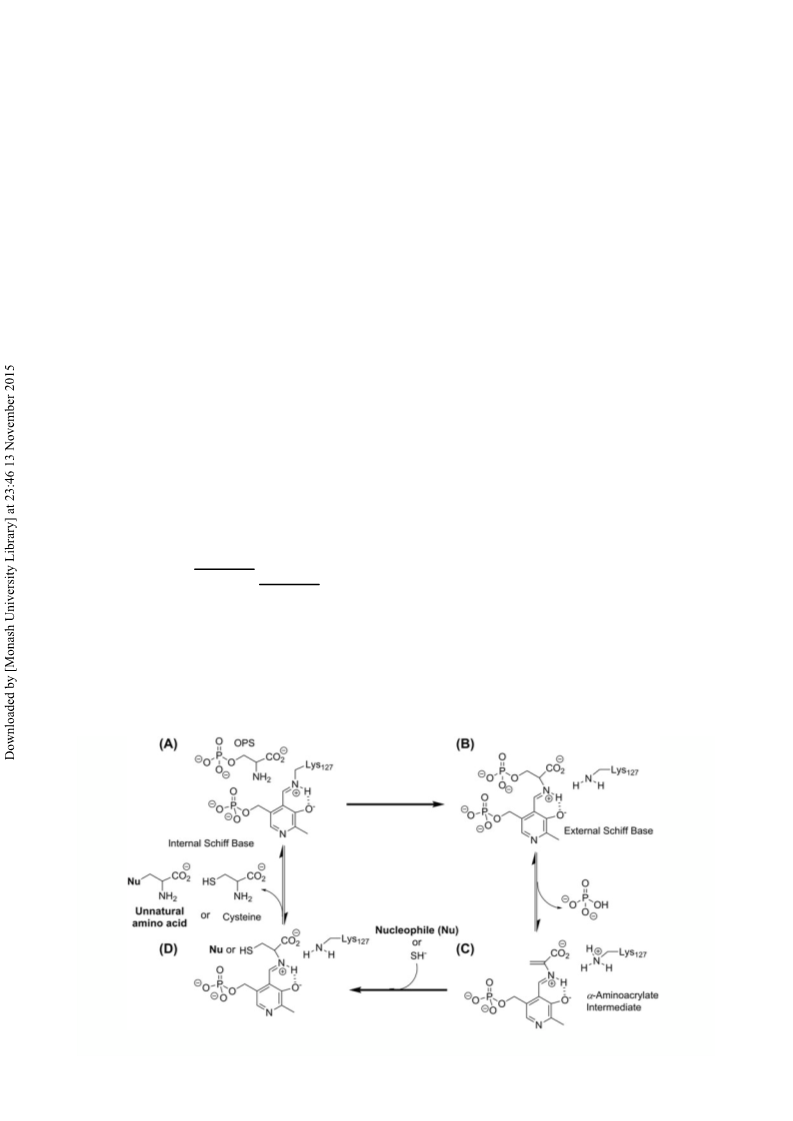
O-phospho-L-serine sulfhydrylase (OPSS) from archaeon Aeropyrum pernix K1 is able to synthesize L-cysteine even at 80 °C. In this article, we compared thermal stability and reactivity in organic solvent of OPSS with those of O-acetyl-L-serine sulfhydrylase B (OASS-B) from Escherichia coli. As a result, the thermostability of OPSS was much higher than that of OASS-B. Moreover, the activity of OPSS increased in the reaction mixture containing the organic solvent, such as N, N'-dimethyl formamide and 1,4-dioxane, whereas that of OASS-B gradually decreased as the content of organic solvent increased. From the crystal structural analysis, the intramolecular electrostatic interactions of N-terminal domain in OPSS seemed to be correlated with the tolerance of OPSS to high temperature and organic solvent. These results indicate that OPSS is more superior to OASS-B for the industrial production of L-cysteine and unnatural amino acids that are useful pharmaceuticals in the presence of organic solvent.
View More






Tai zhou world Pharm & Chem Co., Ltd
Contact:+86-576-85301198
Address:Rome 1001,wangjiang plaza,unti 2,jinshan east Road linhai,zhejiang,china
website:http://www.hope-chem.com
Contact:86-21-58090396-805
Address:Floor 4, Building 5, No.588 Tianxiong Road, Zhoupu International Medical Zone, ShangHai, China
Suzhou Ryan Pharmachem Technology Co.,Ltd
Contact:+86-0512-68780025
Address:B-301,No.2 Taishan Road,Suzhou New District,Jiangsu,P.R. China
Guangxi Nanning Guangtai Agriculture Chemical Co.,Ltd
Contact:+86-771-2311266
Address:Room703,Building12, Software Park Phase II,NO.68,Keyuan Road,Nanning City,Guangxi,China
Baoding City Light Industry And Textiles Imp.& Exp. Corp. Chemical Department.
Contact:86-312-3262436
Address:NO.658 CHAOYANG SOUTH STREET,BAODING CITY HEBEI CHINA
Doi:10.1016/0040-4039(96)00941-0
(1996)Doi:10.1089/105072503322401032
()Doi:10.1134/S0036024416090193
(2016)Doi:10.1016/j.ejmech.2016.12.037
(2017)Doi:10.1021/acs.organomet.8b00740
(2018)Doi:10.1016/S0040-4039(00)62013-0
(1966)